Ovarian cancer is a public health problem that affects women of reproductive age and is a major cause of morbidity and mortality, accounting for 85% of all malignant ovarian neoplasms. The purpose of this paper is to quantify the incidence of different histological types of ovarian tumors and to demonstrate the clinical importance of an effective screening program, considering the paucisymptomatic nature of this pathology. This study represents a retrospective evaluation of 249 consecutive cases of epithelial ovarian tumors diagnosed and treated in the “Prof. Dr. Panait Sîrbu” Clinical Hospital of Obstetrics and Gynecology, Bucharest, Romania, between 2014 and 2016, out of which we extracted the data related only to carcinomas (23 cases). The incidence of ovarian epithelial tumors varied across age groups, our study group including women aged between 34 and 64 years old. Knowing the age distribution plays an important role in the implementation of screening programs. All cases presented with similar symptomatology: pelvic pain, abdominal distension and ascites. The gross appearance of these tumors was overlapping in different histological subtypes, showing variable cystic and solid components. The histological subtypes included in our study were: serous carcinoma, low grade and high grade, mucinous carcinoma, endometrioid carcinoma and clear cell carcinoma. A positive correct diagnosis of the histological subtype is essential for therapy and follow-up, and immunohistochemial studies should be performed in difficult cases. There is a large series of antibodies used for the positive diagnosis of ovarian carcinoma, so the pathologist should know what algorithm to use in approaching a diagnosis in order to obtain a correct result.
Utilitatea imunohistochimiei în diagnosticul carcinomului ovarian
The utility of immunohistochemistry in the diagnosis of ovarian carcinoma
First published: 18 decembrie 2017
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/Gine.18.4.2017.1328
Abstract
Rezumat
Cancerul ovarian este o problemă de sănătate publică ce afectează femeile de vârstă reproductivă, fiind o cauză majoră a morbidității și mortalității și reprezentând 85% din toate neoplasmele ovariene maligne. Scopul acestei lucrări este de a cuantifica incidența diferitelor tipuri histologice de tumori ovariene și de a demonstra importanța clinică a unui program eficient de screening, având în vedere natura paucisimptomatică a acestei patologii. Acest studiu reprezintă o evaluare retrospectivă a 249 de cazuri consecutive de tumori ovariene epiteliale diagnosticate și tratate în Spitalul Clinic de Obstetrică-Ginecologie „Prof. Dr. Panait Sîrbu” în perioada 2014-2016, din care am extras datele referitoare numai la carcinoame (23 de cazuri). Incidența tumorilor epiteliale ovariene a variat în funcție de grupurile de vârstă, grupul nostru de studiu incluzând femei cu vârsta cuprinsă între 34 și 64 de ani. Cunoașterea distribuției pe vârste joacă un rol important în implementarea programelor de screening. Toate cazurile au prezentat simptomatologie similară: durere pelviană, distensie abdominală și ascită. Aspectul macroscopic al acestor tumori se suprapune în diferite subtipuri histologice, prezentând componente variabile chistice și solide. Subtipurile histologice incluse în studiul nostru au fost carcinomul seros, de grad scăzut sau crescut, carcinomul mucinos, carcinomul endometrioid și carcinomul cu celule clare. Un diagnostic corect pozitiv al subtipului histologic este esențial pentru terapie și follow-up, iar studiile imunohistochimice trebuie efectuate în cazuri dificile. Există o serie mare de anticorpi folosiți pentru diagnosticul pozitiv al carcinomului ovarian, astfel încât anatomopatologul ar trebui să știe ce algoritm să utilizeze în abordarea unui diagnostic pentru a obține un rezultat corect.
Introduction
Ovarian cancer is a public health problem that affects women of reproductive age and is a major cause of morbidity and mortality. Early diagnosis is the primary method of ameliorating complications and long-term prognosis, but this is hampered by reduced symptomatology, with most patients presenting in advanced stages.
Ovarian carcinoma accounts for 85% of all malignant ovarian neoplasms(1), being the seventh most common cause of cancer mortality in the global female population(2). From 2003 to 2012, the incidence rate and the mortality rate decreased by 0.9% and 2%, respectively, each year(3).
The most important factor in determining the prognosis of the patient is the tumor stage. Although the survival at 5 years for stage I ovarian cancer is 90%, approximately 75% of patients are in stage III/IV, with a survival rate of 30%(2). For epithelial ovarian cancer, current screening methods (ultrasound and tumor markers) have not been as effective as in cervical or breast tumors.
Ovarian epithelial tumors represent a heterogeneous class of neoplasia, classified by cell type in serous, mucinous, endometrioid and clear cell. Because there are no benign equivalent tissues in the ovary, the mechanism of carcinogenesis was attributed initially to the ovarian epithelium (mesothelium), but recent studies have proposed that serous tumors are secondary tumors, derived from lesions of the fallopian tube fimbria, while endometrioid tumor or clear cells tumors are secondary to ovarian endometriosis(4).
Ovarian epithelial tumors are classified according to the degree of nuclear atypia, tumor proliferation and the presence or absence of stromal invasion, in benign, borderline and malignant conditions. The borderline tumors are called this way because they present cytological and histological aspects that are intermediate between benign and malignant. Ovarian carcinomas are classified according to histological and immunohistochemical carcinomas in five main categories: high grade serous carcinoma (70%), endometrioid carcinoma (10%), clear cell carcinoma (10%), mucinous carcinoma (3%), low grade serous carcinoma (5%)(5).
