Pregnancy is a time characterized by physiological changes that are related to a high vulnerability of the oral health. The variation of the prevalence of changes of the periodontal status correlated to pregnancy can be related to specific particularities of the different investigated populations (cultural factors, oral hygiene habits, concentration and distribution of estrogen and progesterone receptors in gingival fibroblasts and periodontal ligament cells). The onset of the pregnancy gingivitis is related to the increased levels of estrogen and progesterone that change the levels of periodontal subgingival pathogens, increase the levels of prostaglandins, modify the immunologic response by influencing the levels of some cytokines and interleukines, and decrease the antioxidants levels in gingival crevicular fluid. Also, localized or generalized gingival enlargement can be related to pregnancy, as hormonal changes increase the response to local irritants (bacteria, calculus). The authors present a relevant case from their casuistry. In the conclusion of our study, the variation of the prevalence of changes of the periodontal status correlated to pregnancy can be related to specific particularities of the different investigated populations, and also the dental practitioners need to provide in-depth knowledge to the pregnant women about the changes that occur in the oral cavity status during pregnancy.
Modificările statusului periodontal în sarcină – review şi prezentare a unui caz clinic
Pregnancy and periodontal status changes – review and a clinical case
First published: 08 martie 2019
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/Gine.23.1.2019.2222
Abstract
Rezumat
Sarcina este o perioadă caracterizată de schimbări fiziologice legate de o vulnerabilitate ridicată a sănătăţii orale. Variaţia prevalenţei modificărilor stării parodontale legate de sarcină poate fi corelată cu particularităţile specifice ale diferitelor populaţii investigate (factori culturali, obiceiuri de igienă orală, concentrarea şi distribuţia receptorilor de estrogen şi progesteron în fibroblastele gingivale şi celulele ligamentului parodontal). Debutul gingivitei în sarcină este legat de creşterea nivelului de estrogen şi progesteron, care modifică nivelurile agenţilor patogeni parodonţiali, creşte nivelul de prostaglandine, modifică răspunsul imun prin influenţarea nivelurilor unor citokine şi interleukine şi reduce nivelurile antioxidante din lichidul coletului gingival. De asemenea, edemul gingival localizat sau generalizat poate fi legat de sarcină, deoarece modificările hormonale cresc răspunsul la iritanţii locali (bacterii, tartru dentar). Autorii prezintă, totodată, un caz relevant din propria cazuistică. Ca o concluzie a studiului nostru, variaţia prevalenţei schimbărilor de status parodontal legate de sarcină poate fi corelată cu particularităţile specifice ale diferitelor populaţii investigate şi, de asemenea, este esenţial ca medicii stomatologi să ofere cunoştinţe detaliate femeilor însărcinate despre schimbările care apar în starea cavităţii orale în timpul sarcinii.
Pregnancy is a period characterized by physiological changes that are related to a high vulnerability of the oral health(1). Moreover, these transient changes onset physical oral signs and symptoms that can affect the people’s perceptions and interactions with others, as well as productivity and well-being state(2).
The most frequent oral condition related to pregnancy is pregnancy gingivitis, an inflammation of the gingival tissues associated to bleeding, swelling and tenderness. The different results of the researches performed to assess the prevalence of this condition during pregnancy reflects the specific features of the different studied populations(3), as pregnancy gingivitis affects 95% of women in India(4), 86.2% of women in Thailand(5), but only 47% of women in Brazil(6). Pregnancy gingivitis is a condition with a etiology that is not fully known, despite researches which revealed to be related to the increased levels of estrogen and progesterone that are supposed to be responsible for gingivitis progression due to changes of subgingival microbiota and immunologic mediators in gingival tissues and periodontal ligaments(7). The literature data could not provide an accurate relation between pregnancy and the changes of gingival tissues, as the difference of populations groups, criteria defining periodontal disease, clinical measurements, as well as the numbers of teeth or patients examined may distort the results of any review or meta-analysis(7). It can be associated with the increasing of the periodontal probing depths, bleeding upon probing or mechanical stimulation, and the increase of the gingival crevicular fluid flow, changes that disappear after birth(8). Even pregnant women with excellent oral hygiene, low plaque index and healthy periodontium showed an increase of gingival inflammation, especially in the first and second trimesters, that was not related to dental plaque; the gingival inflammation decrease significantly after birth, proving that the increase of the inflammatory changes of gingival tissues was induced by pregnancy(9).
