Impactul factorilor de mediu asupra patologiei respiratorii la copil
Impact of environmental factors on the airway pathology in children
Abstract
The exposure to environmental factors is incriminated not only in the increase of the prevalence of allergic diseases, but also in their severity. Most of the air pollutants have an action similar to that of air allergens in asthma and allergic rhinitis. Children are particularly vulnerable to air pollutants due to age-related anatomical, physiological and immunological characteristics. The authors review the main outdoor and indoor pollutants, highlighting their impact on the respiratory tract of the healthy or chronically ill children. Despite all the efforts made at national level, pollution in our geographical region and especially in urban areas is increasing, as reflected in the growing prevalence of pediatric allergic diseases. A multidisciplinary approach is necessary in order to promote specific preventive and curative measures to reduce morbidity related to allergic diseases, school absenteeism, and to improve the child and family’s quality of life.Keywords
air pollutionchildrenrespiratory tractRezumat
Expunerea la factorii de mediu este incriminată nu numai în creşterea prevalenţei bolilor alergice, dar şi în severitatea acestora. Majoritatea poluanţilor atmosferici au o acţiune similară cu cea a aeroalergenilor în astmul bronşic şi în rinita alergică. Copiii sunt deosebit de vulnerabili la poluanţii atmosferici din cauza caracteristicilor anatomice, fiziologice şi imunologice legate de vârstă. Autorii examinează principalii poluanţi exteriori şi interiori, subliniind impactul lor asupra tractului respirator al copiilor sănătoşi sau cu boli cronice. În ciuda tuturor eforturilor depuse la nivel naţional, poluarea în regiunea noastră geografică şi în special în zonele urbane este în creştere, aceasta reflectându-se în prevalenţa crescândă a bolilor alergice pediatrice. O abordare multidisciplinară este necesară pentru a promova măsuri preventive şi curative specifice de reducere a morbidităţii prin boli alergice, de scădere a absenteismului şcolar şi pentru a îmbunătăţi calitatea vieţii copilului şi a familiei.Cuvinte Cheie
poluarea aeruluicopiitract respirator1. Introduction
The prevalence of allergic diseases has grown considerably over the past few decades, reaching approximately 15% to 35%(22). Population studies for large groups particularly vulnerable have demonstrated an overall prevalence of allergic disorders between 25% and 30%: 15-20% for atopic dermatitis, 7-10% for bronchial asthma, and 15-20% for allergic rhinitis and conjunctivitis(15). The “endemic” raising of allergic disorders is explained by the appearance of some new risk factors, of which we were unaware in the past, either nutritional or environmental (maternal smoking, atmospheric pollution, certain viral strains). There was also a reduction in the “traditional lifestyle” protection factors: contact with domestic animals, the presence of forests and green spaces (in urban areas), increased number of family members, collectivity attendance, certain infections early contracted (viral or with mycobacterias)(17). Other theories incriminate the reduction of consumption of animal fats which contain omega-3 polyunsaturated fatty acids (w-3 PUFA) and increased intake of margarine and vegetable oils rich in omega-6 polyunsaturated fatty acids (w-6 PUFA), along with decreased consumption of fruits and vegetables rich in powerful antioxidants such as b-carotene or vitamin C, and vitamin D deficiency.
The exposure to environmental factors, as imposed by urbanization, which is a feature of the “Western lifestyle”, is a concern not only due to the increasing prevalence of airway disorders, but also due to their severity. In accordance to European Environmental Agency, the major changes of the environment with great influence on the health state of the population, in particular the elderly, young children or people with existing chronic respiratory or cardiac diseases, are: ozone layer destruction, degradation of oceans and continental waters, the emergence of smog, changes in flora and fauna biodiversity, climate changes and the high pollution of the urban environment(10).
