Atrezia anorectală, malformaţie izolată diagnosticată în primul trimestru de sarcină – prezentare de caz
Isolated anorectal atresia diagnosed in the first trimester – case report
Abstract
Anorectal atresia (ARA) is a congenital anomaly with an incidence of 1:1500 to 1:5000 in newborns. This malformation can be isolated, but is often associated with multiple congenital anomalies. Until recently, the diagnosis was often made in the third trimester of pregnancy or even after birth but, according to a review, it was established that prenatal diagnosis can be made even in the first trimester. Early diagnosis is essential for the management and planning of birth and postpartum treatment. We present the case of a fetus diagnosed with ARA at 12+0 weeks at the first-trimester ultrasound anomalies scan. The karyotype was performed and the result was normal. ARA was diagnosed and considered isolated since further detailed examinations performed in the second and third trimesters found no other anomalies. After birth, at 36 weeks, the diagnosis was confirmed as low anorectal atresia with perineal fistula. The newborn was scheduled for surgery at 3 months of life.Keywords
anorectal atresiaprenatal diagnosisisolated anomalyRezumat
Atrezia anorectală (ARA) este o anomalie congenitală cu o incidenţă de 1:1500 la 1:5000 de nou-născuţi. Această malformaţie poate fi izolată, dar este adesea asociată cu multiple anomalii congenitale. Până de curând, diagnosticul se făcea adesea în al treilea trimestru de sarcină sau chiar după naştere, dar în urma unui review s-a stabilit că diagnosticul prenatal se poate face chiar din primul trimestru. Diagnosticul precoce este esenţial pentru managementul şi planificarea naşterii şi a conduitei postnatale. Prezentăm cazul unui făt diagnosticat cu ARA la vârsta gestaţională de 12 săptămâni, în cadrul morfologiei de trimestrul întâi. S-a efectuat cariotipul fetal, iar rezultatul a fost normal. ARA a fost diagnosticată şi considerată izolată, deoarece în urma evaluărilor ecografice efectuate în al doilea şi al treilea trimestru de sarcină nu s-au constatat alte anomalii. După naştere, care a avut loc la 36 de săptămâni de gestaţie, s-a stabilit diagnosticul de atrezie anorectală joasă cu fistulă perineală. Nou-născutul a fost programat pentru intervenţie chirurgicală la vârsta de 3 luni de viaţă.Cuvinte Cheie
atrezie anorectalădiagnostic prenatalanomalie izolatăIntroduction
Anorectal atresia (ARA) is a congenital anomaly, with a significant incidence, of 1:1500 to 1:5000 in newborns(1,2). This malformation can be isolated, but is often associated with multiple congenital anomalies as part of the VACTERL syndrome (vertebral, anal, cardiovascular, tracheoesophageal, renal and limb)(3-5), complex genitourinary defects, caudal regression syndrome, or omphalocele-exstrophy-imperforate anus-spinal defects syndrome (OEIS)(3,4,6). ARA is the result of a deficient development of the urogenital septum, a mechanism where the distal rectal pouch is prevented from reaching the perineum. As a result of this mechanism, abnormal development of the perianal muscular complex also occurs(7). Until recently, the diagnosis was often made in the third trimester of pregnancy or even after birth(2) but, after a review, it was established that prenatal diagnosis can be made even in the first trimester(8). Early diagnosis is essential for the management and planning of birth and for the postpartum course of action. We present a case of a fetus diagnosed with ARA at 12+0 weeks, within the first-trimester morphology.
Case report
A 46-year-old primipara was referred to our center for a first-trimester anomaly scan at 12+0 weeks. The pregnancy was obtained after in vitro fertilization (IVF) with egg donation at the fourth attempt. No pre-implantation genetic diagnosis (PGD) was performed and the egg donor was 23 years old. The patient reported no drug consumption, non-smoking, antiplatelet medication, and vaginal progesterone substitution.
