Introduction. Endometriosis with different stages of severity affects approximately 5% of women. It can involve the ovaries, the pelvic peritoneum, the pouch of Douglas (POD), the rectum, the rectosigmoid junction, the rectovaginal septum (RVS), the uterosacral ligaments (USL), the vagina and the bladder. The ultrasound evaluation is the firsthand method to detect ovarian endometriosis, but recently it has proven its value in the diagnosis of deep infiltrating endometriosis (DIE). Materials and method. We have evaluated the medical database and international guides regarding ultrasound examination of DIE. Results. Recent data prove that the purpose of ultrasound examination of a woman suspected of endometriosis is to explain the main symptoms, to identify the lesions and to evaluate the grade of severity before choosing the appropiate conservatory or surgical means of therapy. Using transvaginal ultrasound as the first means of diagnosis, the examiner must first evaluate the uterus and adnexa. Ovarian endometriomas are frequently associated with other endometriosis-specific lesions such as adhesions and DIE. The second step is to look for “soft markers” and to test the mobility of the ovaries. The presence of these signs increases the chance that DIE and adhesions are to be found. By applying pressure between the uterus and ovaries, one can test whether the ovaries are fixed to the uterosacral ligaments, medially or laterally to adjacent structures. The third step is the evaluation of the pouch of Douglass using the sliding sign. The fourth and final step is to search for DIE nodules in the anterior and posterior compartments of the pelvis. Conclusions. Transvaginal ultrasound is the firsthand method of evaluating patients with ovarian endometriosis and DIE. The prediction of severe forms of DIE, as well as the obliteration of the pouch of Douglas are essential in planning a multidisciplinary surgical approach.
Ecografia în endometrioza infiltrativă profundă
Ultrasound evaluation of deep infiltrating endometriosis
First published: 13 martie 2020
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/Gine.27.1.2020.2886
Abstract
Rezumat
Introducere. Endometrioza cu diferite grade de severitate afectează aproximativ 5% dintre femei. Aceasta poate implica ovarele, peritoneul pelvian, fundul de sac Douglas (POD), rectul, rectosigmoidul, septul rectovaginal (SRV), ligamentele uterosacrate (LUS), vaginul şi vezica urinară. Ecografia este tehnica imagistică de primă linie utilizată în diagnosticul endometriozei ovariene, dar recent şi-a dovedit aplicabilitatea şi pentru endometrioza infiltrativă profundă (EIP). Materiale şi metodă. Am evaluat bazele de date medicale şi ghidurile internaţionale cu privire la evaluarea ecografică a endometriozei infiltrative profunde. Rezultate. Datele recente demonstrează că scopul efectuării unui examen ecografic la o femeie suspectată de endometrioză este de a explica simptomele de bază, de a localiza leziunile şi de a evalua severitatea bolii înainte de terapia medicală sau chirurgicală. Utilizând ecografia transvaginală ca instrument de imagistică de primă linie, examinatorul trebuie să evalueze iniţial uterul şi anexele. Endometriomele ovariene sunt asociate frecvent cu alte leziuni endometriozice, cum ar fi aderenţele şi EIP. Al doilea pas este de a căuta „soft markeri” sonografici, adică de a testa sensibilitatea zonei şi mobilitatea ovarelor. Prezenţa acestor markeri creşte probabilitatea endometriozei profunde şi a aderenţelor. Prin aplicarea presiunii între uter şi ovar, se poate evalua dacă ovarul este fixat medial, lateral sau la LUS. Al treilea pas este evaluarea Douglasului, utilizând un „indicator de alunecare”. Al patrulea pas este identificarea nodulilor de EIP în compartimentele anterioare şi posterioare. Concluzii. Ecografia transvaginală este metoda imagistică de primă intenţie în evaluarea pacientelor cu endometrioză ovariană şi EIP. Predicţia formelor severe de EIP, precum şi obliterarea Douglasului sunt esenţiale în planificarea unei abordări chirurgicale multidisciplinare.
Introduction
Endometriosis is a chronic estrogen-dependent inflammatory disease, associated with chronic pelvic pain and infertility. It has a wide spectrum of symptoms and a significant socioeconomic impact on pacients(1). Histologically, it is defined by the presence of ectopic endometriotic tissue outside the uterus. It has a prevalence of 2-10% in women of reproductive age and is cited in 50% of cases of women who suffer from infertility(2).