Regarding the etiology, at the beginning of 1970s it was for the first time when the increase of estrogen and progesterone levels in pregnant women were correlated with the increased inflammatory processes in the gingival tissues(10).
It is known that the production of estrogen and progesterone increases progressively during pregnancy, reaching by the end of the third trimester peak plasma levels of 100 and 6 ng/mL, respectively, equivalent with 10 and 30 times from the normal levels measured during the menstrual cycle(11). Fundamental researches proved the existence of receptors for estrogen and progesterone in human gingival tissues. Both Kawahara et al. and Jönsson et al. have reported that human gingival fibroblasts expressed high signal for ER-β subtype of estrogen, as ER-β immunoreactivity was observed in the nuclei of about 40% of cultured human periodontal ligament cells. This result suggested that estrogen influences the functional properties of gingival fibroblasts and periodontal ligament cells(12,13). Another research concluded that the receptor for ER-β is localized not only in nuclei, but also in the mitochondria of human periodontal ligament cells. The conclusion of this study was that estrogen influences mitochondrial function and energy metabolism in human periodontal ligament cells(14). Regarding the effects of progesterone, researches found different results between different populations. While a study performed in Sweden revealed the absence of expression of receptors for progesterone in periodontal ligament cells(15), another study performed in China found the expression of progesterone receptor in human periodontal ligament cells(16). The variation of the expression of estrogen receptors and progesterone receptors in different populations could be an interesting research issue. Further researches performed in various geographic areas, by using reverse transcriptase-polymerase chain reaction and immunocytochemistry, could explain the wide range of the pregnancy gingivitis prevalence determined by epidemiological studies.
Another factor implied in the pregnancy gingivitis could be the change of subgingival pathogenic periodontal microbiota directly related to the increase of estrogen and progesterone levels. One study found that estradiol and progesterone influence the fumarate reductase system of subspecies of Bacteroides intermedius, altering the metabolic pathways of these pathogens and thus the entire ecology of subgingival microbiota(17). Another study reported a 55-fold increase of the level of Bacteroides species during pregnancy compared to non-pregnant women and a 16-fold increase in the level of Bacteroides species in women taking contraceptives comparing to control(18). Also, other periodontal pathogenic species, Porphyromonas gingivalis and Prevotella intermedia, were positively correlated with the increase of estradiol and progesterone levels during pregnancy(19). Another periodontal pathogenic bacterium, Fusobacterium nucleatum, is able to invade not only the gingival epithelial cells, the gingival fibroblasts and the periodontal ligament fibroblasts, but also other different types of human cells, and it was associated to term stillbirth, as the bacteria originated from the mother’s subgingival plaque and translocated to the placenta and fetus caused acute inflammation and fetal demise(20).
The third mechanism responsible for pregnancy gingivitis is some degree of immunosuppression (which minimizes the risk of fetal rejection) due to impaired neutrophil functions by estrogen and progesterone modulation and considered to be linked to the increase of the inflammatory gingival processes(21). Also, gingival fibroblasts and periodontal ligament cells, participants in the oral immune defense system, seem to produce chemokines and cytokines when exposed to minimal levels of periodontal bacteria or to inflammatory cytokines, aggravating the evolution of gingival inflammation or the periodontal disease(22). Progesterone also influences the increase of gingival inflammatory processes and the onset of pregnancy gingivitis by the increase of the production of prostaglandin E2 by LPS-stimulated human monocytes and by reducing the secretion of IL-6 by human gingival fibroblasts(23,24).