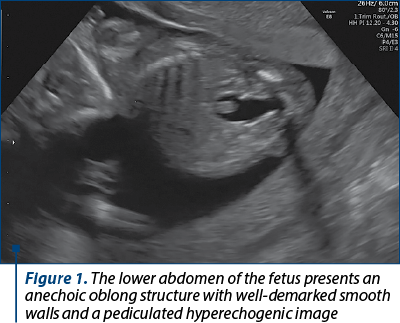
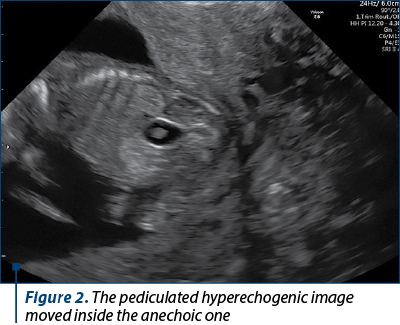
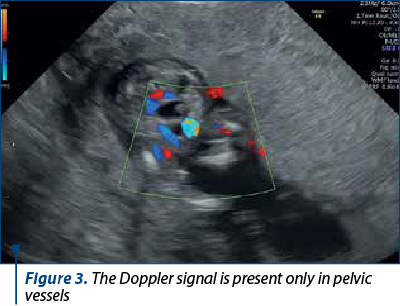
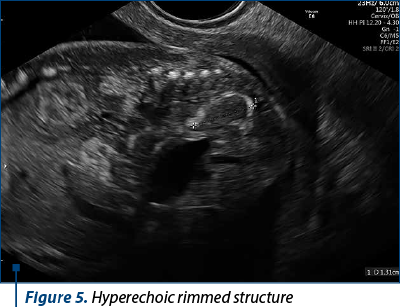
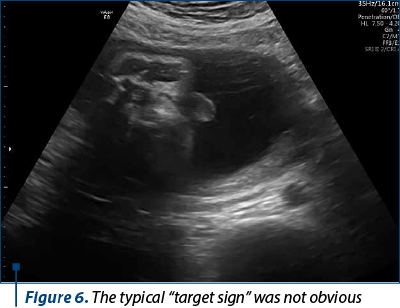
The scan revealed a slighter increased NT (3.2 mm), but no other soft markers for aneuploidy. At the lower abdominal level, mostly in the left quadrant, we noticed an anechoic oblong structure with well-demarked smooth walls, without a Doppler signal containing a pediculated hyperechogenic image that moved inside the anechoic one (Figures 1, 2, 3 and 4). The complex image was labeled as distal bowel dilatation after checking the complete separation from the neighboring structures (urinary bladder). Fetal sex was confirmed as male, and a thorough evaluation of the fetal anatomy was performed. No other anomalies were found. Anorectal atresia diagnosis was advanced. Considering the possible association with chromosomal anomalies, free fetal DNA test was recommended and it was repeated because of the inconclusive first result. The second result indicated a high risk (6/100) for trisomy 21. Amniocentesis with MCA was performed with a normal result (arr (1-22)x2,(XY)x1). Close follow-up of the fetus was conducted monthly. At the mid-trimester anomaly scan, the anechoic image disappeared, and a hyperechoic rimmed structure of 13 mm was found instead (Figure 5). No other anomalies were obvious. Later in pregnancy, even this image vanished. Considering the initial diagnosis, we evaluated the perianal muscular complex (PAMC). The examination was difficult due to the fetal position, but at 29 weeks the PAMC aspect was abnormal and the typical “target sign” was not obvious (Figure 6). From this consideration, we counseled the couple as having a very high likelihood of ARA, and pediatric surgery evaluation was scheduled.
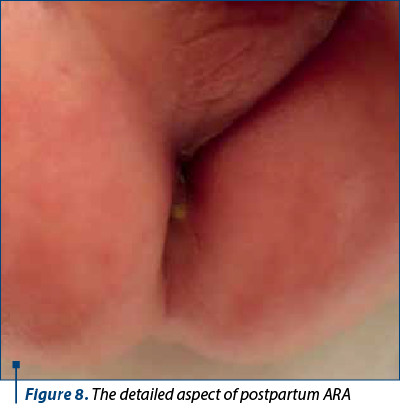
At 36 weeks, the baby was delivered by caesarean section, with 2770 g and a 9 Apgar score. Postnatal examination revealed anorectal atresia with a fistula at the perineal level, allowing meconium passage (Figures 7 and 8). The newborn underwent surgical evaluation with dilatation of the fistula and was scheduled for surgery at 3 months of life.
