Perinatal depression is considered one of the most important topics of the new insights in obstetrics and affects 13% of women during pregnancy or in the first year after delivery, leading to important changes in the mother’s life or her family. Objective and methodology. To review the literature on perinatal depression, including diagnosis, complications and treatment. We searched PubMed, Medline and medical texts pertaining to perinatal depression up to January 2019. Results. Validated scales such as Edinburgh Postnatal Depression Scale and clinical criteria approved by the Diagnostic and Statistical Manual of Mental Disorders V (DSM-V) are used to obtain a proper diagnosis. The consequences of untreated perinatal depression can be devastating, affecting both the mother and the child. Despite the fact that there are still drawbacks regarding diagnosis and treatment, it is recommended that all pregnant women be screened at least once during the perinatal period, using accepted and validated tools.
Depresia perinatală
Perinatal depression
First published: 13 martie 2020
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/Gine.27.1.2020.2884
Abstract
Rezumat
Depresia postpartum reprezintă un subiect important în obstetrică, afectând 13% dintre femei în timpul sarcinii sau în primul an post-partum, ducând la schimbări importante în viaţa mamei şi a familiei. Obiectiv şi metodologie. Revederea literaturii cu privire la diagnosticul, complicaţiile şi tratamentul depresiei post-partum. Am cercetat bazele de date PubMed şi Medline şi documente medicale care făceau referire la depresia post-partum până în ianuarie 2019. Rezultate. Diagnosticul de depresie post-partum este stabilit prin intermediul scalelor validate, precum Scala Edinburgh de Depresie Postnatală sau chestionarul Beck, care evaluează simptomele episoadelor depresive majore, elaborate de Asociaţia Americană de Psihiatrie, prin intermediul ghidului DSM-V. Consecinţele lipsei tratamentului în depresia post-partum pot fi grave, afectând mama şi nou-născutul. Deşi diagnosticul şi tratamentul depresiei peripartum sunt încă deficitare, este recomandat ca fiecare femeie să fie evaluată măcar o dată pe parcursul sarcinii, pentru a putea beneficia de tratament adecvat.
Introduction
An important event in the life of the future mother and her family, pregnancy is characterized by a normal state of health, both psychologically and physically. Extensive research, including a prospective cohort study(1), has shown that women with prior diagnosis of depression are more likely to develop new episodes or to relapse during pregnancy(2,3). Depression, a serious mood disorder, affects 8% to 11% of non-pregnant women from developing countries(4), reaching 13% during pregnancy, being one of the most prevalent medical conditions, outpacing gestational diabetes. Studies have shown that anxiety and depression during parturiency represent risk factors for complications during the perinatal period such as preterm birth, low weight at birth and low Apgar score(5).
Although it represents an important medical condition, depression during pregnancy can be easily neglected by the health care provider or the obstetrician-gynecologist, given the fact that there are still flaws in the diagnosis and treatment of this condition. One of the major drawbacks is the teratogenic effect of the most antidepressants, so the objective of this article is to evaluate the risk factors, clinical image, complications and the treatment of depression during pregnancy.
![Table 3.Identification of women prone to mental health disorders during pregnancy (National Institute for Health and Care Excellence [NICE] – Antenatal and postnatal mental health: clinical management and service guidance clinical guideline)](/image/10249/0/table_3_identification_of_women_prone_to_mental_health_disorders_during_pregnancy_national_10249.png)
The reviews available in literature usually estimate the prevalence and incidence of perinatal depression or treatment options, but as far as we know there has been little effort to include diagnosis, complications and treatment options in the same review. The present systematic review contributes significantly by connecting these three gaps in the literature.
Epidemiology
Addressing the international medical literature through PubMed and Medline and the latest studies on depression during pregnancy, it can be noticed that depression is the second cause of Disability-Adjusted Life-Years (DALYs) in women and the leading cause of maternal morbidity. During the second and third trimesters of pregnancy, the percentage of depressive symptoms are almost twice as high as those in the first trimester, 13% versus 7%, as shown in a meta-analysis by Benett et al.(6) However, controversial data exist, given the fact that changes of mood, appetite and sleeping pattern during pregnancy are often misdiagnosed.