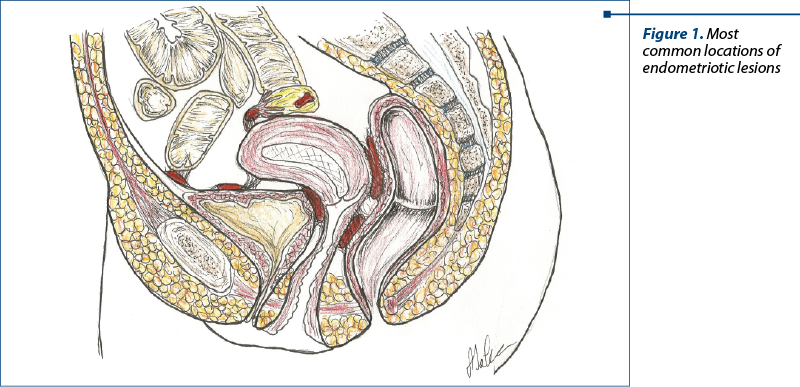
Endometriosis can be detected in multiple regions of the body, most commonly in the ovaries, pelvic peritoneum, pouch of Douglas, rectum, sigmoid, bladder, but rare cases of endometriosis of the lungs, liver and pericardium have been described (Figure 1).
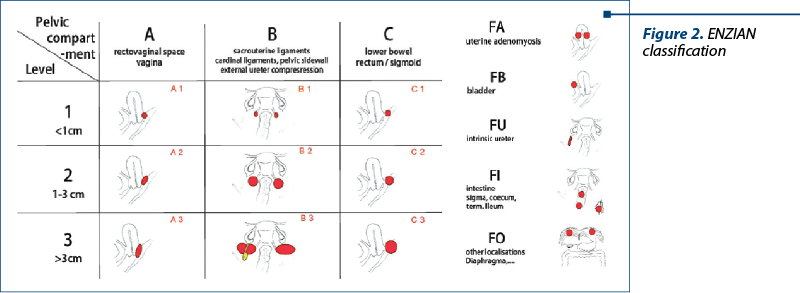
A particular form of endometriosis is the deep infiltrating endometriosis (DIE), which is represented by lesions that are found more than 5 mm below the peritoneal surface. The location and severity of lesions are established by means of the ENZIAN classification(3) (Figure 2).
Materials and method
We have evaluated the medical database and international guides regarding ultrasound examination of DIE.
Results
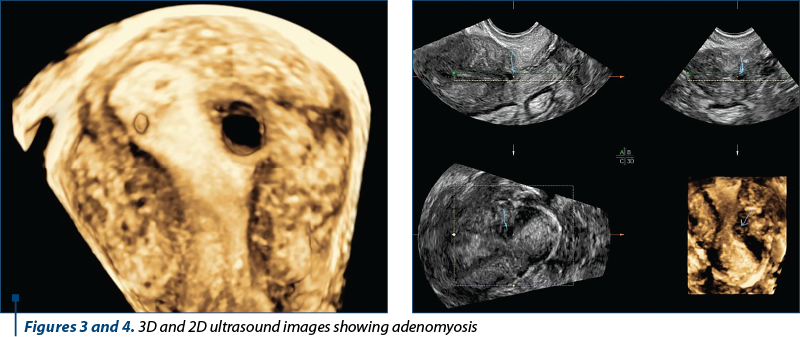
In our study, we have defined four steps for DIE evaluation. Firstly, we examine the uterus and adnexa. Secondly, certain signs which have been defined as “soft markers” must be looked for. The evaluation will be continued by examining the pouch of Douglas, followed by the anterior and posterior compartments of the pelvis. The primary elements that must be searched for during the ultrasound of the uterus and adnexa are adenomyosis (Figures 3 and 4) and ovarian endometriomas, which are in most cases associated with other endometriotic lesions such as pelvic adhesions and DIE. This brings us to the second step in our examination: searching for soft markers. By applying pressure between the uterus and ovary with the vaginal probe, the mobility of the ovary can be assessed: whether it is fixed medially to the uterosacral ligaments or to other nearby structures. The aforementioned soft markers are the points where pelvic organ mobility is limited and therefore the examination with the vaginal probe combined with manual palpation on the lower abdomen is painful for the patient. These signs lead the examiner to suspect peritoneal adhesions due to DIE. The kissing ovaries sign can also be attributed to the presence of endometriotic lesions.
The third step consists of the examination of the pouch of Douglas, using a maneuver called the sliding sign (Figure 5). The structures that will be evaluated are the uterus, the sigmoid colon, the rectum and the rectosigmoid junction, and the examination will differ according to uterine positioning. Thus, when the uterus is positioned in AVF, using the vaginal probe, pressure will be applied to the cervix to check if the rectum slides freely along the posterior cervical wall, respectively the uterine corpus. To continue the evaluation, the examiner will place his free hand on the lower abdomen of the patient and apply pressure in order to move the uterus between the vaginal probe and his hand and see whether the intestines slide on the uterine fundus and its posterior wall. When the uterus is positioned in RVF, the examiner will proceed similarly by applying pressure to the posterior uterine wall and during the second phase of the evaluation he will asses whether the sigmoid colon glides over the inferior segment of the uterus.