Discussion
Anal atresia occurs as an isolated congenital anomaly, with a reported frequency of 1.11 per 10,000 births in Europe(1). Anal atresia is more often associated with other congenital anomalies, such as VACTERL syndrome (vertebral defects, anal atresia, tracheoesophageal fistula, esophageal atresia, renal anomalies and limb malformations) or caudal regression syndrome(9-11). Prenatal diagnosis is based on the presence of a dilated fetal bowel on ultrasound(12). This diagnosis is commonly corroborated by the presence of further congenital anomalies since anal atresia rarely occurs as an isolated anomaly(9). In a cohort study of patients with ultrasound consistent anal atresia, Harris et al. confirmed the diagnosis either at birth or at fetal autopsy. Among them, 11 of 12 patients (92%) with anal atresia had abnormalities associated with either VACTREL syndrome or caudal regression syndrome(13). In our case, the abnormality was isolated.
It is well known that anorectal anomaly is more frequent in male fetuses(14), and in our case the fetus was male.
The etiology of ARA appears to be multifactorial. Chromosomal aberrations have been reported in only one case of Down syndrome. Genetic mutations involved in this condition are rare(15). In our case, it was assumed that the abnormality was related to the assisted reproductive procedure.
The diagnosis of ARA may be difficult in the first trimester when the diagnosis is based only on indirect ultrasound signs. ARA has a low prenatal detection rate of only 16%(2). In a 10-year study on 56 children, prenatal diagnosis of ARA by MRI versus ultrasound could not be established below 20 weeks of gestation(16).
Fourteen cases have been reported in the literature in which the diagnosis was suspected following visualization of fetal intraabdominal or pelvic cystic structures(17-20). Cystic structures are represented by distended intestines filled with fluid of undetermined origin. The accumulation of fluid can be explained by the fact that the intestinal mucosa is not capable of absorption in early gestation and the rectum ends in the fundus(21).
The most common form of these structures is tubular, which is determined by associated anomalies (rectovesical fistula, megacystis, complex cloacal anomalies). A tubular structure was also the defining feature in our case.
The presence of intracystic hyperechogenic structures may suggest the existence of intestinal disorders due to enterolithiasis(7). In our case, the hyperechogenic structure was interpreted as an image produced by peristalsis.
The pregnancy assessment and management of a cystic abdominal tubular formation in a fetus at 12 weeks of gestation should take into consideration the differential diagnosis(22), including the possibility that bowel dilatation is a transient finding(23). Cystic transient images in pregnancy with spontaneous resolution in the second and third trimesters are reported in the literature, particularly if the right colon is affected(21). In our case, the anechogenic image faded in the second trimester. It is thus recommended that serial sonographic examinations and perhaps other tests, including karyotyping, be offered to try to exclude other significant malformations. According to a review by Pleş et al., the appearance of abnormalities on later examination was reported not only as transient findings, but also as a change in echogenicity from anechoic to hyperechoic structures in two cases(8).


Conclusions
The diagnosis of anorectal atresia in the prenatal period is clinically important because prenatal diagnosis allows the planning of birth and the postnatal management. This case demonstrates the importance of maintaining a high clinical suspicion for anal atresia in a fetus with intraabdominal tubular anechogenic imaging in the first trimester and monitoring with serial ultrasound to establish a diagnosis of certainty.
Conflict of interests: The authors declare no conflict of interests.
Bibliografie
-
Cuschieri A, EUROCAT Working Group. Anorectal anomalies associated with or as part of other anomalies. Am J Med Genet. 2002;110:122–30.
-
Brantberg A, Blaas HG, Haugen SE, Isaksen CV, Eik-Nes SH. Imperforate anus: a relatively common anomaly rarely diagnosed prenatally. Ultrasound Obstet Gynecol. 2006;28(7):904–10.
-
Cho S, Moore SP, Fangman T. One hundred three consecutive patients with anorectal malformations and their associated anomalies. Arch Pediatr Adolesc Med. 2001;155(5):587–91.
