Thrombocytopenia (a platelet value of less than 150x109/L) is a very important hematologic disorder in pregnancy and is encountered in 7-10% of the pregnant women population. There are multiple causes of thrombocytopenia, related or not to pregnancy. The focus of this paper is immune thrombocytopenia (ITP) in pregnancy, and we review the pathological mechanisms, the diagnostic methods, the management, the treatment options and neonatal outcome by analysing and discussing the most recent literature data. It is mandatory to accurately diagnose, manage and treat hematologic pathologies during pregnancy. Immune thrombocytopenia in pregnancy may be an apparently limited disorder, but it has often proven to be a challenge for obstetricians, hematologists and neonatologists, who need to work together and supervise these cases in order to prevent complications and ensure the best outcome available for both mother and newborn.
Trombocitopenia imună în sarcină – o revizuire a literaturii
Immune thrombocytopenia in pregnancy – a literature review
First published: 28 septembrie 2018
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/Gine.21.3.2018.1945
Abstract
Rezumat
Trombocitopenia (numărul trombocitelor sub 150x109/L) este o afecţiune hematologică importantă în timpul sarcinii şi este întâlnită cu o frecvenţă de 7-10% în populaţia feminină gravidă. Sunt multe cauze ale trombocitopeniei imune, legate sau nu de sarcină. Subiectul acestui articol este trombocitopenia în sarcină, revederea mecanismelor patologice şi a metodelor diagnostice, a conduitei şi a opţiunilor terapeutice, precum şi a rezultatelor neonatale, prin analiza şi discuţia celor mai recente date din literatură. Este obligatoriu ca diagnosticul, conduita şi tratamentul afecţiunilor hematologice în sarcină să aibă o acurateţe maximă. Trombocitopenia imună în sarcină pare să fie o afecţiune autolimitantă, dar se dovedeşte adesea o provocare pentru obstetrician, hematolog şi neonatolog, care trebuie să lucreze în echipă, prevenind complicaţiile şi asigurând cele mai bune rezultate posibile atât pentru mamă, cât şi pentru nou-născut.
Introduction
Thrombocytopenia (a platelet value of less than 150x109/L) is a very important hematologic disorder in pregnancy, being encountered in 7-10% of the pregnant women population.
Although platelets value have a physiologic tendency to decrease in the normal pregnancy, and light bleeding episodes may occur even in less severe cases of thrombocytopenia, the patient must be investigated thoroughly in order to prevent major complications (Table 1)(1,2).

Our main focus is the subject of immune thrombocytopenia (ITP). Pregnancy may induce or complicate a pre-existing ITP. Usually, after delivery the platelet count returns to the values recorded before pregnancy. It is important to clearly diagnose ITP and to correctly evaluate the patient in order to exclude other pathologies.
The condition is encountered in 1 to 10 in 10.000 pregnancies, and a treatment is required in about 30% of the patients(4,9).
Mechanism of action and diagnostic methods
Due to the fact that over the recent years the pathophysiology of the disorder has been better understood, the International Work Group (IWG) decided to rename it immune thrombocytopenia. Although patients may present petechiae and bruising, most patients do no develop purpura. Unlike thrombotic thrombocytopenic purpura (TTP), where the main mechanism is represented by autoantibodies directed to ADAMTS13 protease(10), primary ITP is an acquired immune disorder which is characterized by pathologic platelet antibodies, impaired megakaryocytopoiesis and T-cell mediated destruction of platelets(4,8). The autoantibodies recognize specific epitopes such as fibrinogen receptor, glycoprotein Ib/IX and IIb/IIIa, Ia/IIa, V and IV. After binding to the macrophages’ Fcg receptors, the platelets are then destroyed in the reticuloendothelial system of the spleen. Also, it has been discovered that certain changes in T cell population, such as the loss of regulatory T cells and an increase in T-proinflammatory responses, are responsible for low platelet value. Recent studies demonstrated the existence of cytotoxic CD8+ T cells which can directly destroy platelets. Furthermore, it has been proven that some antiplatelet antibodies (GP Ib/Ix Ab) determine the apoptosis of megakaryocytes, thus altering differentiation and maturation and diminishing the production of new platelets(2,9,11).