Climate change may aggravate respiratory diseases evolution by increasing concentration of pollens, carbon dioxide, ozone and particulate substances and/or molds proliferation(10). The average global temperature has increased by 0.6±0.2°C since the late 19th century and is expected to increase by 1.1-6.4°C by the end of the 21st century. The overall increase of temperature and the extreme variations, as well as the extensive use of fertilizers are responsible for increasing the amount of pollens in the ambient air and the quantity of allergic proteins they contain, changing their geographic distribution(7). An increase in atmospheric concentration of carbon dioxide correlates directly proportional with pollen seasonal extension, which may represent a trigger for the bronchial asthma and allergic rhinitis exacerbations(6). Last but not least, there is evidence for a positive correlation between school absenteeism and the levels of atmospheric pollutants, especially PM10, ozone, carbon monoxide and sulphur dioxide.
2. Air pollutants – general data
Pollutants from the environment are substances with harmful effects on living organisms, responsible for increased mortality and morbidity, even from intrauterine life. Air pollutants are derived from natural and human activities. Over time, the chemical composition of air pollutants has significantly changed, as well as their physical properties, ability to persist in the atmosphere and the impact on the population health status(14). For this reason, these pollutants will be included in the list of risk factors for fetal epigenetic reset (DNA modifications), with severe consequences, inducing diseases and death in intrauterine life.
The major pollutants which may come from outside sources or from inside areas are represented by: ozone, nitrogen dioxide, particles of Diesel combustion and cigarette smoke.
The atmospheric pollutants can be grouped into four categories: gaseous substances (sulphur dioxide, ozone, nitrogen oxide and volatile organic compounds with a great contribution in the variations of atmospheric air composition), persistent organic pollutants (dioxins), heavy metals (mercury, lead) and particulate substances. The effects of pollutants on health status depend on their composition, the dose and the type of exposure and, not ultimately, on their concentration(4). Pregnant women’s exposure to heavy metals – in particular, lead – increase the risk of spontaneous abortion and may delay the intrauterine growth (premature birth and low birth weight), which can lead to congenital malformations and central nervous system injuries responsible for disabilities and cognitive impairments.
All types of pollutants can affect the respiratory tract health, especially in children and elderly people. There was demonstrated the existence of individual susceptibility factors, such as age, nutritional status, existing diseases, particularly respiratory and cardiovascular disorders, but also genes’ polymorphisms involved in tissue detoxification and repair processes(14).
Children are more susceptible to air toxicants’ action, especially in the first years of life, when the respiratory system is completing its growth and maturation. There is strong evidence that they exert harmful effects even in the fetal period. Maternal exposure to elevated levels of pollutants during pregnancy may cause early loss of conception, fetal growth restriction, birth defects, preterm delivery or small-for-gestational-age status, depending on the age of gestation.
After birth, the extreme vulnerability of children to air pollutants is favoured by some age-related particularities: small-size airways, “physiological” immune immaturity, the inefficiency of metabolic and detoxification mechanisms, and the increased incidence of respiratory infections. It is also true the fact that, in general, children are more active than adults and spend more time outside, especially during the day, when the air is more polluted(19).
Pollutants are generally non-allergenic by themselves, but they are able to exacerbate the allergic diseases through well-documented interferences with the synthesis of immunoglobulins, cytokines and inflammatory mediators. Many studies have demonstrated that air pollutants are associated with an increased allergic type 2 helper T (Th2) cell pattern, similar to the immune reactions found in allergic rhinitis or asthma(22). Although short-term effects are widely accepted, there are very few studies regarding the possibility to initiate asthma.
The possible pathogenic mechanisms by which air pollutants aggress the airway mucosa are still a matter of debate in literature. Among those mechanisms, there could be mentioned: airway inflammation (particulate substances, ozone), free radical generation by oxidative stress (heavy metals, organic chemicals), covalent changes of intracellular proteins (enzymes), alteration of immune adaptive responses (endotoxins, glicans), nociception receptors stimulation and autonomic nervous system activation, procoagulant action of ultrafine particles which could penetrate the systemic circulation, alveolar macrophages’ suppression, and increased susceptibility to viral infections(7).