Risk factors
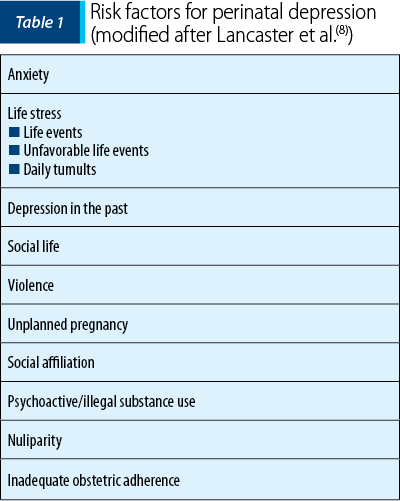
There is still no research to show why for some women pregnancy serves as a protective factor against depression. Pregnancy represents a complex experience, with psychological, physical and social changes. According to a study made by Woods et al., antenatal psychosocial stress is frequent and is associated with depression, panic disorder, drug use and domestic violence(7). Extensive research has shown that there is a number of risk factors that can trigger perinatal depression (Table 1)(8), including history of depressive episodes, substance abuse, miscarriage/abortion, low-income families and lack of social support. In this category, there are also included major life events such as divorce or the loss of a close person.
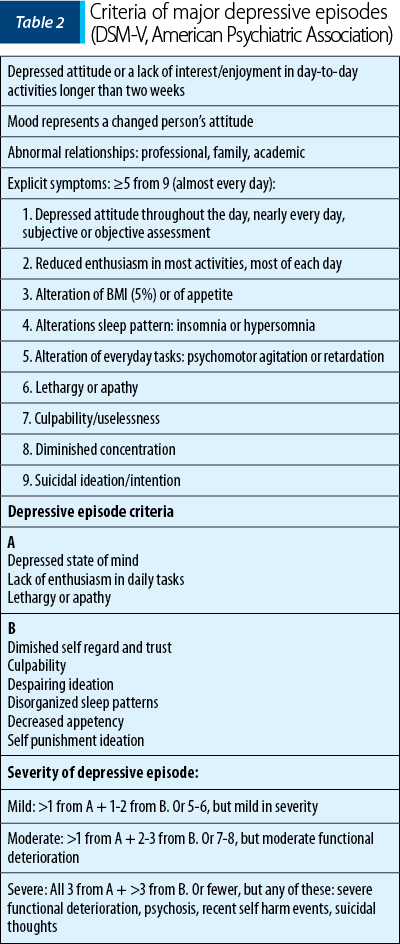
In 2013, the American Psychiatric Association (APA), through the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-V), established the clinical criteria for diagnosing major depressive episodes, both in general population and pregnant women (Table 2).
Validates scales, such as Edinburgh Postnatal Depression Scale (EPDS), are used to assess clinical depression symptoms. EPDS was first developed in 1987 and uses a 10-item questionnaire that correlates to clinical depression symptoms, with a maximum score of 30 and possible depression at a score of 10 or greater(9). The severity of depression is evaluated through the Beck Depression Inventory (BDI), a 21-question multiple choice inventory, each question created to evaluate distinct symptoms prevalent among population with depression(10). In December 2014, the National Institute for Health and Care Excellence (NICE) from the United Kingdom published a guideline for diagnosing, evaluating and attending women within perinatal period with mental health disorders. Two questions were considered to be asked during the first visit of the pregnant woman to the health care provider (Table 3)(11). If the patient responded positively to either of the two questions, she was at risk to develop a mental health disorder during pregnancy, and EPDS was considered in order to achieve a complete assessment.
Risks of untreated depression during pregnancy
It is acknowledged that depression in pregnant women increases mortality and morbidity, leading to obstetrical and puerperal complications(12). Gestational hypertension and preeclampsia have been associated with untreated perinatal depression(13). Studies have shown that the most frequent complications of untreated perinatal depression were: miscarriage or abortion, bleeding during pregnancy, neonatal growth restriction, preterm birth, low Apgar scores at birth, high cortisol levels in neonate(14). In a 2009 study conducted by Marcus et al., it was demonstrated that pregnancy is a period characterized by variation in gonadal hormones that lead to behavioral changes mostly concerning sleep and appetite, with complications such as weight gain, fewer visits to the health care provider during pregnancy, and substance abuse(15). In 2010, Field published a systematic review on the prenatal depression effects and interventions, studying the implication of high levels of cortisol in perinatal complications such as premature birth or low weight at birth(16). It was emphasized the fact that women with prenatal depression have lower levels of dopamine and serotonin, resulting in premature births, low weight at birth, newborn with higher levels of cortisol and lower levels of dopamine and serotonin. Women who had premature births had higher cortisol levels between 15 and 19 weeks of gestation(17). In a prospective study led by Kurki in 2000, it was analyzed the relationship between anxiety or depression during pregnancy and the risk of subsequent preeclampsia(18). From 623 nulliparous women, 4.5% of them developed preeclampsia, with an increased risk in those who associated depression or anxiety (odds ration [OR] 2.5 versus 3.2).
