Managementul durerii episodice intense la pacienţii oncologici
Management of breakthrough pain in cancer patients
Abstract
Background. Breakthrough pain reflects a transitory exacerbation of pain that breaks through chronic pain (background, or persistent, or basal pain) which is controlled by around-the-clock medication (opioids).Materials and method. We assessed 107 patients treated with strong opioids (oxycodone) for baseline pain. Thirty-three patients needed the second opioid for breakthrough pain. We evaluated the pain intensity in patients who presented breakthrough pain, before and after the initiation of the treatment.
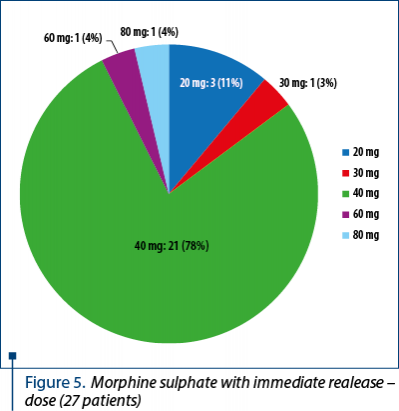
Results. The patients’ distribution according to the dose of strong opioids with immediate release, per day, for breakthrough pain, was the following: morphine 20 mg – 2 patients, 40 mg – 3 patients; morphine sulphate tablets immediate release 20 mg – 3 patients, 30 mg – 1 patient, 40 mg – 21 patients, 60 mg – 1 patient, 80 mg – 1 patient; fentanyl tablets with immediate release – 1 patient.
Discussion and conclusions. Basal pain lasts more than 12 hours per day and is controlled by the around-the-clock medication (opioids). The assessment of breakthrough pain is very important (physical examination, evaluation of the extent of the disease, pain history, pain intensity, temporal features, and interference with activity). Breakthrough pain has an important prevalence and a negative impact on the quality of life of cancer patients. Managing breakthrough pain leads to an improved quality of life, which has positive outcomes for patients. These outcomes will be reflected in their physical and psychological symptoms, and also in their social life.
Keywords
cancerbreakthrough painstrong opioidsRezumat
Introducere. Durerea episodică intensă reflectă o exacerbare tranzitorie a durerii, în cazul unor pacienţi care suferă deja de durere cronică (persistentă sau bazală), care este controlată de medicaţia de 24 de ore (opioide).Materiale şi metodă. Am evaluat 107 pacienţi trataţi cu opioide puternice (oxicodonă) pentru durerea de bază. Treizeci şi trei de pacienţi au avut nevoie de cel de-al doilea opioid pentru durerea episodică intensă. Am evaluat intensitatea durerii la pacienţii care au prezentat durere episodică intensă, înainte şi după iniţierea tratamentului.
Rezultate. Distribuţia pacienţilor în funcţie de doza de opioizi puternici, cu eliberare imediată, pe zi, pentru durerea episodică intensă, a fost următoarea: morfină 20 mg – doi pacienţi, 40 mg – trei pacienţi; comprimate de sulfat de morfină cu eliberare imediată 20 mg – trei pacienţi, 30 mg – un pacient, 40 mg – 21 de pacienţi, 60 mg – un pacient, 80 mg – un pacient; fentanil cu eliberare imediată – un pacient.
Discuţii şi concluzii. Durerea bazală durează mai mult de 12 ore pe zi şi este controlată de medicaţia de 24 de ore (opioide). Evaluarea durerii episodice intense este foarte importantă (examinare fizică, evaluarea amplorii bolii, istoricul ei, intensitatea durerii, trăsăturile temporale şi interferenţa cu activitatea). Durerea episodică intensă are o prevalenţă importantă şi un impact negativ asupra calităţii vieţii pacienţilor cu cancer. Gestionarea durerii episodice intense conduce la o îmbunătăţire a calităţii vieţii, având rezultate pozitive pentru pacienţi. Aceste rezultate se vor reflecta în simptomele lor fizice şi psihologice, precum şi în viaţa lor socială.
Cuvinte Cheie
cancerdurere episodică intensăopioide puterniceBackground
Basal pain lasts more then 12 hours per day and is controlled by the around-the-clock medication (opioids). A pain which is controlled adequately means a pain with a score of 0-4 on the Numerical Rating Scale (0 meaning no pain, and 10 meaning the more severe pain possible)(1).
The original definition of breakthrough pain (BTP) was given by Portenoy and Hagen in 1989: “a transitory increase in pain to greater than moderate intensity on a base-line pain of moderate intensity or less”(2).