Because gestational thrombocytopenia accounts for about 70-80% of the cases, it must be differentiated from ITP, especially because many patients are diagnosed during pregnancy (Table 2).

About 90% of women with ITP have platelet-associated IgG; however, this is not a specific marker because the same marker tested positive in patients with gestational thrombocytopenia and preeclampsia (Table 3)(12).
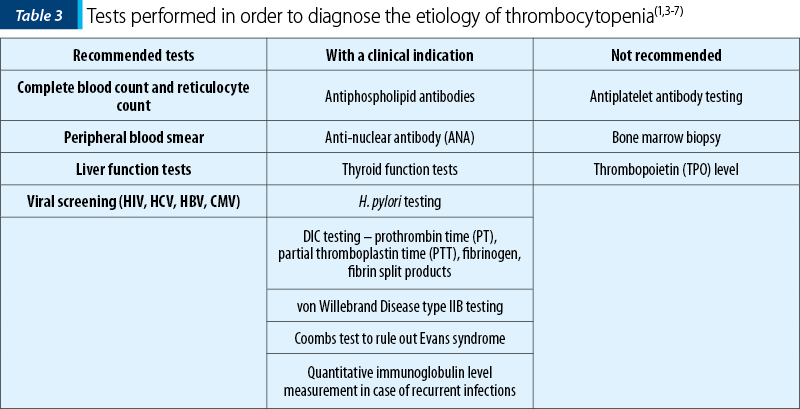
Zhang et al. demonstrated in their study that thrombopoietin levels in pregnant patients suffering from ITP were higher than the values found in patients diagnosed with GT (29 out of 35 ITP patients had thrombopoietin levels of >500 pg/mL, whereas all the GT patients had TPO values of under 500 pg/mL)(13).
A study carried out on 446 pregnant women with ITP reported a higher risk for stillbirth or fetal loss, premature delivery and specific congenital anomalies (ostium secundum) for cases diagnosed with ITP before pregnancy(14).
Management and treatment options
Throughout the pregnancy, the physician must keep a close eye on the patient’s platelet values, and if bleeding symptoms start to appear. No drug therapy is required if the platelet level is >30x109/L and in the absence of bleeding symptoms. If bleeding symptoms are present and platelet values are less than 20-30x109/L in the third trimester or less than 20x109/L in any trimester, therapy is required, glucocorticoids being the first line of treatment and in a minimal dose, due to their side effects, with a starting dose of 10 mg/day. It has been reported that approximately 70% of the ITP cases which did not have an adequate response to corticoid therapy are likely to be efficiently treated using immunoglobulins (IVIG). These are recommended if glucocorticoids fail or if a fast rise of platelets is necessary. Usually, a dose of 1 mg/kg per day for two days or 400 mg/kg per day for five days is recommended alone or in association with low-dose prednisone(15). However, a retrospective study regarding first-line therapy choices in a population of 91 women reported that there were no significant differences between the platelet count response in patients who were given an initial treatment of either corticosteroids (77x109/L), or IVIG (69x109/L)(16).
Studies show that high-risk pregnancies (previous pregnancies with neonates who suffered from an intracranial hemorrhage) should be administered 1 g/kg/week of IVIG, with the treatment ideally starting somewhere between the 12th and the 20th week of gestation. On the other hand, in the case of standard pregnancies, the ideal interval to commence the treatment would be between the 20th and the 24th week of gestation, in association with steroids if needed(17).
The alternative and second-line therapies come with a high risk. It has been proven that anti-(Rh) D can lead to hemolytic reactions and is not used in patients with low values of hemoglobin (<10 g/dl). Rituximab, which has a long half-life, can cross the placenta and it has been shown in studies along the years that the recommendation for women is to avoid pregnancy for six months – one year after being administered the drug. There are studies that highlighted the fact that the use of rituximab during pregnancy could cause prolonged B-cell lymphocytopenia, leading to a delayed vaccination for newborns(3,4,18,19).
