Genital melanomas are the second most common cancers of the female external genitalia after squamous cell carcinoma. It is a rare pathology considering that vulvar, vaginal, and cervical melanomas represent less than 1-3% of all melanomas in women. Amongst these, vulvar melanoma is the most common. The aim of this article is to present a collection of data stated in literature about genital malignant melanoma since the interest in this disease has significantly increased, although, until now, there have been few studies made about it. It is known that melanomas arising from urogenital mucosa have many diferrences from cutaneous melanomas, including higher aggressiveness, worse prognosis and, not at least, different molecular abnormalities, such as c-KIT mutations, that modify the therapeutic management. Most of the data regarding treatment are assumed from cutaneous malignant melanoma, but recent studies show that care should be taken when applying the same conduct. Even more, the prognosis and treatment are guided by the depth of tumor rather than the lesion diameter, which means that FIGO staging used for squamos cell carcinoma has no use for this type of genital neoplasm.
Localizări genitale ale melanomului malign
Genital localization of malignant melanoma
First published: 15 noiembrie 2016
Editorial Group: MEDICHUB MEDIA
Abstract
Rezumat
Melanoamele genitale sunt al doilea cel mai frecvent cancer al organelor genitale externe feminine, după carcinomul cu celule scuamoase. Este o patologie rară, având în vedere că localizările vulvare, vaginale şi cervicale constituie mai puţin de 1-3% din totalul melanoamelor la femei. Printre acestea, melanomul vulvar este cel mai frecvent. Scopul acestui articol este de a prezenta o colecţie de date menţionate în literatura de specialitate despre melanomul malign genital, deoarece interesul pentru această boală a crescut în mod semnificativ, cu toate că, până în prezent, au existat doar câteva studii efectuate cu privire la aceasta. Este cunoscut faptul că melanoamele care rezultă din mucoasa urogenitală prezintă multe diferenţe comparativ cu melanoamele cutanate, inclusiv agresivitate mai mare, prognostic mai prost şi, nu în ultimul rând, diferite anomalii moleculare, cum ar fi mutaţiile c-KIT, care modifică managementul terapeutic. Cele mai multe date cu privire la tratament sunt preluate din conduita în melanomul malign cutanat, dar studiile recente arată că trebuie avut grijă atunci când se aplică acelaşi comportament. Chiar mai mult, prognosticul şi tratamentul sunt ghidate mai degrabă de profunzimea tumorii, decât de diametrul acesteia, însemnând că stadializarea FIGO folosită pentru carcinomul cu celule scuamuase nu are nici o utilitate pentru acest tip de neoplasme genitale.
Primary mucosal malignant melanoma is a rare and aggressive neoplasm, characterized by a higher aggressiveness and a worse prognosis than its cutaneous counterpart(1). This pathology accounts for only 1.4% of all melanomas, and of these, only about 20% are diagnosed in the female genital tract.
Among all melanomas in women, vulvar, vaginal and cervical malignant melanomas account for 1% to 3 %(2). The most common site is the vulva (70%), followed by the vagina (21%) and the cervix (9%)(3). Malignant melanoma of the female urethra remains rare, accounting for approximately 0.2% of primary melanomas(4).
Malignant melanoma is the second most common cancer of the vagina, representing 2.8% to 5% of all vaginal neoplasms. Vaginal malignant melanoma is extremely lethal, occurring most often in white women population. The average age of the patients is 58-years-old. The most common location of these malignancies is in the lower one-third of the vagina(5), mostly on the anterior vaginal wall(6) (Figure 1).
The presenting symptoms are not particular at all, most of them being related to vaginal bleeding, the appearance of a vaginal tumor and vaginal discharge. The prognosis is among the worst of vaginal malignancies, with a reported 5-year survival rate ranging from 10% to 20%(3).
On rare occasions, melanosis was seen in the cervix. Thus, malignant melanoma may arise de novo in this area. Histopathologically, it is similar to melanoma elsewhere. The prognosis depends on the depth of invasion into the cervical stroma(5). For propper diagnosis, immunohistochemical methods, especially for the exclusion of other primary sites of melanoma, are necessary(7).