In 2002, an expert working group of the European Association for Palliative Care recommended the term “episodic pain” or “transient pain”, because the English term “breakthrough pain” has no translation in other languages(3).
Breakthrough pain reflects a transitory exacerbation of pain which occurs in a patient with a controlled baseline pain, and that can be caused by different or the same stimuli of baseline pain(4). It breaks through chronic pain (basal, background or persistent pain), which is controlled by around-the-clock medication (opioids).
Materials and method
We assessed 107 patients treated with strong opioids, admitted at “St. Luke” Chronic Disease Hospital, Department of Chronic Oncology – Palliative Care, Bucharest, between 1st of January and 1st of July 2017.
We initiated strong opioids treatment (oxycodone – slow-release tablets) for severe pain in these patients.
Thirty-three patients (33.84%) had also breakthrough pain, being necessary the second opioid (morphine/ morphine sulphate tablets with immediate release/ fentanyl sublingual tablets with immediate release).
We evaluated the patients’ pain intensity using the Edmonton Symptom Assesssment System (ESAS) at the initiation of breakthrough pain treatment and after one week of treatment.
Results
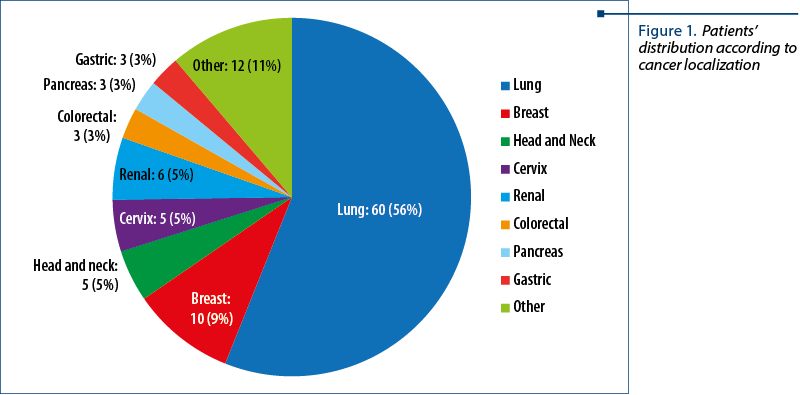
The patients were between 42 and 83 years old. The patients’ distribution according to cancer diagnosis was: lung – 60 patients; breast – 10 patients; renal – 6 patients; head and neck – 5 patients; cervix – 5 patients; colorectal – 3 patients; gastric – 3 patients; pancreas – 3 patients; other – 12 patients (Figure 1).
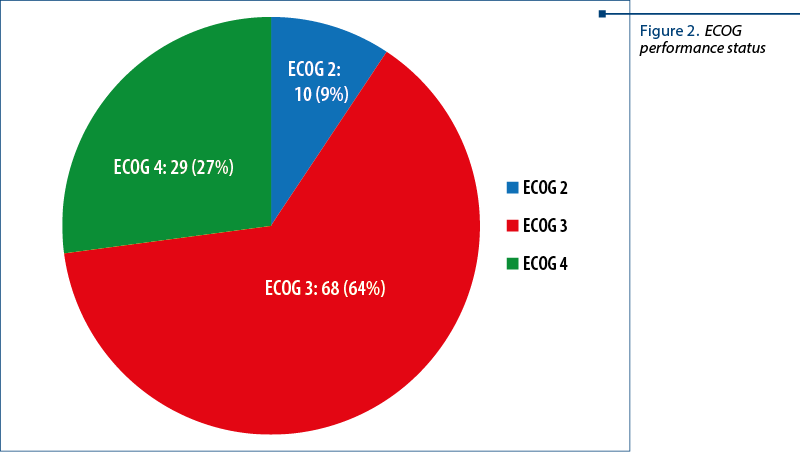
The patients’ distribution according to ECOG performance status was: ECOG 2 – 10 patients; ECOG 3 – 68 patients; ECOG 4 – 29 patients (Figure 2).
