The phyllodes tumors are rarely found, accounting for up to 1% of cases. Phyllodes tumors tend to grow quickly, but they rarely spread outside the breast. In 2003, the World Health Organization classified the phyllodes tumors into three subtypes (benign, borderline and malignant), according to various clinicopathological characteristics, including the degree of stromal cell atypia and stromal overgrowth, tumor necrosis, the status of mitosis and the tumor margin. In this paper we present a case of giant phyllodes tumor of the breast in a young woman, with its clinicopathological particularities and the different treatment options.
Tumoră phyllodes gigantică
Giant phyllodes tumor of the breast
First published: 08 martie 2019
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/Gine.23.1.2019.2221
Abstract
Rezumat
Tumorile phyllodes sunt tumori rare, reprezentând doar 1% din toate cazurile de tumori ale sânului. Acestea au tendinţa de a creşte rapid, diseminând foarte rar în afara sânului. În 2003, Organizaţia Mondială a Sănătăţii a clasificat tumorile phyllodes în trei subtipuri (benigne, borderline şi maligne), în funcţie de diferitele caracteristici clinicopatologice, incluzând gradul de atipie celulară stromală, gradul de necroză tumorală, numărul mitozelor şi prezenţa invaziei marginilor de rezecţie. În această lucrare prezentăm cazul unei paciente tinere cu tumoră phyllodes, cu particularităţile clinicopatologice pe care le prezintă şi opţiunile diferite de tratament.
Introduction
Phyllodes tumors are rather rare entities, accounting for only 1% of all breast tumors(1). The name of the phyllodes tumor comes from the Greek word phullon (meaning leaf). Phyllodes tumors have been described for the first time by Cumin and Chilius in 1827, and in 1838 Muller called them “cystosarcome phylode”. Phyllodes tumors tend to grow quickly, but they rarely spread outside the breast. Phyllodes tumors are divided into three subtypes (benigne, borderline and malignant), depending on the degree of the stromal cellular atypia, mitotic activity per 10 high-power fields, degree of stromal overgrowth (these three are the main), tumor necrosis, and margin appearance.
Borderline tumors have the greatest tendency for local recurrence(2,3). All forms of phyllodes tumors have malignant potential and can behave like sarcomas, with metastases to various organs, commonly the lungs, bone and abdominal viscera(4). The majority of phyllodes tumors have been described as benign (35% to 64%), with the remainder divided between borderline and malignant subtypes. A five-year survival rate was observed in almost 100% of the patients with benign tumors – 98% with borderline, and about 88% with malignant tumors(5). The only treatment option for these tumors is the surgical removal.
Case report
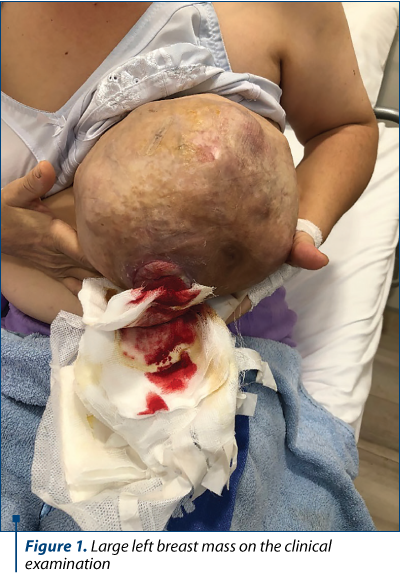
A 48-year-old female presented to our department in December 2018, with a lump in the left breast that had been gradually increasing for three months. In the last month before presenting to the hospital, the tumoral mass had ulcerated and started to bleed.
Laboratory and imaging findings
The hemoglobin level was 7 g/dl and all other biological investigations were within normal limits. The CEA and CA15-3 were in normal limits. A biopsy showed a phyllodes tumor. Computed tomography scanning for chest, abdomen and pelvis revealed: voluminous tissue formation (approximately 12/20 cm) with lobular contour that almost completely occupied the left breast, left axillary pathological adenopathies (between 1.2 and 1.7 cm), nodular imaging (approximately 1.5/2.5 cm), iodophyll in the inferior right breast. The patient perfomed total mastectomy without axillary dissection.