There have been reported rare cases of malignant melanoma metastatic to the ovaries.
In these circumstances, the melanomas are commonly widely disseminated. Therefore, surgical removal is to be done for palliation of abdominal or pelvic pain, bleeding, or preventing torsion. Malignant melanoma can rarely arise in a mature cystic teratoma(8).
Primary urethral malignant melanomas are often amelanotic, leading to difficulties in early clinical diagnosis and biopsy delays. The distal urethra is most often involved. The average peak incidence is 65-years-old. Symptoms are associated with the development of a urethral mass, hematuria, local bleeding, perineal pain, dysuria, increased urinary frequency and urinary incontinence. Generally, primary urethral malignant melanomas carry a worse prognosis than their cutaneous counterparts, most probably because of the following reasons: advanced stage at diagnosis, often presenting one decade later, richly vascularized anatomic location and unique genetic mutational spectrum(4).
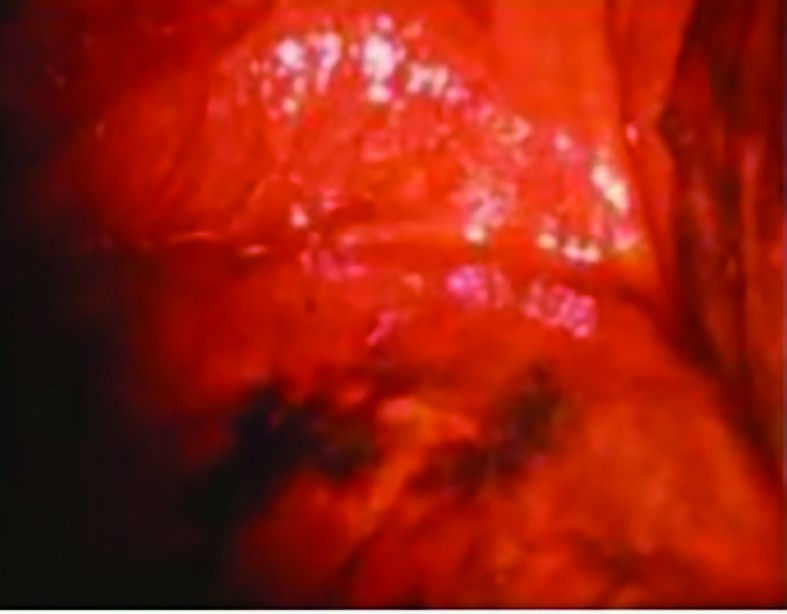
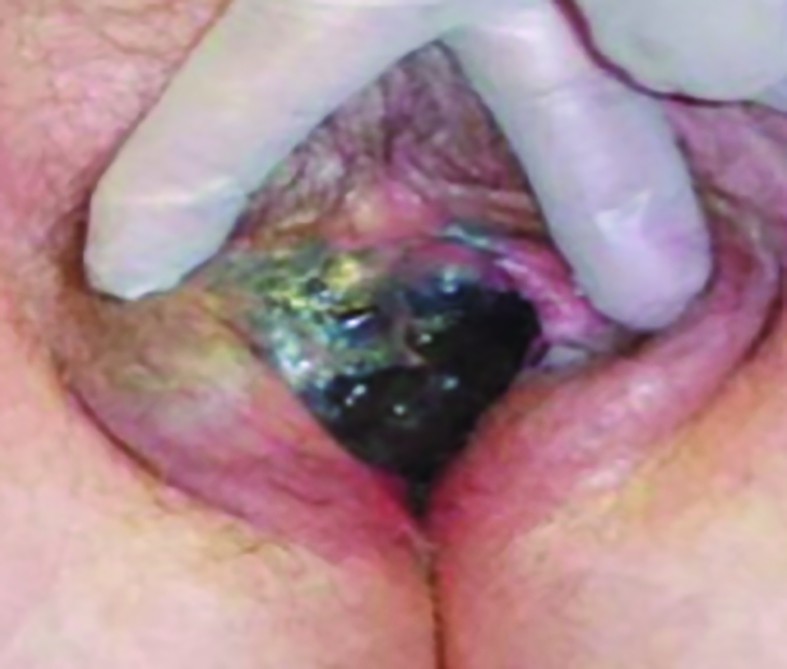
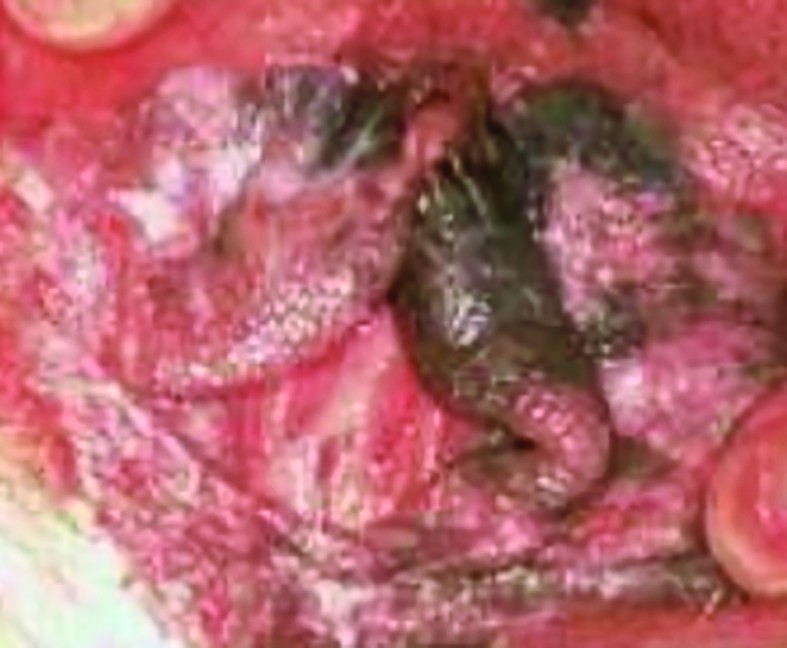
Vulvar malignant melanoma
Malignant melanoma of the vulva represents the most frequent localization of melanotic lesions in the female genital tract and accounts for 2-4% of invasive vulvar cancers(5), having an estimated annual incident rate of 1 per 1,000,000 women (Figures 2 and 3).