The fourth mechanism that influences gingival status in pregnancy, the reduction of antioxidants and superoxide dismutase enzyme levels in gingival crevicular fluid, was demonstrated in a study which compared the pregnant patients with chronic periodontitis, pregnant patients with gingivitis, and periodontally healthy pregnant women (control group), non-pregnant women with chronic periodontitis, and non-pregnant periodontally healthy women. The results showed that systemic and local antioxidants levels decreased in pregnancy and periodontitis, while antioxidants defense agents and superoxide dismutase enzyme reached the lowest level in the last phase of pregnancy, which was also associated with the increase of the inflammatory gingival processes and with the alteration of the periodontal status. The study demonstrated that the reduction of the antioxidants levels was influenced more by pregnancy and less by periodontal inflammation, indicating that pregnancy is a risk factor for periodontal status(25).
Another change of the periodontal status is related to the onset of “pregnancy tumors” during the second trimester of pregnancy, due to the increase of periodontal bacteria levels in gingival sulcus in relation to less proper oral hygiene. The most frequent is localized or generalized gingival hyperplasia, influenced by the hormonal changes that increase the response to local irritants (bacteria, calculus). The interproximal gingiva, more frequently affected, is soft and friable, bright red or magenta, with a smooth, shiny surface, associated with bleeding, spontaneous or provoked by mild stimulation. The gingival enlargement may be reduced spontaneously after birth. However, some of these tumors may not disappear after birth and require additional surgical procedures (classic or laser-assisted) of any fibrotic remanants(26).
Clinical case
We present the gingival-periodontal condition of a patient from our practice, a 27-year-old woman in the sixth month of pregnancy who presented herself under dental check-up due to the inability to perform a correct dental brushing. The main accusations are related to mucosal bleeding gums at the smallest touch with the brush. Gingival mucous membranes have a slightly swollen, glossy and bleeding appearance both in the frontal area (Figure 1 and Figure 3) and in the lateral area (Figure 2).
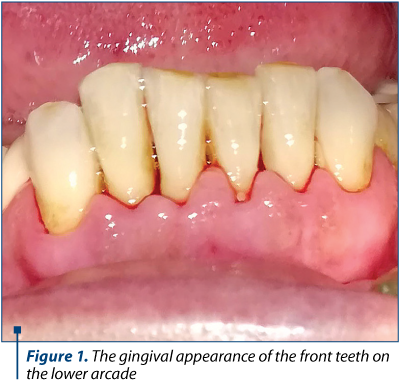
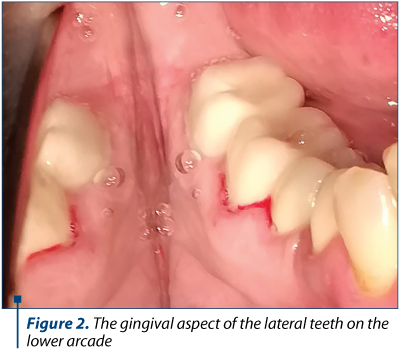
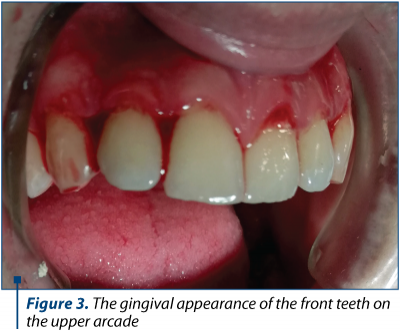
Considering that the patient is in her second trimester of pregnancy, during this period minor dental-periodontal care treatments are allowed, and general oral hygiene (ultrasonic scrubbing and professional brushing) has been performed, followed by advice related to a correct maintenance of oral cavity hygiene and programming at periodic control in the postnatal period. It was recommended to use a manual brush instead of a mechanical brush, thus allowing a more appropriate control over the mechanical action. Considering the physiological state of the future mother, the painful odontal treatments and possible prosthetic restorations were timed, in agreement with the patient, for the postnatal period.