Dapsone has been shown to increase the risk for a neonate to develop hemolytic anemia and hyperbilirubinemia. Other drugs, such as vinca alkaloids, cyclophoshamide or danazol, are not allowed during pregnancy(18,20).
Some studies reported the use of new methods of therapy by administering recombinant human thrombopoietin (rhTPO) or thrombopoietin mimetic (romiplostim, eltrombopag) for patients who did not respond to the initial treatment with either corticoids, or intravenous immunoglobulin. Recombinant human thrombopoietin (rhTPO) is a full-length glycosylated TPO with a molecular weight of 90k Daltons and has the advantage of not being able to cross the placental barrier, unlike thrombopoietin mimetics, such as eltrombopag, which have a low molecular weight. Although it has a molecular weight of 60k Daltons, romiplostim binds to Fc receptors, giving it long circulating half-life and also the ability to cross the placenta. The multicenter prospective study presenting the effects of rhTPO given as therapy for a population of 31 pregnant patients diagnosed with ITP reported that the treatment is “potentially safe, effective and fast-acting” for cases with inadequate response to first-line therapy and transfusions with thrombocytes. Out of 31 patients, 23 responded to the 14-day treatment with 300 U/kg, achieving a platelet level of >100x109/L, while the other 13 had values ranging from 30 to 100x109/L(21).
A 28-year-old multidrug resistant patient received increased romiplostim doses in association with corticotherapy with dexamethasone in order to maintain a stable level of platelets before having induced labor at 33 weeks and 6 days of gestation because of increasing episodes of thrombocytopenia. She gave birth to a 1.910 kg male neonate, with an Apgar score of 8/9 and a platelet level of 70x109/L at birth, which decreased after 8 hours postpartum to 33x109/L. Also, he was diagnosed with a grade III intraventricular hemorrhage (which was a result of maternal antiplatelet antibodies), and received transfusion with a unit of thrombocytes(22).
Eltrombopag was introduced to a patient who, even though she was on treatment of 8 mg/day of prednisolone, had platelet values under 10x109/L. Prior to this, she suffered two induced abortions as a result of uncontrolled ITP, although the treatment included IVIG, oral prednisolone at 20 mg/day and platelet transfusions. The patient refused a splenectomy procedure and was administered 12.5 mg/day of eltrombopag. She became pregnant for a third time and continued the use of eltrombopag throughout her pregnancy, and also took 2.5 mg/day of prednisolone, thus establishing an adequate platelet count. The dose of prednisolone was increased at 36 weeks because of a platelet value of 19x109/L. She had a caesarean section due to preeclampsia and delivered a 1,670 g newborn with no reported malformations(23).
Another use of eltrombopag during pregnancy was reported in the case of a patient with a history of recurrent abortions who at 26 weeks accused multiple episodes of mucosal bleeding. Eltrombopag was introduced at 29 weeks first at a dose of 25 mg/day for one week. The dose was then doubled and a platelet value of 30x109/L was achieved, with no episodes of bleeding being reported. She gave birth at 36 weeks, following a preterm-induced vaginal delivery, to a 1860 g newborn with no apparent malformations(24).
Rezk et al. suggested that a splenectomy procedure performed for patients diagnosed with ITP before achieving a pregnancy may have obstetric benefits by “lowering the rates of bleeding episodes, severe thrombocytopenia in the antenatal period, the need for therapy, sequelae of steroid therapy, occurrence of postpartum hemorrhage and defective lactation”(25). However, even though such a surgical procedure should be avoided in pregnancy, it may be required in cases that do not have an adequate platelet value using first-line therapy options. The ideal moment to perform such a procedure is in the second trimester and whenever possible, using a laparoscopic approach. Such a case was presented by Griffiths et al., where a 35-year-old patient underwent a laparoscopic splenectomy at 20 weeks of gestation, after which she continued her prednisone and intermittent IVIG treatment until she spontaneously delivered at 34 weeks(26). Another important issue to be pinpointed is the fact that women diagnosed with ITP who benefited from a splenectomy should continue penicillin prophylaxis throughout the pregnancy(27).