3. Major air pollutants with negative impact on child’s respiratory tract
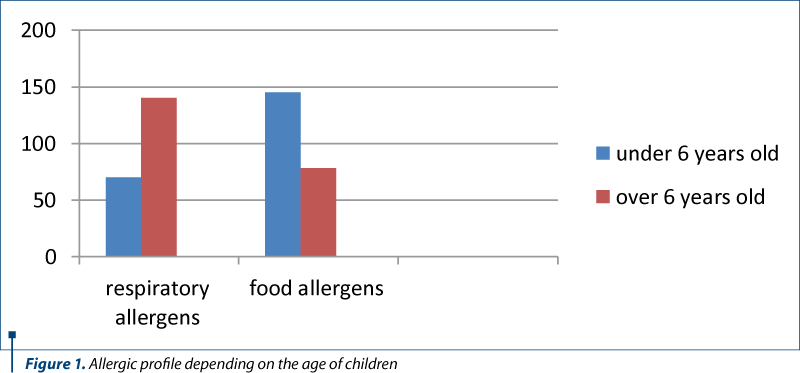
In 2009, in the 2nd Pediatrics Clinic of the “Sf. Maria” Emergency Clinical Hospital for Children in Iaşi, we performed a retrospective study on 423 children diagnosed with different allergic diseases, based on the international criteria. Specific serum IgE levels performed (for the most frequent 20 allergens) in 300 cases, along with patient’s history and clinical features, made the diagnosis more precise and suggested the severity of the allergic diseases. Polysensitizations were more frequent, especially for respiratory allergens, and monosensitization was more common under 6 years old (especially for dust and cow’s milk). Respiratory allergy (asthma +/- allergic rhinitis) had been also correlated with polysensitization (Figure 1). Some of the allergic diseases may have been influenced by environmental factors described thereafter.
3.1. Diesel combustion particles
Diesel combustion produces particulate substances, nitrogen oxide and ozone precursors. Diesel particles are an important source of particulates in urban areas. The effects on mucous membranes of respiratory tract are dependent on their size. The most harmful substances are fine particles, those less than 10 µm in diameter (PM10 – particulate matter 10), which are considered inhalable, and smaller particles, those less than 2.5 µm in diameter (PM 2.5), which are considered respirable. The latter can contain toxic products of combustion, metals (iron, cobalt, copper, zinc), allergens, and can be retained in the lung parenchyma, causing alveolar inflammation, changes in blood coagulability and free radicals’ generation. The high vulnerability of asthmatic airways is clinically reflected by decreased respiratory functional parameters, increased number of exacerbations, more days of hospitalization and admissions in the emergency units, along with a declining lung function.
These effects could be explained by the oxidative potential of PM and, thus, the ability to induce bronchial reactivity via reactive oxygen species. The variations in individual consequences of exposure to the same concentrations of particulate substances seem to have a genetic basis. For example, genetic polymorphism of the glutathione S-transferase (GSTP1, GSTM1, GSTT1), found in up to 50% of the population, is involved in the defence mechanisms against oxidative stress, thus facilitating the emergence of allergic diseases in children(14). The supplementation of diet with vitamins having antioxidant properties – for example, vitamin C and/or vitamin E – can attenuate this configurable genetics.
Experimental studies have demonstrated an increased number of neutrophils in sputum and bronchial mucosa, as well as an increased concentration of interleukin-6, interleukin-8 and growth-related oncogene-a in patients whose residence is near areas with intense road traffic. Other studies also showed that concomitant exposure to Diesel particles and a food or air allergen could lead to a more rapid and prominent allergic response than that seen in allergen exposure alone(5).
Chronically aggressed airway vulnerability correlates with increased frequency and severity of viral infections, triggering and/or aggravating the respiratory allergy. Particulate substances contribute to spatial transport of respiratory allergens and increase their biodisponibility, roles favoured by weather changes (temperature, air flows, humidity)(9).
3.2. Ozone
Ozone is a more powerful oxidant than nitrogen dioxide. It is the main pollutant of the atmosphere in the highly industrialized regions(13). The harmful effects are expressed at the level of both proximal, as well as small airways(14). Experimental studies have demonstrated that the exposure to ozone determines progressive remodelling processes of respiratory tract characterized by hyperplasia and hypertrophy of the epithelial cells, but also by progressive interstitial fibrosis(12). These effects are most important for individuals with a specific configurable genetics: alpha-TNF gene polymorphism increases the bronchoconstriction response after the exposure to sulphur dioxide and ozone(8).