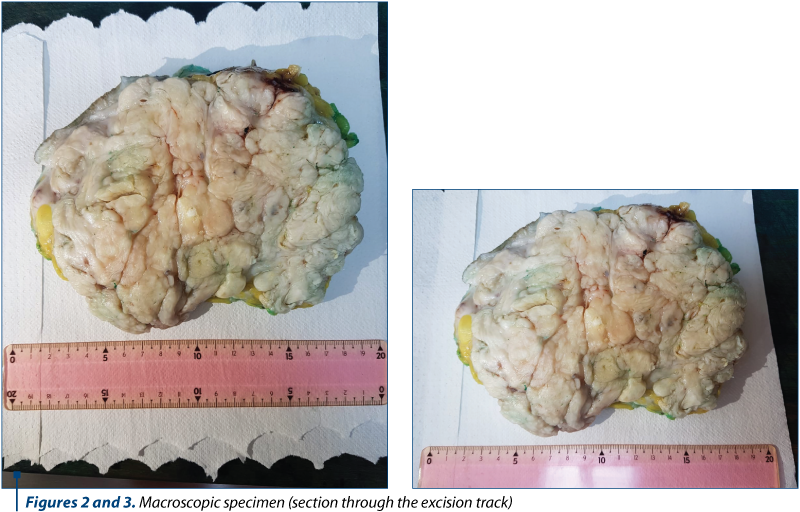
Macroscopic findings
A breast tissue measuring 20×18×13 cm in dimensions was resected (Figure 2 and Figure 3). Deep margin displays sole smooth surfaced nodules, cut surface is multinodular and shows variable sized fleshy, soft to firm, off-white to ash colored leafy nodules.
Microscopic findings
Mammary gland left tumor transforming histopathologically, showing a nodular tumor proliferation well delimited to the surrounding tissue with biphasic aspect, with intracanalicular growth pattern; the stromal component has moderate cellularity, presents limited areas with increased cellularity, moderate cyto-nuclear pleomorphism and relatively numerous mitosis (mitotic index = 9 mitoses/10 HPF); areas sclerohialinization and laxa, mixedomas; the epithelial component consists of variable ductile dots, some dilated epithelial and myoepithelial cell wall with areas of ductal epithelial hyperplasia; focal, dilated duct structures, some with cystic appearance, epithelial wallpaper with squamous metaplasia and keratotic; focal, in the periphery of the structures ductal proliferation of lobular mucosal epithelial structures, smooth contours and myoepithelial cell skin, relatively monoorphic epithelial cells with rare mitoses (adenose zones); cell columnar hyperplasia. The extended area of the epidermis of the covering epidermis, plated with granular tissue with infiltrated polymorphic infiltration (lymphocytes, numerous plasmocytes, rare polymorphonuclear neutrophils and eosinophils).
There are no areas of necrosis, sarcomatoid areas, marked cellular pleomorphism or infiltrative areas in the peritumoral tissue; the maximum number of mitoses was <10/10 HPF.
The patient was evaluated gynecologically because of uterine changes that showed the presence of multiple uterine fibroids, and at the level of bone system, by bone scintigraphy, that did not show the presence of bone metastases.
Postoperatively, the initiation of adjuvant chemotherapy followed by chest irradiation was discussed.
Discussion
Phyllodes tumors are rather rare entities, accounting for only 1% of all breast tumors(1). In 2003, the World Health Organization classified phyllodes tumors into three subtypes – benign, borderline and malignant –, according to various clinicopathological characteristics, including the degree of stromal cell atypia and stromal overgrowth, tumor necrosis, the status of mitosis and the tumor margin(7-9). However, the histologic classification does not always predict the outcome. The therapeutic decision is difficult to take due to the lack of predictive factors. Different studies have shown that infiltration of resection margins, elevated mitosis rates, and rapid stromal growth are important prognostic and predictive factors(10). Not all patients with positive margins develop recurrence. Malignant phyllodes tumors can cause metastases in 33% of cases despite radical mastectomy with negative margins(11). KI67 is an important prognostic marker. In the case of benign and borderline tumors when the KI67 index is grater than 10%, it is necessary to treat the patient and follow him up properly to avoid recurrence and malignant transformation(12).
Another tumor prognostic factor was considered the tumoral size, which was found to positively correlate with distant metastasis, and it was also observed that women with distant metastases tended to present with larger tumors(13).
Several authors found radiotherapy to reduce the local recurrence rates, but unlike with the usual invasive breast cancers, radiation did not improve the survival in malignant phyllodes(14). Local recurrences still developed in patients who received radiation, but few studies demonstrated that these women had large tumors and involved surgical margins, which rendered them at high risk of recurrence regardless. Tan and colleagues developed a nomogram to predict recurrence-free survival, based on stromal atypia, mitoses, stromal overgrowth and surgical margins(15). Even though the nomogram was not developed specifically for distant metastasis, it is likely still relevant, since recurrences are more often systemic, rather than isolated local recurrences, in malignant phyllodes.
Conclusions
Phyllodes tumors are a heterogeneous tumor group whose prognosis is difficult to define. The prognosis depends on the histological and biological characteristics of the tumors. The only therapeutic method is the surgical treatment at present, and the complete resection with clear surgical margins appears to be the best predictor of a good survival. In case of phyllodes benign and borderline tumors, post-resection follow-up remains the method of choice, although adjuvant chemotherapy and radiotherapy are discussed in borderline tumors. Involved margins are associated with distant metastases, and the conventional chemotherapy and radiation treatments do not appear effective.