The disease can affect women of all ages, but is more common in the older population, with almost half of the patients aged 70 or older. More than 90% of malignant melanomas occur in white women(2).
The histopathological types of vulvar cancer are the following(5): squamous (92%), melanoma (2-4%), basal cell (2-3%), Bartholin gland (adenocarcinoma, squamous cell, transitional cell, adenoid cystic) - 1%, metastatic (1%), verrucous (<1%), sarcoma (<1%).
Clinically, most patients with vulvar malignant melanoma have no symptoms except for a pigmented lesion that seems to be enlarging. Local itching and bleeding may be acused, and just a small percent have groin adenopathy(5).
Pigmented vulvar lesions are present in approximately 1 in 10 women and include melanocytic and nonmelanocytic tumors(9). Various benign pigmented lesions including lentigo simplex, vulvar melanosis, acanthosis nigricans, seborrheic keratosis, and junctional, compound, intradermal, or dysplastic nevi may also be found in these areas. We consider pigmented lesions on the vulva suspicious if the color is blue-black, they present a fuzzy border, are raised or ulcerated or are larger than 1 cm(10). Vulvar nevi, melanosis and melanoma are particularly challenging because of the similarity of their clinical and/or histopathological presentation(9). Thus, tissue sampling is mandatory, and immunohistochemical studies or electron microscopy may help to clarify the diagnosis(2).