Exposure to ozone of healthy individuals can be followed by slight symptoms (thoracic discomfort), but also objective signs (decreased FEV1, nonspecific bronchial responsiveness), especially if the exposure is concomitant with physical effort. These effects are felt more intensely in children with intermittent asthma compared with those with persistent asthma who seem to be protected by inhaled corticosteroid therapy. In asthmatic patients, the concomitant exposure to ozone and air allergens is associated with an increased intensity of IgE-mediated hypersensitivity reactions, reflected by an increased number of eosinophils in sputum.
Indoor pollution is responsible for more than 1.6 million deaths annually, more than half of them occurring in children under the age of 5 years old, in geographical areas where the climate changes are important(16).
3.3. Cigarette smoke
Cigarette smoke is the most common pollutant of dwellings. Toxic components can be found in both gas phases, as well as in the form of particles. Particulate substances include microelements, tar, catechol, polynuclear hydrocarbon, phenols, which are considered to be carcinogenic, and also nicotine, indole and carbazole. Carbon monoxide, formaldehyde, nitrosamines and nitrogen oxide with carcinogenetic action are found in gaseous form(23).
The negative effects of smoking on the baby begin in the womb. Maternal smoking is an important risk factor for pathologies such as intrauterine growth retardation, placenta praevia, placental abruption, premature birth, low birth weight, sudden death, difficult adaptation to extrauterine life, and difficulty in breathing at birth. There are also studies which showed that there is an association between smoking and the occurrence of congenital malformations: cleft lip and palate, organ malformations (by mutagenic effects directly on the DNA of the components of cigarette smoke)(3,21).
After birth, the exposure of the infant and child to cigarette smoke is an important risk factor for the development of chronic lung pathologies (asthma, chronic respiratory diseases such as chronic respiratory failure, allergic polysensitization, malnutrition, various nutritional deficiencies, progressive long-term respiratory damage), behavioral disorders, sleep disorders etc.
There are various types of smoking:
active (first-hand smoke) – direct use of products (classic cigarettes, electronic cigarettes, heated tobacco, other tobacco products);
passive (second-hand smoke) – exposure of the child (fetus, newborn, infant, child) to cigarette smoke or directly to products derived from it;
third-hand smoke – exposure of the child to previously tobacco-exposed products (environmental pollution with harmful products after a cigarette has been put off).
There were demonstrated some differences in the composition of the oral flora of smokers compared to those who do not smoke and also in exposed children, such as a greater percentage of Streptococcus pneumoniae; smoking cessation restores the normal composition within 12 to 15 months(2). Active and passive smoking has harmful effects on airways by increasing adhesion of microorganisms to the epithelial cells of the respiratory tract, alteration in mucocilliar clearance, inflammatory oedema of mucous membrane, depressed local immune response and decreased production of secretory IgA, along with alteration in migration or phagocytes’ chemotaxis. Recent studies have shown that certain particulate fractions from cigarette smoke activate kappa nuclear factor, induce the expression of mucine MUC5B genes and produce mucus hypersecretion, an obstruction by increased mucus viscosity and impaired mucocilliar transport. In addition, nicotine blocks the lymphocytes proliferation and differentiation, accelerates the apoptosis and increases the cytotoxic T-lymphocytes production.
There have been proposed several biological markers to assess the amount of cigarette smoke exposure: measurement of sanguine carboxyhemoglobin or exhaled carbon monoxide, detection of thiocyanates and nicotine in saliva, plasma or urine, or assessment of cotinine, the main nicotine metabolite, also in plasma, urine or hair (US Environmental Protection Agency, 1992). Cotinine is present in human hair for 30 to 90 days after exposure, making it useful in retrospective diagnosis. The value of dosing the thiocyanates is limited because they can have other sources. Nicotine remains present in the plasma only for a few hours, afterwards being metabolised in cotinine (National Cancer Institute California, 1999).