Treatment
As in other medical conditions, choosing the right treatment for the perinatal depression is an important step of the entire process, with two main options being available: psychotherapy and pharmacological treatment.
Psychotherapy
For a pregnant woman diagnosed with depression, psychotherapy could be the first option or an adjuvant to the pharmacological treatment. A 2006 meta-analysis conducted by Bledsoe concluded that medication and various therapies, such as cognitive-behavioral therapy (CBT), group therapy and interpersonal psychotherapy (IPT), gave the best results in women with perinatal depression(19). O’Hara showed in a 2000 study that women with major depressive disorders who received interpersonal psychotherapy (IPT) had decreased depression symptoms and enhanced social relationships, IPT being an alternative to pharmacological treatment, especially for women who were breastfeeding(20). Other trials recommended bright light therapy, acupuncture and massage as an alternative treatment(21), as well as omega-3 fatty acids, considered to being superior to placebo(22).
Pharmacological treatment
Women with prior depression who are on a pharmacological treatment may prefer to cease treatment before conception or during pregnancy. In a 2006 longitudinal psychiatric assessment led by Cohen, on 201 pregnant women with psychiatric illness, 26% of those who maintained their medication relapsed comparative to 68% of those who discontinued medication(23).
The first-line treatment for depression consists of selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs). As antidepressants pass through the placenta, Hendrick et al. determined that in 86.8% of the studied cases, antidepressants and their metabolites were detectable in umbilical cord samples, with mean ratios fluctuating from 0.29 to 0.89 (umbilical cord to maternal serum concentration)(24). Selective serotonin reuptake inhibitors (SSRIs) exposure in pregnancy may lead to spontaneous miscarriage, low weight at birth, low Apgar scores, fetal death and seizures in infants(25). In a 2006 review by Rahimi et al., it was reported that SSRIs did not increase the risk of congenital malformations, be it major, minor or cardiovascular, but did increase the rate of spontaneous abortion(26). However, a 2007 case-control study showed a small increased risk of omphalocele, craniosynostosis and anencephaly(27). A review of cohort and case studies reported that exposure to pharmacological antidepressant medication during the third trimester of pregnancy may lead to a neonatal behavioral syndrome, also being called toxicity in neonates with symptoms like hypotonia, weak or absent cry, respiratory distress, hypoglycemia, low Apgar score and seizures(28). Persistent pulmonary hypertension of the newborn (PPHN) is found in 1/700 in normal newborns, while in pregnancies exposed to SSRIs after 20 weeks of gestations, the risk increases to 7/1000, as it was showed in a case study report from 2006, as a result of inhibition of nitric oxide or direct effect on pulmonary smooth muscle cell that leads to pulmonary artery constriction(29). Altered blood concentrations of serotonin and sleep disorders were mentioned as long-term effects of antidepressants(30).
As in other pharmacological therapies, anxiolytic substances such as benzodiazepines are used as single or adjuvant therapies in women with perinatal depression. Benzodiazepines do not have an increased teratogenic risk, as it was demonstrated in a study from 2002(31), but they can be associated with the risk of oral cleft(32). Mood stabilizers, such as lithium, might be associated with cardiac anomalies such as Ebstein’s disease, if the exposure was present in the first trimester of pregnancy(33). Haloperidol exposure, an antipsychotic, may lead to limb defects, although it was not demonstrated an increased risk of congenital malformations(34). The newer antipsychotics (i.e., clozapine, olanzapine, risperidone, quetiapine, aripiprazole and ziprasidone) have side effects like glucose intolerance and weight gain, but do not have an increased risk of malformations; it can lead to low-weight at birth and higher rates of spontaneous abortion(35).