Lichen sclerosus is also suspected to cause vulvar malignant melanoma, although it is konown to be an inflammatory dermatosis of unknown origin that presents as a pruriginous whitening lesion of the skin(11), being the most common precursor of HPV-negative squamous cell carcinoma of the vulva.
Three histologic subtypes of vulvar malignant melanoma have been described: superficial spreading melanoma (SS), nodular melanoma (NM), and acral lentiginous melanoma (AL)(3): SS tends to remain relatively superficial early in its development; NM which is the most aggressive, is characterized by a raised lesion that penetrates deeply and may metastasize widely; AL is a flat freckle that may become quite extensive but tends to remain superficial.
The usual clinical form is the superficial spreading pattern and much less common nodular pattern, which is associated with a poorer prognosis generally(1).
Staging of vulvar malignant melanoma
The FIGO staging used for squamous lesions is not applicable to melanomas because the lesions are usually much smaller and the prognosis is related to the depth of tumor invasion rather than to the diameter of the lesion(5). There are several microstaging systems that are used for staging vulvar malignant melanoma, including the Chung, the Clark, and the Breslow systems(3).
The staging system elaborated by Clark (I - Intraepithelial, II - Into papillary dermis, III - Filling dermal papillae, IV - Into reticular dermis, V - Into subcutaneous fat) for cutaneous melanomas is less applicable to vulvar lesions because of the differences in skin morphology (the vulvar skin lacks a well-defined papillary dermis). Breslow measured the thickest portion of the melanoma from the surface of intact epithelium to the deepest point of invasion (I - Intraepithelial, II - <1 mm from granular layer, III - 1.1-2 mm from granular layer, IV - >2 mm from granular layer, V - Into subcutaneous fat). As for the vulva, this system is more adequate.
The revised 2002 American Joint Committee on Cancer (AJCC) staging for cutaneous melanoma replaced the Clark level of invasion by tumor thickness. There have been taken into consideration other important prognostic factors, such as presence of primary tumor ulceration, the number of metastatic lymph nodes, micrometastatic disease based on sentinel lymph node biopsy or elective node dissection, the areas of distant metastatic disease and LDH levels in serum(12).
Using immunohistochemical methods, KIT (CD117) expression was detected. c-KIT is a receptor tyrosine kinase that regulates a large variety of biological interactions, such as cell proliferation, chemotaxis, apoptosis and adhesion in many cell types, including melanocytes, that is why activating KIT mutations are indispensable for tumour growth and progression(13). Some studies demonstrate that KIT mutations appear to be specific for vulvar malignant melanomas, suggesting that in spite of the anatomic proximity, the emergence of vulvar and vaginal malignant melanomas involves different molecular shifts which may be targeted by novel treatment approaches. By contrast, gene mutations for cutaneous melanomas are irrelevant in vulvar malignant melanomas (BRAF, NRAS), indicating that these two diseases have a different origin(14). The identification of mutated genes, such as c-KIT, or increased levels of c-KIT in vulvar malignant melanomas seems to support the current consensus that vulvar melanomas arise de novo from the malignant transformation of a single junctional melanocyte in situ(15). Therefore, c-KIT expression is a valuable predictor of prognosis and survival, especially in tick (>4 mm) melanoma(16).
Treatment of malignant vulvar melanoma
Treatment of vulvar malignant melanoma is controversial, mostly because of the lack of large retrospective studies. Given the fact that mucosal melanoma of the lower genital tract (LGT) is a rare condition, much of the data about treatment and care have been extrapolated from larger studies that include cutaneous and mucosal malignant melanomas of varied origin(17).
The pylon of treatment for these tumors is primary surgical resection, with the goal of achieving negative margins. This objective is often hard to achieve for melanomas of the LGT due to close approximation of tumor to vital anatomic structures such as the bladder and rectum. Attempting to obtain negative margins through an exenterative procedure is not recommended in this setting, as many studies have demonstrated that radical surgery confers no survival benefit(18,19).