Intrauterine and postpartum exposure to cigarette smoke correlates with an increase incidence of otic infections, bowel inflammatory diseases(2), leukemia, neurocognitive and behavioural disorders, sudden death in infants and, not ultimately, a greater susceptibility to Mycobacterium tuberculosis infection. Various studies have associated the passive smoke with higher frequency of respiratory pathology, including asthma, recurrent wheezing, bronchial hypersecretion, adenoid or tonsil hypertrophy. Lower respiratory infections in the first two years of life and the incidence of tonsillectomies are twice as high in children exposed to cigarette smoke (National Cancer Institute California, 1999).
The mechanisms by which maternal smoking during pregnancy or postpartum exposure interfere with the pathogenesis of asthma are not fully understood. Experimental and clinical studies suggest the irritant effect on airways, altering pulmonary circadian rhythm and increasing air allergen sensitivities(1).
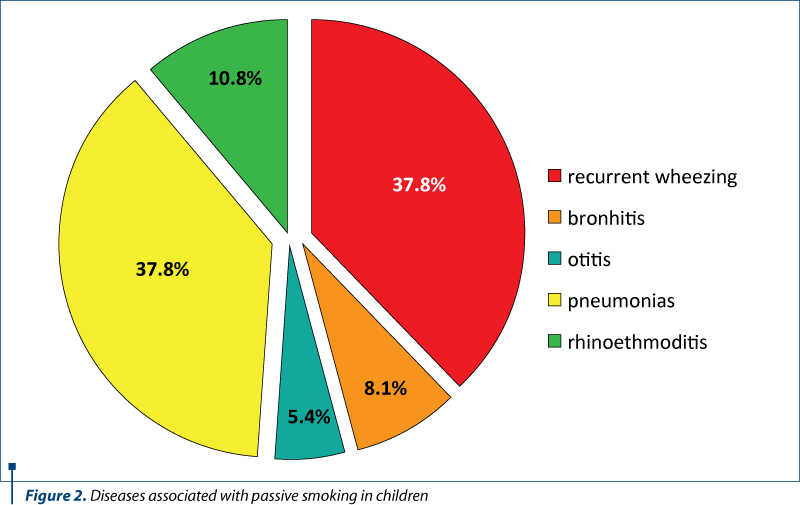
In 2008, in the 2nd Pediatrics Clinic of the “Sf. Maria” Emergency Clinical Hospital for Children in Iaşi, we performed another study on 402 children aged between 0 and 4 years old, in order to assess the impact of passive smoking on the diseases severity and the resistance to the treatment. The determination of cotinine in urine samples, as a marker of cigarette smoke exposure, involves this pollutant as a risk factor for recurrent wheezing, rhino-etmoiditis, bronchitis, otitis and pneumonias (Figure 2).
4. Conclusions
The intrauterine and childhood periods are particularly vulnerable times, during which air pollutants may harmfully interact with the developing and maturing lung. Their action is sometimes concomitant with other exposures, such as viruses, air or food allergens. Primary disease prevention should be based on the avoidance of prenatal or postnatal exposure to these environmental risk factors. Along with the political decision factors, the entire civil society and medical personnel, and both national and international forums should join their efforts to support the improvement of air quality.
There are proposed as protective measures the following: continuous improvement of fuels in order to control the harmful substances emission in the atmosphere, increasing the use of non-pollutant sources of energy, reducing car use in favour of public transport, reducing the use of fossil fuels, avoiding the use of gasoline-powered machines or vehicles, and planting trees with low allergenic potential in urban areas.
Conflicts of interests: The authors declare no conflict of interests.
Bibliografie
- Agabiti N, Mallone S, Forastiere F. SIDRIA Collaborative Group. The impact of parental smoking on asthma and wheezing, Epidemiology. 1999; 10, 692– 698.
- Al-Sayed EM, Ibrahim KS. Second hand tobacco smoke and children. Toxicology and Industrial Health. 2014; 30, 635-644.
- Arshad SH, Kurukulaaratchy RJ, Fenn M, Matthews S. Early life risk factors for current wheeze, asthma, and bronchial hyperresponsiveness at 10 years of age. Chest. 2005; 127, 502–508.