Conclusions
Pregnancy represents an important moment in the life of the mother and her family, with psychological and physical changes. Unrecognized and untreated depression symptoms may lead to disastrous consequences, both for the mother and the child. In DSM-V, American Psychiatric Association defined major depressive disorders with symptoms that can be assessed through validated scales such as Edinburgh Postnatal Depression Scale and Beck Depression Inventory. Treatment options are available, including psychotherapy and pharmacological medication. In the end, it is important to identify and treat perinatal depression in order to lower the impact on pregnancy and the future newborn. n
Conflict of interests: The authors declare no conflict of interests.
Bibliografie
- Müldner-Nieckowski Ł, Cyranka K, Smiatek-Mazgaj B, Mielim˛aka M, Sobanski J, Rutkowski K. Psychotherapy for pregnant women with psychiatric disorders. Psychiatr Pol. 2015; 49:49–56.
- Cohen LS, Altshuler LL, Harlow BL, Nonacs R, Newport DJ, Viguera AC, et al. Relapse of major depression during pregnancy in women who maintain or discontinue antidepressant treatment. JAMA. 2006; 295:499–507.
- Kupfer DJ, Frank E, Phillips ML. Major depressive disorder: New clinical, neurobiological, and treatment perspectives. Lancet. 2012; 379:1045–55.
- Gaynes BN, Gavin N, Meltzer-Brody S, Lohr KN, Swinson T, Gartlehner G, et al. Perinatal depression: prevalence, screening accuracy, and screening outcomes. Evid Rep Technol Assess (Summ). 2005; 119:1–8.
- Dunkel Schetter C, Tanner L. Anxiety, depression and stress in pregnancy. Curr Opin Psychiatry. 2012; 25:141–8.
- Bennett HA, Einarson A, Taddio A, Koren G, Einarson TR. Prevalence of depression during pregnancy: systematic review. Obstet Gynecol. 2004; 103:698–709.
- Woods SM, Melville JL, Guo Y, Fan M-Y, Gavin A. Psychosocial stress during pregnancy. Am J Obstet Gynecol. 2010; 202:61.e1-61.e7.
- Lancaster CA, Gold KJ, Flynn HA, Yoo H, Marcus SM, Davis MM. Risk factors for depressive symptoms during pregnancy: a systematic review. Am J Obstet Gynecol. 2010; 202:5-14.
- Cox JL, Holden JM, Sagovsky R. Detection of postnatal depression: development of the 10-item Edinburgh Postnatal Depression Scale. Br J Psychiatry. 1987; 150:782–6.
- Beck AT. Depression: Causes and Treatment. Univ Pennsylvania Press, 1972.
- National Institute for Health and Care Excellence (NICE). Antenatal and postnatal mental health: clinical management and service guidance Clinical guideline Pub: 2014 Dec 17 nice.org.uk/guidance/cg192 retrieved 3rd Feb 2019
- Chung TK, Lau TK, Yip AS, Chiu HF, Lee DT. Antepartum depressive symptomatology is associated with adverse obstetric and neonatal outcomes. Psychosom Med. 2001; 63:830–4.
- Paarlberg KM, Vingerhoets AJ, Passchier J, Dekker GA, van Geijn HP. Psychosocial factors and pregnancy outcome: a review with emphasis on methodological issues. J Psychosom Res. 1995; 39:563–95.
- Field T, Diego M, Hernandez-Reif M, Salman F, Schanberg S, Kuhn C, et al. Prenatal anger effects on the fetus and neonate. J Obstet Gynaecol. 2002; 22:260–6.
- Marcus SM. Depression during pregnancy: rates, risks and consequences. Motherisk Update 2008. Can J Clin Pharmacol. 2009; 16(1):e15-22.
- Field T, Diego M, Hernandez-Reif M. Prenatal depression effects and interventions: a review. Infant Behav Dev. 2010; 33(4):409-18.
- Sandman CA, Glynn L, Dunkel-Shetter C, Wadhwa P, Garite T, Chicz-DeMet A, Hobel C. Elevated maternal cortisol early in pregnancy predicts third trimester levels of placental corticotropin releasing hormone (CRH): priming the placental clock. Peptides. 2006; 27:1457–63.
- Kurki T, Hiilesmaa V, Raitasalo R, Mattila H, Ylikorkala O. Depression and anxiety in early pregnancy and risk for preeclampsia. Obs Gynecol. 2000;95:487–90.
- Bledsoe SE, Grote NK. Treating depression during pregnancy and in the postpartum: a preliminary meta-analysis. Res Soc Work Pract. 2006; 16:109-20.