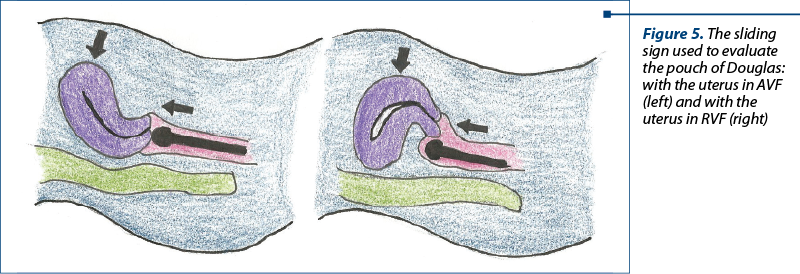
The presence of pelvic adhesions due to DIE is proven if, during at least one of the phases of the examination, the immobility between the uterus and adjacent structures is observed.
The last step consists in the evaluation of the anterior and posterior compartment of the pelvic region. The structures of the anterior compartment include the bladder, the uterovesical region and the ureters, while the posterior compartment includes the uterosacral ligaments, the rectum, the rectosigmoid junction, the posterior vaginal fornix and the sigmoid colon.
It is preferred that during this step the bladder should contain a small amount of urine, but not be fully empty.
Anterior compartment
In order to perform a systematic review of the bladder, we have divided it into four components: the trigonal zone, the bladder base, the bladder dome and the extra-abdominal component. Bladder DIE occurs more frequently in its base. Any found lesion will be measured in three lanes (anterior-posterior, transverse and sagittal). In order for a lesion to be considered DIE, it must have advanced at least into the muscular layer of the bladder wall.
The uterovesical region will be examined similarly to the pouch of Douglas using the sliding sign, only in this case the vaginal probe will be placed in the anterior vaginal fornix. The uterus will be moved between the probe and the examiners’ free hand positioned on the lower abdomen of the patient, above the pubic bone. Previous births through caesarean section should be taken into consideration as postoperative adhesions can reduce the mobility of the aforementioned structures and lead to a false positive result.
The ureters can be found using ultrasound by first identifying the urethra in a sagittal plane and then moving the probe laterally towards the iliac fossae. Strictures on their trajectory should be identified and other indirect signs such as ureteral dilatations and hydronephrosis.
Reduced caliber of the ureteral lumen occurs as a consequence of external compression or intrinsical infiltration. The distance between the distal ureteral orifice and the site of the lesion should always be measured in case of the need for the insertion of a ureteral stent(4).
Posterior compartment
Examining the posterior compartment requires the identification of the number, the size and the anatomical positioning of DIE nodules. The ultrasound aspect of these lesions vary from a hypoechoic thickening of the vaginal or intestinal wall to solid hypoechoic nodules that can be either precisely delimited or not, and may have a wide range of dimensions. DIE lesions of the rectovaginal septum are seen as the absence of normal echostructure at this level, which consists of a hypoechoic space between the vagina and rectum. Isolated nodules in this region are considered rare; they are usually an extension of endometriomas situated in the posterior vaginal wall or the anterior rectal wall. It is necessary to measure the distance between the inferior limit of the nodule and the anal sphincter in order to decide the preferred therapeutic conduit.
Endometriotic nodules located in the vaginal wall can have either a homogenous or non-homogenous structure and can be associated with cystic areas surrounding the primary lesion. In order to identify these lesions, the hypoechoic layer of the vaginal wall will be closely examined, searching for areas where this layer is thickened or nodules are present, and for possible extensions towards the rectovaginal space.
Similarly to the bladder, DIE lesions of the digestive tract are defined as the presence of endometrial glands and stroma deep in the intestinal wall, advancing at least into the tunica muscularis, where it induces smooth muscle hyperplasia and fibrosis(5).
At this level, lesions can be isolated, multifocal, when there are more nodules in the same intestinal segment, or multicentric, these being defined as the presence of endometriotic nodules in more than one intestinal segment. In the case of suspected multicentric lesions, MRI evaluation is preferred over ultrasound examination.