A platelet value of less than 20x109/L, suggesting severe thrombocytopenia, has been shown to increase the risk of intracranial hemorrhage, and it is important to monitor and handle pregnancy complications like emesis and constipation, which could lead to an increase intracranial pressure(28).
There were cases mentioned in the literature where platelet counts increased after a successful Helicobacter pylori (HP) eradication therapy administered after 12 weeks of gestation in four patients suffering from ITP(29).
The treatment options for ITP are summarized in Table 4.
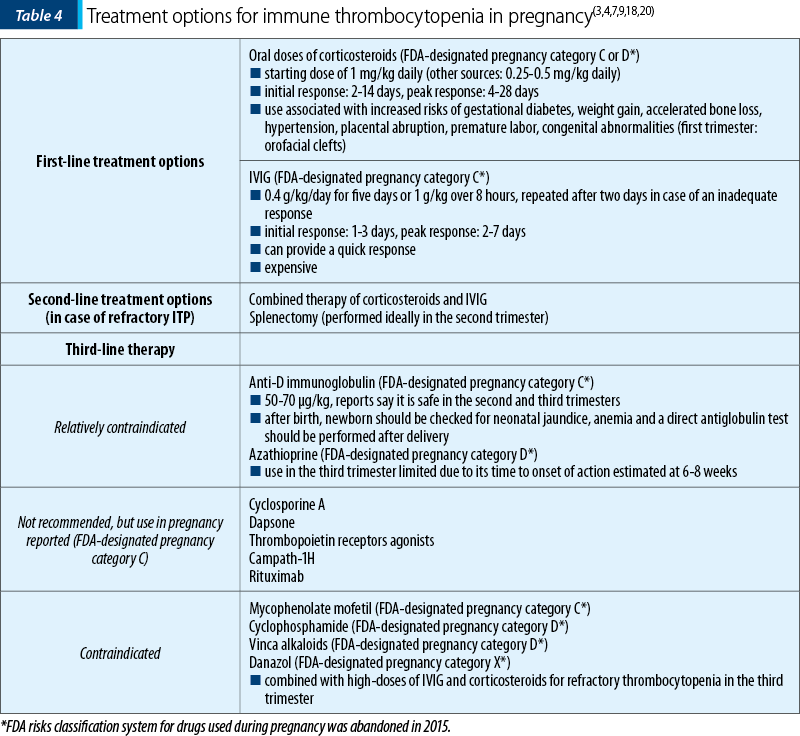
Along the years, there have been several opinions whether patients diagnosed with ITP should deliver by caesarean section or vaginally. Although a risk for neonates to develop intracerebral hemorrhage exists, there are no evidences across the literature that a caesarean section would decrease the chances of such an event. If we are talking about vaginal delivery, then the thrombocyte value should be at least 50x109/L, whereas if a caesarean section or an epidural anesthesia is necessary, then the values should be over 70 to 80x109/L(3,18.20).
Gilmore et al. report that there is a higher risk for women with ITP and a platelet level of less than 50x109/L to develop postpartum hemorrhage at the moment of delivery. Moreover, patients who needed a treatment for ITP had a higher estimated blood loss and the authors suggested the need for a higher platelet count at delivery in order to prevent such complications at birth(30).
Neonatal outcome
A platelet value of less than 20x109/L is correlated with severe neonatal thrombocytopenia (3-5% of ITP pregnancies), whereas a value of under 50x109/L is associated with moderate neonatal thrombocytopenia. Immunoglobulin (IVIG) is administered in severe cases with close monitoring of the neonates’ platelet values (daily for one week). If severe bleeding is associated, transfusions and glucorticoids are recommended(3,16,18).