-
Stoll C, Alembik Y, Dott B, Roth MP. Associated malformations in patients with anorectal anomalies. Eur J Med Genet. 2007;50(4):281–90.
-
Black CT, Sherman JO. The association of low imperforate anus and Down’s syndrome. J Pediatr Surg. 1989;24(1):92–4.
-
Mallmann MR, Reutter HM, Geipel A, Berg C, Gembruch U. Early prenatal diagnosis of the OEIS complex with different appearance in early compared with late pregnancy before spontaneous rupture of the cloacal membrane. Prenat Diagn. 2014;34(8):803–5.
-
Ochoa JH, Chiesa M, Vildoza RP, Wong AE, Sepulveda W. Evaluation of the perianal muscular complex in the prenatal diagnosis of anorectal atresia in a high-risk population. Ultrasound Obstet Gynecol. 2012;39(5):521–7.
-
Pleş L, Chicea R, Poenaru MO, Neacşu A, Sima RM, Micu R. Can anorectal atresia be diagnosed in the first trimester of pregnancy? A systematic literature review. Medicina (Kaunas). 2020;56(11):583.
-
Quan L, Smith DW. The VATER association: vertebral defects, anal atresia, tracheoesophageal fistula with esophageal atresia, radial dysplasia. Birth Defects. 1972;8(2):75-8.
-
Nora RH, Nora JJ. A syndrome of multiple congenital anomalies associated with teratogenic exposure. Arch Environmental Health. 1975;30(1):17-21.
-
Ladd WE, Gross RF. Congenital malformations of the anus and rectum. Report of 162 cases. Am J Surg. 1934;23(1):167-83.
-
Bean WJ, Calonje MA, April, CN, Geshner J. Anal atresia: a prenatal ultrasound diagnosis. J Clin Ultrasound. 1978;6(2):111-2.
-
Huppert BJ, Brandt KR, Ramin KD, BF. King. Single-shot fast spin-echo MR imaging of the fetus: a pictorial essay. Radiographics. 1999;19 Spec No:S215-27.
-
Murken JD, Albert A. Genetic counselling in cases of anal and rectal atresia. Prog Pediatr Surg. 1976; 9:115–8.
-
Zwink N, Jenetzky E, Brenner H. Parental risk factors and anorectal malformations: Systematic review and meta-analysis. Orphanet J Rare Dis. 2011;6:25.
-
Rohrer L, Vial Y, Gengler C, Tenisch E, Alamo L. Prenatal imaging of anorectal malformations - 10-year experience at a tertiary center in Switzerland. Pediatr Radiol. 2019;50(1):57–67.
-
Carroll SG, Hyett J, Eustace D, Seller MJ, Nicolaides KH. Evolution of sonographic findings in a fetus with agenesis of the urethra, vagina, and rectum. Prenat Diagn. 1996;16(10):931–3.
-
Correia P, Moreira M, Rodrigues M, Inocêncio G. Prenatal diagnosis of vesicorectal fistula. Ultrasound Q. 2017;33(1):112–4.
-
Gilbert CE, Hamill JK, Metcalfe RF, Smith P, Teele RL. Changing antenatal sonographic appearance of anorectal atresia from first to third trimesters. J Ultrasound Med. 2006;25(6):781–4.
-
Lam YH, Shek T, Tang MHY. Sonographic features of anal atresia at 12 weeks. Ultrasound Obstet Gynecol. 2002;19(5):523–4.
-
Liberty G, Bardin R, Gilboa Y, Tover A, Mashiach R, Mazaki E, Shen O. First trimester expression of anorectal malformation: Case report and review of the literature. J Clin Ultrasound. 2018;46(9):591–7.
-
Taipale P, Heinonen K, Kainulainen S, Seuri R, Heinonen S. Cloacal anomaly simulating megacystis in the first trimester. J Clin Ultrasound. 2004;32(8): 419–22.
-
Bronshtein M, Gover A, Beloosesky R, Blumenfeld Z. Transient distention of right posterior located sigma, a new sonographic sign for the prenatal diagnosis of anal atresia. J Clin Ultrasound. 2016;45(3):160–2.