The ultrasound aspect is similar to that of DIE nodules found in the vaginal wall. These nodules can sometimes present a narrow extension, making the lesion look like a comet. When pelvic adhesions are present, the lesion will take on the aspect of moose horns due to the traction that these adhesions make on the intestinal wall. Intestinal DIE lesions can be defined according to the rectal or colon segment they form in. Usually, the uterosacral ligaments are not visible on ultrasound. Therefore, endometriotic lesions on these structures will be seen as a hypoechoic thickening of the adjacent adipose tissue. They can present as isolated lesions or an extension of endometriomas formed on nearby structures. Nodules located close to the insertion of the uterosacral ligaments on the cervix, near the torus uterinus, will take on the aspect of a central thickening of the retrocervical region. Rectovaginal nodules are an extension of posterior vaginal fornix lesions through the rectovaginal septum, towards the anterior rectal wall or the other way around. The ultrasound aspect will resemble an hourglass since the lesions become narrow as they pass through the rectovaginal septum. These are usually quite large lesions, with a diameter larger than 3 cm.
Other means for the evaluation of pelvic DIE:
-
Color Doppler ultrasound is useful in colorectal cancer differential diagnosis.
-
Sonovaginography with gel contrast facilitates the complete visualization of the vaginal walls and the posterior and anterior vaginal fornix.
-
Transrectal ultrasound using the vaginal probe for virgo intacta patients.
-
Tenderness-guided ultrasound, which requires that the patient lead the examiner towards the points of discomfort so that he might identify possible DIE lesions in those regions.
-
Rectal water contrast transvaginal ultrasound during which saline water is inserted in the rectum through a catheter for a better view of the rectal walls and the rectosigmoid junction(6,7).
Conclusions
Transvaginal ultrasound is the firsthand method in evaluating cases of DIE, but when multicentric lesions are suspected, MRI examination is necessary, as more than 50% of intestinal endometriosis lesions are associated with another lesion located on a different segment of the digestive tract(8). Extensive localization of endometriotic lesions is essential in order to correctly choose the appropiate therapeutic conduct, whether it is a surgical or a conservatory approach.
Conflicts of interests: The authors declare no conflict of interests.
Bibliografie
- Fritz MA, Speroff L. Clinical gynecologic endocrinology and infertility. 8th ed. Lippincott Williams & Wilkins. 2010.
- Hickey M, Ballard K, Farquhar C. Endometriosis. BMJ. 2014; 348: g1752.
- Fadhlaoui A, Bouquet de la Jolinière J, Feki A. Endometriosis and infertility: how and when to treat? Front Surg. 2014; 1:24.
- Suardika A, Astawa Pemayun TG. New insights on the pathogenesis of endometriosis and novel non-surgical therapies. J Turk Ger Gynecol Assoc. 2018; 19(3):158–6.
- Alimi Y, Iwanaga J, Loukas M, Tubbs RS. The clinical anatomy of endometriosis: A review. Cureus. 2018; 10(9):e3361.
- Fedele L, Bianchi S, Portuese A, Borruto F, Dorta M. Transrectal ultrasonography in the assessment of rectovaginal endometriosis. Obstet Gynecol. 1998; 91:444–8.
- Belghiti J, Thomassin-Naggara I, Zacharopoulou C, Zilberman S, Jarboui L, Bazot M, Ballester M, Darai E. Contribution of computed tomography enema and magnetic resonance imaging to diagnose multifocal and multicentric bowel lesions in patients with colorectal endometriosis. J Minim Invasive Gynecol. 2015; 22:776–84.
- Menakaya U, Reid S, Lu C, Gerges B, Infante F, Condous G. Performance of an ultrasound based endometriosis staging system (UBESS) for predicting the level of complexity of laparoscopic surgery for endometriosis. Ultrasound Obstet Gynecol. 2016; 48(6):786-95.
Articole din ediţiile anterioare
Evaluarea corpului calos fetal în examinarea de rutină - ecografie 2D versus ecografie 3D
În cadrul proiectul „Neurosonografia fetală - evaluarea potenţialului ecografiei 3D în examinarea de rutina” am analizat posibilităţile evaluării c...
IRM – avantaje şi limite în diagnosticul şi tratamentul endometriozei
Endometrioza este o boală ginecologică multifocală comună, care se manifestă în timpul anilor de reproducere, cauzând adesea dureri pelviene croni...
Tumoră uterină asemănătoare tumorilor de cordoane sexuale ovariene: o diagnosticăm dacă o cunoaştem. Prezentare de caz şi review al literaturii
Tumora uterină asemănătoare tumorilor de cordoane sexuale ovariene (UTROSCT) reprezintă un tip rar de neoplasm uterin, având un potenţial malign in...
A rare case of complete hydatidiform mole with prolonged evolution – clinical case
Mola hidatiformă, cunoscută drept sarcină molară, reprezintă o boală gestaţională trofoblastică cu punct de plecare placentar şi capacitate de meta...