The risk factors for a newborn to be suffering from ITP are: a mother who suffered a splenectomy, cases of ITP refractory to splenectomy, a platelet value of 100x109/L at the moment of delivery (which is inconsistent with other studies) or a sibling previously diagnosed with ITP(28,31,32). Kawaguchi et al. reported that platelet values recorded in previous newborns are a good marker for the values of the current sibling. Moreover, it was noted that platelets in newborns after vaginal delivery were more likely to decrease compared to the ones found in newborns delivered via caesarean section(33).
Two studies indicated that breast milk of mothers with active ITP contained antiplatelet specific antibodies. In both cases there was a reported increase for antibodies binding to fibrinogen receptor aIIbb3, demonstrating the fact that persistent neonatal thrombocytopenia can be associated with the transfer of antiplatelet antibodies from mothers diagnosed with ITP via their breast milk(34,35).
Kim et al. reported a case involving a 34-year-old female, pregnant at 33 weeks of gestation, diagnosed with ITP (platelet level of 2x109/L at the moment of referral) who had an ultrasound scan which showed an intracerebral hypoechoic lesion adjacent to the lateral ventricle, suggesting an intracerebral hemorrhage. A reevaluation was done at 37 weeks and two days, which highlighted newly instated hydrocephaly. The patient was administered IVIG, with a rise to 8x109/L platelets being noted. After having a platelet transfusion of 12 U, she underwent a caesarean delivery, giving birth to a newborn of 2540 kg, with an Apgar score of 7/8. The newborn had petechiae on the whole body skin, a cleft palate, and moderate to severe hydrocephaly on the ultrasound scan and confirmed by a CT exam. The platelet count was 1x109/L and there were no signs of antiplatelet antibodies and platelet-associated IgG. She received two days of IVIG therapy. The author believes that the intracranial hemorrhage and the breakdown products from the hemorrhage that led to the obstruction of the arachnoid granulations were the cause of hydrocephaly(36).
Conclusions
It is mandatory to accurately diagnose, manage and treat hematologic pathologies during pregnancy. Immune thrombocytopenia may be an apparently limited disorder, but it has often proved to be a challenge for obstetricians, hematologists and neonatologists, who need to work together and supervise these cases in order to prevent complications and ensure the best outcome available for both the mother and the newborn.
Conflict of interests: The authors declare no conflict of interests.
Bibliografie
- Williams Hematology, 9th edition, McGraw-Hill Education, 2016, 2010-2011.
- Jodkowska A, Martynowicz H, Kaczmarek-Wdowiak B, Mazur G, Thrombocytopenia in pregnancy – pathogenesis and diagnostic approach. Postepy Hig Med Dosw (Online). 2015 Nov 12; 69:1215-21.
- American Society of Hematology 2013 Clinical Practice Guide on Thrombocytopenia in pregnancy, available from: www.hematology.org/Clinicians/Guidelines-Quality/Quick-Ref/530.aspx
- Williams Obstetrics, 24th edition, McGraw-Hill Education, 2014, 1114-5.
- Ciobanu AM, Colibaba S, Cimpoca B, Peltecu G, Panaitescu AM. Thrombocytopenia in Pregnancy. Maedica. 2013; 11(5):55-60.
- Neunert C, Lim W, Crowther M, Cohen A, Solberg L Jr, Crowther MA, The American Society of Hematology. 2011 evidence based practice guideline for immune thrombocytopenia. Blood. 2011; 117(16):4190-207.
- Adams TM, Allaf MB, Vintzileos AM. Maternal Thrombocytopenia in Pregnancy: Diagnosis and Management. Clin Lab Med. 2013; 33(2):327–41.
- Shimanovsky A, Patel D, Wasser J. Refractory Immune Thrombocytopenic Purpura and Cytomegalovirus Infection: A Call for a Change in the Current Guidelines. Mediterr J Hematol Infect Dis. 2016; 8(1):e2016010.
- Lambert MP, Gernsheimer TB. Clinical updates in adult immune thrombocytopenia, Blood. 2017; 129(21):2829-35.