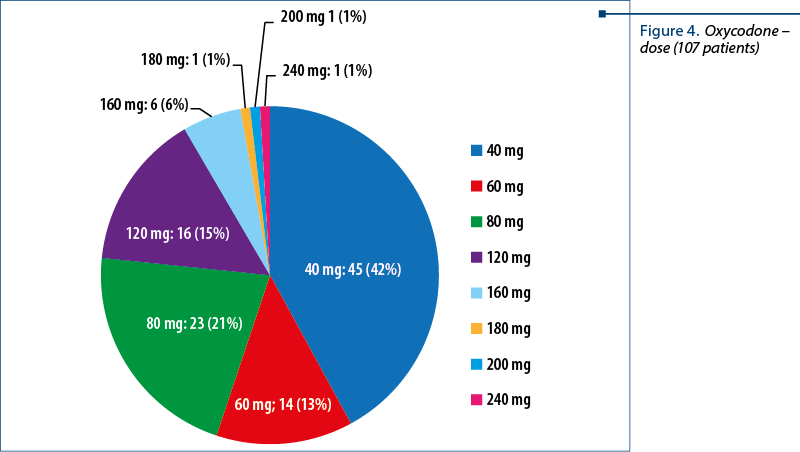
The patients’ distribution according to the oxycodone dose per day was: oxycodne 40 mg – 45 patients; 60 mg – 14 patients; 80 mg – 23 patients; 120 mg – 16 patients; 160 mg – 6 patients; 180 mg, 200 mg and 240 mg – one patient each (Figures 3 and 4).
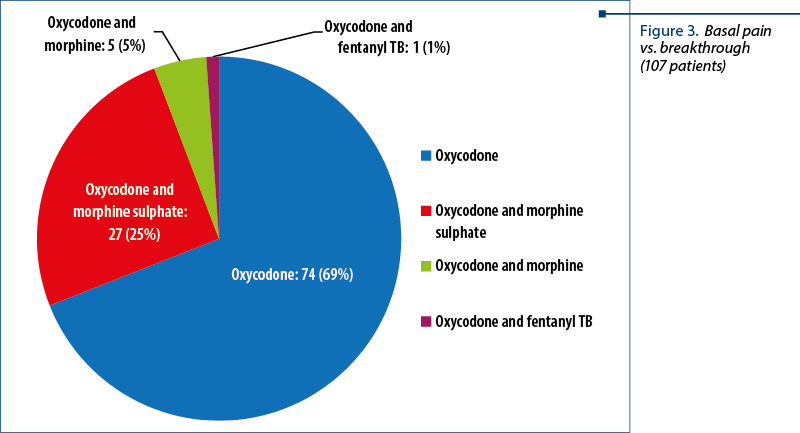
The patients’ distribution according to the dose of strong opioids immediate release, per day, for breakthrough pain is presented in Figures 4 and 5:
- morphine 20 mg – 2 patients
- morphine 40 mg – 3 patients
- morphine sulphate, tablets with immediate release 20 mg – 3 patients
- morphine sulphate, tablets with immediate release 30 mg – 1 patient
- morphine sulphate, tablets with immediate release 40 mg – 21patients
- morphine sulphate, tablets with immediate release 60 mg – 1 patient
- morphine sulphate, tablets with immediate release 80 mg – 1 patient
- fentanyl, tablets with sublingual immediate release – 1 patient.
The intensity of breakthrough pain was severe in 5 patients and moderate in 28 patients at the initiation of the treatment. After one week of treatment, the intensity of pain was moderate in 5 patients and mild in 28 patients.
Discussion
The prevalence of breakthrough pain ranges from 40% to 80% worldwide(4). In our study, the prevalence was 33.84%. The assessment of breakthrough pain is very important (physical examination, evaluation of the extent of the disease, pain history, pain intensity, temporal features, and interference with activity).
Breakthrough pain can be predictable (associated with movement), unpredictable (not associated with events or activity), or both(5). The physiopathology of breakthrough pain can be somatic, visceral, neuropathic or mixed(6).
The characteristics of this type of pain are: 3-5 episodes per day, with a medium duration of 30-60 minutes, and a time to peak severity of about 3-15 minutes(7).
Usually, breakthrough pain is treated with oral immediate-release opioids. Generally, the dose of oral immediate-release opioids begins with 10% of the daily dose of the opioid and should be titrated according to the effect.
The opioids dose cannot be predicted by basal analgesic dose.
An episode of breakthrough pain of mild intensity can appear in a patient with a high level of basal pain and, at the same time, an episode of breakthrough pain of high intensity can appear in a patient with low level of basal pain(8-11). There is no relationship between the dose of medication for breakthrough pain and the total daily dose for basal pain(12,13). The patients will receive the dose according to the intensity of breakthrough pain episode.
Meanwhile, patients can have episodes of breakthrough pain with different intensity, and also with different causes in the same day.