It is generally accepted that lesions with less than 1 mm of invasion may be treated with radical local excision alone(20,21). With more invasive lesions, en bloc resection of the primary tumor and regional groin nodes has traditionally been recommended. In the last 15 years, however, radical vulvectomy has been performed less frequently, and survival does not seem to be compromised(22).
Current literature on cutaneous malignant melanoma suggests that a 1-cm margin of skin and subcutaneous tissue is sufficient for the treatment of superficial localized melanoma (Breslow tumor thickness <0.76 mm), whereas a 2-cm margin suffices for intermediate thickness lesions (1 to 4 mm)(21,23). Because the clitoris and labia minora are commonly involved, the vaginourethral margin of resection is a usual site of failure, and care should be taken to obtain an appropriate inner resection margin(24).
As with cutaneous malignant melanoma, it appears that for lesions that tend to remain superficial (Breslow tumor thickness <0.76 mm), the risk for nodal dissemination is so low that routine lymphadenectomy is not indicated as long as the nodes appear clinically to be unaffected. For intermediate thickness (1 to 4 mm) cutaneous malignant melanoma, a randomized controlled trial of elective lymph node dissection versus observation showed a 5-year survival advantage for patients who underwent elective lymph node dissection, who were younger than 60-years-old, and whose tumors were characterized by 1- to 2-mm thickness and no ulcerations(10). Patients with deeply invasive cutaneous malignant melanomas (>4-mm tumor thickness) have a higher risk of regional and systemic metastases and are unlikely to benefit from regional lymphadenectomy(25). Given some of the epidemiologic, histologic, and prognostic differences between vulvar and cutaneous malignant melanoma(26), extrapolating these data to the vulva should be done with caution.
Pelvic node metastases never occur in the absence of groin node metastases(24,27,28). Even more, the prognosis for patients with positive pelvic nodes seems to be very poor that there is no value in performing pelvic lymphadenectomy for this disease.
Neoadjuvant treatment with chemotherapy and/or radiation to cut the need for extensive surgical resection has been limited(19). Dacarbazine (DTIC) is considered the most active single-agent chemotherapy with a response rate of 16%. Standard chemotherapeutics such as Dacarbazine show limited activity in the metastatic setting, and trials of neoadjuvant chemotherapy for patients with resectable melanoma indicate that they are no more likely to respond than those with stage IV disease(29).
Radiotherapy has customarily been used in the palliative support for women with advanced symptomatic disease(30). More recently, the role of immunotherapy in cutaneous malignant melanoma has been explored, with favorable results(31,32). A 2010 phase 3 study investigating the use of ipilimumab - a monoclonal antibody that blocks cytotoxic T lymphocyte-associated antigen 4 (CTLA-4) - in patients with previously treated metastatic melanoma demonstrated a nearly 4-month overall survival advantage as compared to a peptide vaccine alone(31). Recent literature has also pointed to a potential modulation of the immunotherapeutic effect of CTLA-4 blockade with concomitant radiation(33,34). Radiation has a role in the palliative management of brain metastasis and symptomatic bony metastases.
Conclusions
Genital malignant melanoma is a rare condition that has not yet been enough studied. It may be easily misdiagnosed as there are many benign lesions with similar appearance.
It has a different origin from cutaneous malignant melanoma; this fact makes questionable using the same treatment for both of them. Radical surgery confers no survival benefit, vulvectomy being performed less frequently, with survival rates that do not seem to be compromised.
KIT mutations seem to be specific for malignant melanoma of the vulva, therefore studies ought to be made in hope to find an adequate treatment focused on this molecular abnormality. Imunotherapy using CTLA4- blockers is currently the most intensely investigated approach for vulvar malignant melanoma in the hope for a more conservatory treatment.
Nowadays, chemotherapeutics such as Dacarbazine (FDA approved), used alone, have limited use, particularly in metastatic disease. Radiation treatment is used only in palliation for advanced simptomatic disease. There is evidence that genital malignant melanoma - like all the other human melanomas - contains estrogen receptors, therefore, occasional treatment with Tamoxifen has been reported.