- Panaitescu AM, Stoia R, Ciobanu AM, Demetrian M, Peltecu G. Pregnancy shortly after an acute episode of severe acquired thrombotic thrombocytopenic purpura. Transfus Apher Sci. 2016; 55(3):308-10.
- Stavrou E, McCrae KR. Immune thrombocytopenia in pregnancy. Hematol Oncol Clin North Am. 2009; 23:1299–1316.
- Gabbe SG. Obstetrics – Normal and problem pregnancies, 7th edition, Elsevier, 2017; 48.
- Zhang X, Zhao Y, Li X, Han P, Jing F, Kong Z, Zhou H, Qiu J, Li L, Peng J, Hou M. Thrombopoietin: a potential diagnostic indicator of immune thrombocytopenia in pregnancy. Oncotarget. 2016; 16;7(7):7489-96.
- Wyszynski DF, Carman WJ, Cantor AB, Graham Jr. JM, Kunz LH, Slavotinek AM, Kirby RS, Seeger J. Pregnancy and Birth Outcomes among Women with Idiopathic Thrombocytopenic Purpura. J Pregnancy. 2016; 2016:8297407.
- Nicolescu A, Vlădăreanu AM, Voican I, Onisai M, Vlădăreanu R. Therapeutic options for immune thrombocytopenia (ITP) during pregnancy. Maedica. 2013; 8(2):182-8.
- Sun D, Shehata N, Ye XY, Gregorovich S, De France B, Arnold DM, Shah PS, Malinowski AK. Corticosteroids compared with intravenous immunoglobulin for the treatment of immune thrombocytopenia in pregnancy. Blood. 2016; 128(10):1329-35.
- Winkelhorst D, Murphy MF, Greinacher A, Shehata N, Bakchoul T, Massey E, Baker J, Lieberman L, Tanael S, Hume H, Arnold DM, Baidya S, Bertrand G, Bussel J, Kjaer M, Kaplan C, Kjeldsen-Kragh J, Oepkes D, Ryan G. Antenatal management in fetal and neonatal alloimmune thrombocytopenia: a systematic review. Blood. 2017; 16;129(11):1538-47.
- Cines DB, Levine L. Thrombocytopenia in pregnancy. Blood. 2017; 130(21):2271-227.
- Chakravarty, E, Murray E, Kelman A, Farmer P. Pregnancy outcomes following maternal exposure to rituximab. Blood. 2011; 117:1499–1506.
- Gernsheimer T, James AH, Stasi R. How I treat thrombocytopenia in pregnancy. Blood. 2013; 121:38-47.
- Kong Z, Qin P, Xiao S, Zhou H, Li H, Yang R, Liu X, Luo J, Li Z, Ji G, Cui Z, Bai Y, Wu Y, Shao L, Peng J, Ma J, Hou M. A novel recombinant human thrombopoietin therapy for the management of immune thrombocytopenia in pregnancy. Blood. 2017; 130(9):1097-103.
- Patil SA, Dottes-Katz SK, Metjian AD, James AH, Swamy GK. Use of a thrombopoietin mimetic for chronic immune thrombocytopenic purpura in pregnancy. Obstet Gynecol. 2013; 122(2 Pt 2):483-5.
- Suzuki N, Hiraga J, Hariyama Y, Takagi Y, Ohashi H, Kishigami Y, Oguchi H, Kagami Y. A low birth weight with no malformations delivered by a primary immune thrombocytopenia patient treated with eltrombopag. Int J Hematol. 2017; 108(1):109-11.
- Purushothaman J, Puthumana KJ, Kumar A, Innah SJ, Gilvaz S. A case of refractory immune thrombocytopenia in pregnancy managed with elthrombopag. Asian J Transfus Sci. 2016; 10(2):155-8.
- Rezk M, Masood A, Dawood R, Emara M, El-Sayed H. Improved pregnancy outcome following earlier splenectomy in women with immune thrombocytopenia: a 5-year observational study. J Matern Fetal Neonatal Med. 2018; 31(18):2436-40.