Examples of treatments for cancer breakthrough pain are: oral agents (immediate-release morphine, methadone), i.v. or s.c. agents (morphine, hydromorphone), sublingual agents (fentanyl), as we presented in our study.
Managing breakthrough pain, the general status and the quality of life of our patients became better, with positive outcomes. These outcomes will be reflected in their physical and psychological symptoms, and also in their social life.
Conclusions
Breakthrough pain has an important prevalence and a negative impact on the quality of life of cancer patients.
A very important aspect was that each patient needed an individualised treatment for his specific needs, with attention to the details. In the future, more studies are needed for improving the recognition and assessment of breakthrough pain, and also for pharmacologic approaches in order to treat it, this aspect being very important in cancer pain management.
Conflict of interests: The authors declare no conflict of interests.
Bibliografie
- Mercadante S. Managing breakthrough pain. Curr Pain Headache Rep. 2011; 15:244-249.
- Portenoy RK, Hagen NA. Breakthrough pain: definition and management. Oncology (Williston Park). 1989; 3(8 Suppl):25-29.
- Mercadante S, Radbruch L, Caraceni A, et al. Episodic (breakthrough) pain: consensus conference of an expert working group of the European Association for Palliative Care. Cancer. 2002; 94:832–839.
- Hagen NA, Biondo P, Stiles C. Assessment and management of breakthrough pain in cancer patients: Current approaches and emerging research. Current Science Inc. 2008; 12: 241. https://doi.org/10.1007/s11916-008-0042-1
- Davies A, Zeppetella G, Andersen S, et al. Multi-centre European study of breakthrough cancer pain: pain characteristics and patient perceptions of current and potential management strategies. Eur J Pain. 2011;15:756-763.
- Portenoy RK, Hagen NA. Breakthrough pain: definition, prevalence and characteristics. Pain. 1990; Jun;41(3):273-81.
- Taylor DR. Managing Cancer Breakthrough Pain. Springer Healthcare Communication. 2013; DOI: 10.1007/978-1-908517-83-8_2.
- Davies AN, Dickman A, Reid C, Stevens AM, Zeppetella G; Science Committee of the Association for Palliative Medicine of Great Britain and Ireland. The management of cancer-related breakthrough pain: recommendations of a task group of the Science Committee of the Association for Palliative Medicine of Great Britain and Ireland. Eur J Pain. 2009; 13:331-338.
- Davies A, Zeppetella G, Andersen S, et al. Multi-centre European study of breakthrough cancer pain: pain characteristics and patient perceptions of current and potential management strategies. Eur J Pain. 2011; 15:756-763.
- Green CR, Montague L, Hart-Johnson TA. Consistent and breakthrough pain in diverse advanced cancer patients: a longitudinal examination. J Pain Symptom Manage. 2009; 37:831-847.
- Davies AN, Vriens J, Kennett A, McTaggart M. An observational study of oncology patients’ utilization of breakthrough pain medicine. J Pain Symptom Manage. 2008;35:406-411.
- Portenoy RK, Payne D, Jacobsen P. Breakthrough pain: characteristics and impact in patients with cancer pain. Pain. 1999;81:129-134.
- Christie JM, Simmonds M, Patt R, et al. Dose-titration, multicenter study of oral transmucosal fentanyl citrate for the treatment of breakthrough pain in cancer patients using transdermal fentanyl for persistent pain. J Clin Oncol. 1998; 16:3238-3245.
Melanomul malign
Bogdan Mircea Măciuceanu-Zărnescu
Se prezintă un caz tipic de melanom cutanat, localizat la nivelul toracelui posterior. Se descrie protocolul chirurgical al Secţiei de chirurgie plastică de la Spitalul de Urgenţă Floreasca Bucureşti pentru acest tip de cancer. Rezultatul biopsiei (examenul histologic) relevă un melanom malign la care se prec...Rolul nutriţiei în cancer
Lucian MIRON
Factorii alimentari joacă un rol complex în etiopatogeneza cancerelor umane. Se apreciază că dieta, inactivitatea fizică şi obezitatea sunt responsabile pentru producerea a cel puţin 35% din cancerele umane. Aproximativ un sfert din aceste tipuri de cancer apar în ţările cu un nivel socioeconomic scăzut, unde...A fost aprobat un nou tratament pentru hipertensiunea arterială
Cristina Ghioca
Administrația Americană pentru Alimente și Medicamente a aprobat un nou medicament pentru gestionarea hipertensiunii arteriale necontrolate sau rezistente la tratament la pacienții aflați deja sub tratament pentru această afecțiune.
...