In conclusion, the variation of the prevalence of changes of the periodontal status related to pregnancy can be correlated to specific particularities of the different investigated populations (cultural factors, oral hygiene habits, concentration and distribution of estrogen and progesterone receptors in gingival fibroblasts and periodontal ligament cells).
The proper measure to counteract the rapid evolution of the more severe gingivitis is the attendance of the pregnant women to frequent dental sessions for professional cleaning, as well as proper oral hygiene measures.
Also, the dental practitioners need to provide in-depth knowledge to the pregnant women about the changes that occur in the oral cavity status during the pregnancy and how to prevent the onset or the worsening of the existent oral conditions.
Compliance with ethics requirements:
The authors declare no conflict of interest regarding this article.
The authors declare that all the procedures and experiments of this study respect the ethical standards in the Helsinki Declaration of 1975, as revised in 2008, as well as the national law. The informed consent was obtained from the patient included in the study.
No funding for this study.
Bibliografie
- Hartnett E, Haber J, Krainovich-Miller B, Bella A, Vasilyeva A, Lange Kessler J. Oral Health in Pregnancy. J Obstet Gynecol Neonatal Nurs. 2016; 45(4):565-73.
- Dellinger TM, Livingston HM. Pregnancy: physiologic changes and considerations for dental patients. Dent Clin North Am. 2006; 50(4):677-97.
- Vogt M, Sallum AW, Cecatti JG, Morais SS. Factors associated with the prevalence of periodontal disease in low-risk pregnant women. Reprod Health. 2012; 24(9):3.
- Gupta R, Acharya AK. Oral Health Status and Treatment Needs among Pregnant Women of Raichur District, India: A Population Based Cross-Sectional Study. Scientifica (Cairo). 2016; 2016: 9860387.
- Nuamah I, Annan BD. Periodontal status and oral hygiene practices of pregnant and non-pregnant women. East African Medical Journal. 1998; 75(12):712–4.
- Rakchanok N, Amporn D, Yoshida Y, Harun-Or-Rashid M, Sakamoto J. Dental caries and gingivitis among pregnant and non-pregnant women in Chiang Mai, Thailand. Nagoya J Med Sci. 2010;72(1-2):43–50.
- Wu M, Chen SW, Jiang SY. Relationship between gingival inflammation and pregnancy. Mediators Inflamm. 2015; 2015:623427.
- Mariotti A. Sex steroid hormones and cell dynamics in the periodontium. Crit Rev Oral Biol Med. 1994;5(1):27-53.
- Figuero E, Carrillo-de-Albornoz A, Herrera D, Bascones-Martínez A. Gingival changes during pregnancy: I. Influence of hormonal variations on clinical and immunological parameters. J Clin Periodontol. 2010; 37(3):220-9.
- Deasy MJ, Vogel RI. Female sex hormonal factors in periodontal disease. Ann Dent. 1976; 35(3):42-6.
- Zachariasen RD. The effect of elevated ovarian hormones on periodontal health: oral contraceptives and pregnancy. Women Health. 1993; 20(2):21-30.
- Kawahara K, Shimazu A. Expression and intracellular localization of progesterone receptors in cultured human gingival fibroblasts. J Periodontal Res. 2003; 38(3):242-6.
- Jönsson D, Andersson G, Ekblad E, Liang M, Bratthall G, Nilsson BO. Immunocytochemical demonstration of estrogen receptor beta in human periodontal ligament cells. Arch Oral Biol. 2004; 49(1):85-8.