- Griffiths J, Sia W, Shapiro AM, Tataryn I, Turner AR. Laparoscopic splenectomy for the treatment of refractory immune thrombocytopenia in pregnancy. J Obstet Gynaecol Can. 2005; 27(8):771-4.
- Nelson-Piercy C. Handbook of Obstetric Medicine, 5th ed. CRC Press, London, 2015; 270-72.
- Piatek CI, El-Hemaidi I, Feinstein DI, Liebman HA, Akhtari M. Management of immune-mediated cytopenias in pregnancy. Autoimmun Rev. 2015; 14(9):806-11.
- Ono Y, Shiozaki A, Yoneda N, Yoneda S, Yoshino O, Saito S. Effectiveness of Helicobacter pylori eradication in pregnant women with idiopathic thrombocytopenic purpura. J Obstet Gynaecol Res. 2017; 43(7):1212-6.
- Gilmore KS, McLintock C. Maternal and fetal outcomes of primary immune thrombocytopenia during pregnancy: A retrospective study. Obstet Med. 2017; 11(1):12-6.
- Koyama S, Tomimatsu T, Kanagawa T, Kumasawa K, Tsutsui T, Kimura T. Reliable predictors of neonatal immune thrombocytopenia in pregnant women with idiopathic thrombocytopenic purpura. Am J Hematol. 2012; 87(1):15-21.
- Bayhan T, Tavil B, Korkmaz A, Ünal Ş, Hanalioğlu D, Yiğit Ş, Gümrük F, Çetin M, Yurdakök M. Neonates born to mothers with immune thrombocytopenic purpura: a single-center experience of 20 years. Blood Coagul Fibrinolysis. 2016; 27(1):19-23.
- Kawaguchi K, Matsubara K, Takafuta T, Shinzato I, Tanaka Y, Iwata A, Nigami H, Takeuchi Y, Fukaya T. Factors predictive of neonatal thrombocytopenia in pregnant women with immune thrombocytopenia. Int J Hematol. 2014; 99(5):570-6.
- Sharon N, Rosenberg N, Seligsohn U, Simmonds A, Shiff Y, Schechter Y, Hausner H. Breast milk from immune thrombocytopenic mothers can contain anti platelet antibodies that are associated with persistent thrombocytopenia in neonates. Blood. 2014; 124(21):4181.
- Hauschner H, Rosenberg N, Seligsohn U, Mendelsohn R, Simmonds A, Shiff Y, Schachter Y, Aviner S, Sharon N. Persistent neonatal thrombocytopenia can be caused by IgA antiplatelet antibodies in breast milk of immune thrombocytopenic mothers. Blood. 2015; 126(5):661-4.
- Kim MW, Choi HM. Fetal hydrocephalus in a pregnancy complicated by idiopathic thrombocytopenic purpura. J Ultrasound Med. 2006; 25(6):777-80.
Articole din ediţiile anterioare
Etiologia şi mecanismul patogenic al encefalopatiei hipoxic ischemice neonatale
Encefalopatia hipoxic-ischemică perinatală (EHIP) constituie cea mai importantă cauză de invaliditate şi mortalitate infantilă, prin consecinţele...
Caracteristicile neonatale în malformaţiile cardiace congenitale – experienţa noastră
Introducere. Malformaţiile cardiace congenitale (MCC) reprezintă cel mai frecvent defect congenital, apărând la 1% până la 5% din nou-născuţii ...
Donarea de organe neonatale – consideraţii etice şi criminalistice la graniţa dintre dramă şi speranţă
Context. Donarea de organe de la nou-născuţi pentru transplant este un eveniment relativ rar, care necesită cunoaşterea corespunzătoare a prob...
Provocările terapeutice ale sindromului de detresă respiratorie a nou-născutului – prezentare de caz
Insuficienţa respiratorie este frecvent observată la nou-născuţii cu asfixie perinatală severă. Aceasta reprezintă rezultatul afecţiunilor sub...