Bibliografie
2. Nelson R. Gynecologic Melanoma Different from cutaneous counterpart, Medscape, 2015.
3. Wiliams Gynecology, 2nd Ed, 2012, The McGraw-Hill Co, 32: 803, 815.
4. Urban J, Bayart CB, Taylor JA, Hegde U, Forouhar F, Dadras SS. Urethral duct invasion in female urethral melanoma, Human Pathology: Case Reports 6 (2016):48–51.
5. Berek & Novak’s Gynecology, 14th Ed, 2007, Lippincott, Williams & Wilkins, 34:1446, 1415; 36:1553, 1566.
6. Hacker NF, Eifel PJ, van der Velden J. Cancer of the Vagina, FIGO cancer report, 2015.
7. Mihmanli V, Toprakci G, Cetinkaya N, Kilickaya A, Kamali G. Primary malignant melanoma of the cervix: a case report, Eur J Gynaecol Oncol, 2015; 36(5):607-9.
8. Young RH, Scully RE. Malignant melanoma metastatic to the ovary: a clinicopathologic analysis of 20 cases. Am J Surg Pathol 1991; 15:849–60.
9. Murzaku EC, Penn LA, Hale CS, Pomeranz MK, Polsky D. Vulvar nevi, melanosis and melanoma: an epidemiologic, clinical and histopathologic review, J Am Acad Dermatol, 2014; 71(6):1241-9.
10. Balch CM, Soong SJ, Bartolucci AA, et al. Efficacy of an elective regional lymph node dissection of 1-4 mm thick melanomas for patients 60 years of age or younger. Ann Surg 1996; 224:255–63.
11. Powell JJ, Wojnarowska F. Lichen sclerosus. Lancet. 1999; 353:1777–83.
12. Kim CJ, Reintgen DS, Balch CM for the AJCC Melanoma Staging Committee. The new melanoma staging system. Cancer Control, 2002; 9:9–15.
13. Heinzelmann-Schwartz VA, Nixdorf S, Valadan M, Diczbalis M, Olivier J, Otton G, Fedier A, Hacker NF, Scurry JP. A clinicopathological review of 33 patients with vulvar melanoma identifies c-KIT as a prognostic marker. Int J Mol Med, 2014 ; 33(4): 784–94.
14. Aulmann S, Sinn HP, Penzel R, Gilks CB, Schott S, Hassel JC, Schmidt D, Kommoss F, Schirmacher P, Kommoss S. Comparison of molecular abnormalities in vulvar and vaginal melanomas, Epub, 2014.
15. Blessing K, Kernohan NM, Park KG. Subungual malignant melanoma: clinicopathological features of 100 cases. Histopathology. 1999; 19:425–9.
16. Salcedo-Hernández RA, Lino-Silva LS, Isla-Ortiz D, Posada-Torres JA, Chanona-Vilchis JG, Pérez-Montiel DM, Aguilar-Romero JM, Domínguez-Rodríguez JA, Herrera-Gómez A. Sateliosis and CD 117 imunohistochemical expression correlates with poor outcome in thick vulvar melanoma, G Ital Dermatol Venereol, 2016.
17. Schiavone MB, Broach V, Shoushtari AN, Richard D. Carvajal RD, Alektiar K, Kollmeier MA, Abu-Rustum NR, Leitao MM Jr. Combined immunotherapy and radiation for treatment of mucosal melanomas of the lower genital tract, Gynecologic Oncology Reports, 2016; 16:42–6.
18. Brand E, Fu YS, Lagasse LD, Berek JS. Vulvovaginal melanoma: report of seven cases and literature review, Gynecol Oncol, 1989; 33: 54–60.
19. Leitao Jr MM. Management of Vulvar and Vaginal Melanomas: Current and Future Strategies. Am Soc Clin Oncol Educ Book, 2014;e277–e281.