- Jönsson D, Nilsson J, Odenlund M, Bratthall G, Broman J, Ekblad E, Lydrup M, Nilsson BO. Demonstration of mitochondrial oestrogen receptor beta and oestrogen-induced attenuation of cytochrome c oxidase subunit I expression in human periodontal ligament cells. Arch Oral Biol. 2007; 52(7):669-76.
- Jönsson D, Wahlin A, Idvall I, Johnsson I, Bratthall G, Nilsson BO. Differential effects of estrogen on DNA synthesis in human periodontal ligament and breast cancer cells. J Periodontal Res. 2005; 40(5):401-6.
- Yuan G, Cai C, Dai J, Liu Y, Zhang R, Dai Y, Wen L, Ding Y. Progesterone modulates the proliferation and differentiation of human periodontal ligament cells. Calcif Tissue Int. 2010; 87(2):158-67.
- Kornman KS, Loesche WJ. Effects of estradiol and progesterone on Bacteroides melaninogenicus and Bacteroides gingivalis. Infect Immun. 1982; 35(1):256-63.
- Jensen J, Liljemark W, Bloomquist C. The effect of female sex hormones on subgingival plaque. J Periodontol. 1981; 52(10):599-602
- Carrillo-de-Albornoz A, Figuero E, Herrera D, Bascones-Martínez A. Gingival changes during pregnancy: II. Influence of hormonal variations on the subgingival biofilm. J Clin Periodontol. 2010; 37(3):230-40.
- Han YW, Fardini Y, Chen C, Iacampo KG, Peraino VA, Shamonki JM, Redline RW. Term stillbirth caused by oral Fusobacterium nucleatum. Obstet Gynecol. 2010; 115(2 Pt 2):442-5.
- Krause PJ, Ingardia CJ, Pontius LT, Malech HL, LoBello TM, Maderazo EG. Host defense during pregnancy: neutrophil chemotaxis and adherence. Am J Obstet Gynecol. 1987; 157(2):274-80.
- Lapp CA, Lapp DF. Analysis of interleukin-activated human gingival fibroblasts: modulation of chemokine responses by female hormones. J Periodontol. 2005; 76(5):803-12.
- Miyagi M, Morishita M, Iwamoto Y. Effects of sex hormones on production of prostaglandin E2 by human peripheral monocytes. J Periodontol. 1993; 64(11):1075-8.
- Morishita M, Miyagi M, Iwamoto Y. Effects of sex hormones on production of interleukin-1 by human peripheral monocytes. J Periodontol. 1999; 70(7):757-60.
- Akalin FA, Baltacioğlu E, Alver A, Karabulut E. Total antioxidant capacity and superoxide dismutase activity levels in serum and gingival crevicular fluid in pregnant women with chronic periodontitis. J Periodontol. 2009; 80(3):457-67.
- Agrawal AA. Gingival enlargements: Differential diagnosis and review of literature. World J Clin Cases. 2015; 3(9):779–88.
Articole din ediţiile anterioare
Complicaţii materne şi neonatale în sarcina gemelară obţinută spontan versus sarcina gemelară obţinută prin FIV
Acest studiu a avut ca obiectiv identificarea complicaţiilor materne şi neonatale din sarcina gemelară obţinută spontan, comparativ cu sarcina gem...
Diagnosticul precoce al sarcinii ovariene – prezentare de caz
Sarcina ovariană este o varietate foarte rară de sarcină ectopică şi reprezintă o provocare medicală în ceea ce priveşte managementul acesteia...
Trombofilia ereditară şi complicaţiile obstetricale mediate de placentă
Trombofilia se referă la o predispoziţie către o stare de hipercoagulabilitate şi poate fi identificată în 30-50% din cazurile de tromboembolism ve...
Progesteron, vitamine, metformin şi aspirina administrate în sarcină - „mit” sau medicină bazată pe dovezi?
Deşi sarcina este o stare fiziologică normală şi naşterea este un fenomen natural, posibilele complicaţii ale acestora, deşi rare, sunt suficient d...