- Bernstein JA, Alexis N, Barnes C, Bernstein IL, Bernstein JA, Nel A, Peden D, Diaz-Sanchez D, Tarlo SM, Williams PB. Health effects of air pollution. Journal of Allergy and Clinical Immunollogy. 2004; 114, 1116-23.
- Crinnion W. Do environmental toxicants contribute to allergy and asthma? Alternative Medicine Review: a Journal of Clinical Therapeutic. 2012; 17, 6-18.
- Cecchi L, D’Amato G, Ayres JG. Projections of the effects of climate change on allergic asthma: the contribution of aerobiology. Allergy. 2010; 65, 1073–1081.
- Cecchi L, D’Amato G, Annesi-Maesano I. Urban Air Pollution and Respiratory Allergy. In: Climate Vunerability: Understanding and Addressing Threats to Essential Resources, Pielke R.A. (Ed.). 2013; Academic Press, Waltham, 105–113.
- Cohen AJ, Ross Anderson H, Ostro B, Pandey KD, Krzyzanowski M, Künzli N, Gutschmidt K, Pope A, Romieu I, Samet JM, Smith K. The global burden of disease due to outdoor air pollution. Journal of Toxicology and Environmental Health. 2005; Part A: Current Issues, 68, 1301-1307.
- D’Amato G, Cecchi L. Effects of climate change on environmental factors in respiratory allergic diseases. Clinical Experimental Allergy. 2008; 38, 1264–1274.
- D’Amato G, Cecchi L, D’Amato M, Annesi-Maesano I. Climate change and respiratory diseases. Environmental Lung Diseases. 2014; 23, 161-169.
- DiFranza J, Aligne CA, Weitzman M. Prenatal and postnatal environmental tobacco smoke exposure and childrens health. Pediatrics. 2004; 113, 1007-1014.
- Dodge R. The respiratory health and lung function of Anglo-American children in a smelter town. American Review of respiratory Disease. 1983; 127, 158-161.
- Ellingsen K, Gauss M, Van Dingenen R. Global ozone and air quality: a multi-model assessment of risks to human health and crops. Atmospheric Chemistry and Physics. 2008; 8, 2163-2223.
- Holgate S. Mechanisms by which Air Pollution Injures the Child’s Respiratory System, In: Effects of Air Pollution on Children’s Health and Development. Review of evidence. WHO Regional Office for Europe, Copenhagen, 2005; 29-43.
- Host A. Allergy testing in children: why, who, when and how? Allergy. 2003; 58, 559-569.
- Hulin M, Simoni M, Viegi G. Respiratory health and indoor air pollutants based on quantitative exposure assessments. European Respiratory Journal. 2012; 40, 1033–1045.
- McGeady SJ. Immunocompetence and allergy. Pediatrics. 2004; 113, 1107-1111.
- National Cancer Institute. Health Effects of Exposure to Environmental Tobacco Smoke: The Report of the California Environmental Protection Agency. Smoking and Tobacco Control Monograph No. 10. Bethesda, MD: US Department of Health and Human Services, National Institutes of Health, National Cancer Institute (NIH Pub. No. 9-4645), 1999.
- Park H, Lee B, Ha EH, Lee JT, Kim H, Hong YC. Association of air pollution with school absenteeism due to illness. Archives of Paediatrics and Adolescent Medicine Journal. 2002; 156, 1235-1239.
- US Environmental Protection Agency. Respiratory Health Effects of Passive Smoking: Lung Cancer and Other Disorders, USEPA Office of Research and Development (Publication No. EPA/600/6-90/006F), Washington DC, 1992.
- Wehby GL, Prater K, McCarthy AM, Castilla EE, Murray JC. The impact of maternal smoking during pregnancy on early child neurodevelopment. J Hum Cap. 2011; 5 (2), 207–254.
- Yang SN, Hsieh CC, Kuo HF, Lee MS, Huang MY, Kuo CH, Hung CH. The effects of environmental toxins on allergic inflammation. Research. 2014; 6, 478-784.
- Yilmaz G, Caylan ND, Karacan CD. Effects of active and passive smoking on ear infection. Current Infectious Disease Reports. 2012; 14, 166-174.