Conflict of interests: The authors declare no conflict of interests.
Bibliografie
- Jang JH, Choi MY, Lee SK, Kim S, Kim J, Lee J, Jung SP, Choe JH, Kim JH, Kim JS, et al. Clinicopathologic risk factors for the local recurrence of phyllodes tumors of the breast. Ann Surg Oncol. 2012; 19:2612–7.
- Gradishar WJ, Anderson BO, Balassanian R, Blair SL, Burstein HJ, Cyr A, Elias AD, Farrar WB, Forero A, Giordano SH, et al. Breast Cancer, Version 1. J Natl Compr Canc Netw. 2016; 13:1475–85.
- Karczmarek-Borowska B, Bukala A, Syrek-Kaplita K, Ksiazek M, Filipowska J, Gradalska-Lampart M. A rare case of breast malignant phyllodes tumor with metastases to the kidney: Case report. Medicine (Baltimore). 2015; 94:e1312.
- Mituś J, Reinfuss M, Mituś JW, Jakubowicz J, Blecharz P, Wysocki WM, Skotnicki P. Malignant phyllodes tumor of the breast: Treatment and prognosis. Breast J. 2014; 20:639–44.
- Reinfuss M, Mituś J, Smolak K, Stelmach A. Malignant phyllodes tumours of the breast. A clinical and pathological analysis of 55 cases. Eur J Cancer. 1993; 29A:1252–6.
- Sedgwick EL, Ebuoma L, Hamame A, Phalak K, Ruiz-Flores L, Ortiz-Perez T, Sepulveda KA. BI-RADS update for breast cancer caregivers. Breast Cancer Res Treat. 2015; 150:243–54.
- Feakins RM, Mulcahy HE, Nickols CD, Wells CA. p53 expression in phyllodes tumours is associated with histological features of malignancy but does not predict outcome. Histopathology. 1999; 35:162–9.
- Kleer CG, Giordano TJ, Braun T, Oberman HA. Pathologic, immunohistochemical, and molecular features of benign and malignant phyllodes tumors of the breast. Mod Pathol. 2001; 14:185–90.
- Kuenen-Boumeester V, Henzen-Logmans SC, Timmermans MM, et al. Altered expression of p53 and its regulated proteins in phyllodes tumours of the breast. J Pathol. 1999; 189:169–75.
- Millar EK, Beretov J, Marr P, et al. Malignant phyllodes tumours of the breast display increased stromal p53 protein expression. Histopathology. 1999; 34:491–6.
- Hawkins RE, Schofield JB, Fisher C, Wiltshaw E, McKinna JA. The clinical and histologic criteria that predict metastases from cystosarcoma phyllodes. Cancer. 1992; 69(1):141-7.
- August DA, Kearney T. Cystosarcoma phyllodes: mastectomy, lumpectomy, or lumpectomy plus irradiation. Surg Oncol. 2000; 9(2):49-52.
- Gnerlich JL, Williams RT, Yao K, Jaskowiak N, Kulkarni SA. Utilization of radiotherapy for malignant phyllodes tumors: analysis of the National Cancer Data Base. Ann Surg Oncol. 2014; 21(4):1222-30.
- Mitus J, Reinfuss M, Mitus JW, Jakubowicz J, Blecharz P, Wysocki WM, et al. Malignant phyllodes tumor of the breast: treatment and prognosis. Breast J. 2014; 20(6):639-44.
- Tan PH, Thike AA, Tan WJ, Thu MM, Busmanis I, Li H, et al. Predicting clinical behaviour of breast phyllodes tumours: a nomogram based on histological criteria and surgical margins. J Clin Pathol. 2012; 65(1):69-7.
Articole din ediţiile anterioare
Fetal and neonatal diagnosis and management in a case of a neonate with congenital heart disease – case report
Boala cardiacă congenitală (BCC) este cea mai frecventă formă de anomalie congenitală. La făt, canalul arterial (CA) deviază sângele neoxigenat din...
Studiu clinico-patologic al tumorilor ovariene - experienţa de un an într-un centru medical
Scopul acestui studiu este analiza retrospectivă a spectrului de tumori ovariene, din punct de vedere statistic, epidemiologic şi al caracteristi...
Relaţia dintre infiltratul limfocitar tumoral şi progresia în cancerul de sân triplu negativ
Cancerul de sân este o boală foarte heterogenă, atât la nivel molecular, cât şi la nivel histologic. Iniţial au fost descrise cinci subtipuri intri...
Impactul fetal şi neonatal al metodelor de diagnostic şi al chimioterapiei în sarcină la gravidele cu afecţiuni oncologice
Afecţiunile oncologice din timpul sarcinii sunt diagnosticate mult mai frecvent în ultimii ani datorită metodelor de diagnostic avansate. Odată c...