20. Chung AF, Woodruff JM, Lewis JL Jr. Malignant melanoma of the vulva: a report of 44 cases. Obstet Gynecol, 1975;45:638–46.
21. Sturgeon SR, Brinton LA, Devesa SS, et al. In situ and invasive vulvar cancer incidence trends (1973 to 1987). Am J Obstet Gynecol, 1992; 166:1482–5.
22. Rose PG, Piver MS, Tsukada Y, et al. Conservative therapy for melanoma of the vulva. Am J Obstet Gynecol 1988; 159:52–5.
23. Veronesi U, Cascinelli N. Narrow excision (1-cm margin): a safe procedure for thin cutaneous melanoma. Arch Surg, 1991; 126:438–41.
24. Morrow CP, Rutledge FN. Melanoma of the vulva. Obstet Gynecol, 1972; 39:745–52.
25. Balch CM, Soong SJ, Milton GW, et al. A comparison of prognostic factors and surgical results in 1,786 patients with localized (stage I) melanoma treated in Alabama, USA, and New South Wales, Australia. Ann Surg, 1982; 196:677–84.
26. Dunton JD, Berd D. Vulvar melanoma, biologically different from other cutaneous melanomas. Lancet, 1999; 354:2013–4.
27. Beller U, Demopoulos RI, Beckman EM. Vulvovaginal melanoma: a clinicopathologic study. J Reprod Med, 1986; 31:315–9.
28. Jaramillo BA, Ganjei P, Averette HE, et al. Malignant melanoma of the vulva. Obstet Gynecol, 1985; 66: 398–401.
29. Shah GD, Socci ND, Gold JS, Wolchok JD, Carvajal, RD, Panageas KS, et al. Phase II trial of neoadjuvant temozolomide in resectable melanoma patients. Ann. Oncol, 2010; 21:1718–22.
30. Huguenin PU, Kieser S, Glanzmann C, Capaul R, Lutolf UM. Radiotherapy for metastatic carcinomas of the kidney or melanomas: an analysis using palliative end points. Int. J. Radiat. Oncol Biol Phys, 1998; 41:401–5.
31. Hodi FS, O’Day SJ, McDermott DF, Weber RW, Sosman JA, Maanen JB, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med, 2010; 363:711–23.
32. Larkin J, Hodi FS, Wolchok JD.Combined nivolumab and ipilimumab or monotherapy in untreated melanoma. N Engl J Med, 2015; 373:23–34.
33. Postow MA, Callahan MK, Barker CA, Yamada Y, Yuan J, Kitano S, et al. Immunologic correlates of the abscopal effect in a patient with melanoma. N Engl J Med, 2012; 366: 925–31.
34. Twyman-Saint Victor C, Rech AJ, Maity A, Rengan R, Pauken KE, Stelekati E, et al. Radiation and dual checkpoint blockade activate non-redundant immune mechanisms in cancer. Nature, 2015; 520: 373–7.
Articole din ediţiile anterioare
Impactul fetal şi neonatal al metodelor de diagnostic şi al chimioterapiei în sarcină la gravidele cu afecţiuni oncologice
Afecţiunile oncologice din timpul sarcinii sunt diagnosticate mult mai frecvent în ultimii ani datorită metodelor de diagnostic avansate. Odată c...
Tumori ovariene cu celule granuloase
Tumora ovariană cu celule granuloase (TCG) la adulţi este deseori un neoplasm celular stromal, activ hormonal, care se distinge prin capacitatea sa...
Corelaţii clinice şi paraclinice în managementul neoplaziei intraepiteliale cervicale
Tratamentul neoplaziei intraepiteliale cervicale vizează eliminarea zonei de joncţiune, inclusiv a ţesuturilor anormale. Acest lucru se real...
SARS-CoV-2 şi sarcina: leziuni placentare şi conduită terapeutică. Prezentare de caz
Prezentăm cazul unei gravide care s-a infectat în primul trimestru de sarcină cu SARS-CoV-2, prezentând o formă moderată de boală. După puseul acu...