- O’Hara MW, Stuart S, Gorman LL, Wenzel A. Efficacy of interpersonal psychotherapy for postpartum depression. Arch Gen Psychiatry. 2000; 57(11):1039-45.
- Pearlstein T. Perinatal depression: treatment options and dilemmas. J Psychiatry Neurosci. 2008; 33(4):302–18.
- Su KP, Huang SY, Chiu TH, et al. Omega-3 fatty acids for major depressive disorder during pregnancy: results from a randomized, double-blind, placebo-controlled trial. J Clin Psychiatry. 2008; 69:644-51.
- Cohen LS, Nonacs RM, Bailey JW, et al. Relapse of depression during pregnancy following antidepressant discontinuation: a preliminary prospective study. Arch Womens Ment Health. 2004; 7:217-21.
- Hendrick V, Stowe ZN, Altshuler LL, et al. Placental passage of antidepressant medications. Am J Psychiatry. 2003; 160:993-6.
- Simon GE, Cunningham ML, Davis RL. Outcomes of prenatal antidepressant exposure. Am J Psychiatry. 2002; 159:2055-61.
- Rahimi R, Nikfar S, Abdollahi M. Pregnancy outcomes following exposure to serotonin reuptake inhibitors: a meta-analysis of clinical trials. Reprod Toxicol. 2006; 22:571-5.
- Alwan S, Reefhuis J, Rasmussen SA, et al. Use of selective serotonin-reuptake inhibitors in pregnancy and the risk of birth defects. N Engl J Med. 2007; 356:2684-92.
- Koren G, Matsui D, Einarson A, et al. Is maternal use of selective serotonin reuptake inhibitors in the third trimester of pregnancy harmful to neonates? CMAJ. 2005; 172:1457-9.
- Chambers CD, Hernandez-Diaz S, Van Marter LJ, et al. Selective serotonin-reuptake inhibitors and risk of persistent pulmonary hypertension of the newborn. N Engl J Med. 2006; 354:579-87.
- Laine K, Heikkinen T, Ekblad U, et al. Effects of exposure to selective serotonin reuptake inhibitors during pregnancy on serotonergic symptoms in newborns and cord blood monoamine and prolactin concentrations. Arch Gen Psychiatry. 2003; 60:720-6.
- Eros E, Czeizel AE, Rockenbauer M, et al. A population-based case-control teratologic study of nitrazepam, medazepam, tofisopam, alprazolam and clonazepam treatment during pregnancy. Eur J Obstet Gynecol Reprod Biol. 2002; 101:147-54.
- Dolovich LR, Addis A, Vaillancourt JM, et al. Benzodiazepine use in pregnancy and major malformations or oral cleft: meta-analysis of cohort and case-control studies. BMJ. 1998; 317:839-43.
- Cohen LS, Friedman JM, Jefferson JW, et al. A reevaluation of risk of in utero exposure to lithium. JAMA. 1994; 271:146-50.
- Trixler M, Gati A, Fekete S, et al. Use of antipsychotics in the management of schizophrenia during pregnancy. Drugs. 2005; 65:1193-206.
- McKenna K, Koren G, Tetelbaum M, et al. Pregnancy outcome of women using atypical antipsychotic drugs: a prospective comparative study. J Clin Psychiatry. 2005; 66:444-9.
Articole din ediţiile anterioare
Rolul infecţiilor orale în naşterea prematură
Prematuritatea şi greutatea scăzută la naştere sunt unele din complicaţiile frecvente ale sarcinilor. Infecţiile orale, în special bolile parodonta...
Tratamentul endodontic în sarcină
Tratamentele stomatologice pe parcursul sarcinii reprezintă o situaţie cu care medicii dentişti se confruntă în mod frecvent. Pacienta însărcinată ...
Dificultăţi de examinare ecografică la gravidele supraponderale. Studiu retrospectiv şi analiza literaturii
Introducere. În ultimele decenii, sistemul medical se confruntă tot mai des cu paciente obeze sau supraponderale. Obezitatea este asociată cu un ri...
Complicaţiile hipertensiunii arteriale induse de sarcină – management terapeutic şi prognostic
Managementul sarcinii asociate cu hipertensiune gestaţională se realizează în funcţie de severitatea hipertensiunii arteriale, de vârsta ges...