Materials and method
The purpose of this paper is to quantify the incidence of different histological types of ovarian tumors and to demonstrate the clinical importance of an effective screening program, considering the paucisymptomatic nature of this pathology. This study represents a retrospective evaluation of 249 consecutive cases of epithelial ovarian tumors diagnosed and treated in the “Prof. Dr. Panait Sîrbu” Clinical Hospital of Obstetrics and Gynecology between 2014 and 2016, out of which we extracted the data related only to carcinomas (23 cases). Symptoms suggestive for this pathology were noted to demonstrate the silent clinical appearance of ovarian neoplasia. Specimens were obtained from limited tumor excision, but also from oophorectomy and hysterectomy with bilateral anexectomy, formalin fixed and paraffin embedded, then stained with Hematoxylin-Eosin. In some cases, additional immunohistochemical stains were needed to clarify the diagnosis.
Results
This study included data from a batch of 23 ovarian carcinomas, selected from 249 ovarian pathology patients. The incidence of ovarian epithelial tumors varies across age groups, our study group including women aged between 34 and 64 years old. Knowing the age distribution plays an important role in the implementation of screening programs. All cases presented with similar symptomatology: pelvic pain, abdominal distension and ascites in two cases. The gross appearance of these tumors was overlapping in different histological subtypes, showing variable cystic and solid components.
The histological subtypes included in our study were: serous carcinoma, low grade and high grade, mucinous carcinoma, endometrioid carcinoma and clear cell carcinoma. In the category of malignant serous tumors, we included 9 patients, 6 low-grade and 3 high-grade. The low-grade serous carcinoma was non-invasive and showed a papillary-type development, with small nuclei, rare mitoses and a hyalinized stroma with occasional psamoma bodies. Immunohistochemical assays showed positivity to CK7 and ER. P53 showed patchy immunostaining and Ki67 was on average 15% positive (Figure 1).

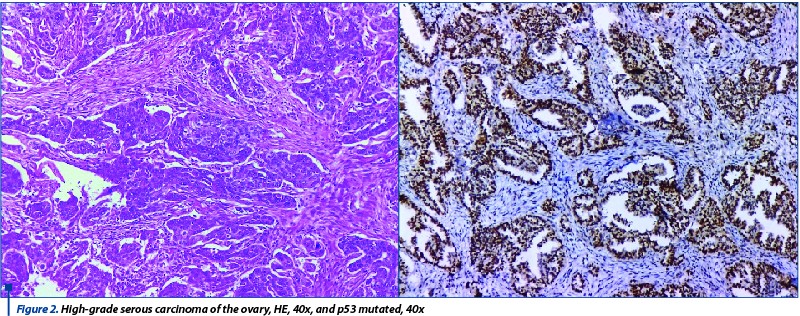
The cases with high-grade serous carcinoma showed areas of invasive cells with marked atypia and mitoses, with areas of necrosis and hemorrhage. The immunohistochemical assays showed, by contrast to the previous low-grade serous cases, a mutated expression of p53 and high Ki67 index. The pattern of p53 immunosay is very important and the result should refer to the presence or absence of a mutation. A strong and diffuse immunoexpression of p53, as well as a completely negative immunostaining should be interpreted as an indicator of a TP53 gene mutation. In our cases, all high grade showed mutated status of TP53 gene. In two cases, the immunostaining showed strong reaction in more than 75% of tumoral cells and in the third case there was no reaction. Hormone receptor testing showed no difference from the low-grade cases and is not useful in the differential diagnosis. Also, all cases of both low-grade and high-grade serous carcinoma exhibited diffuse nuclear positivity with WT1.
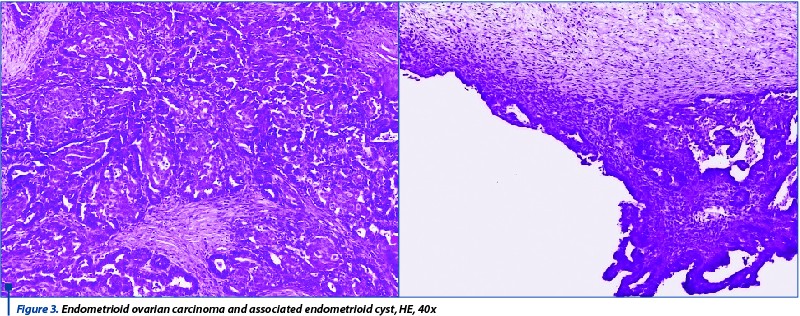
The cases of carcinomas with glandular architecture, atypical cells and foci of squamous metaplasia were classified as endometroid carcinomas due to their resemblance to the endometrium (5 cases). The immunohistochemical stains used for these cases were CK7, P53, Ki67, ER, PGR and CD10. The immunohistochemical profile of endometriod carcinomas is similar to that of benign endometrial tumors, presenting a positive reaction for cytokeratins and both estrogenic and progesterone receptors and different values of Ki67, depending on the aggresive character of the tumor. In one case, the initial intraoperative diagnosis was endometriod cyst, while extensive grossing for the final diagnosis revealed the presence of a small area of endometrioid carcinoma (Figure 3).
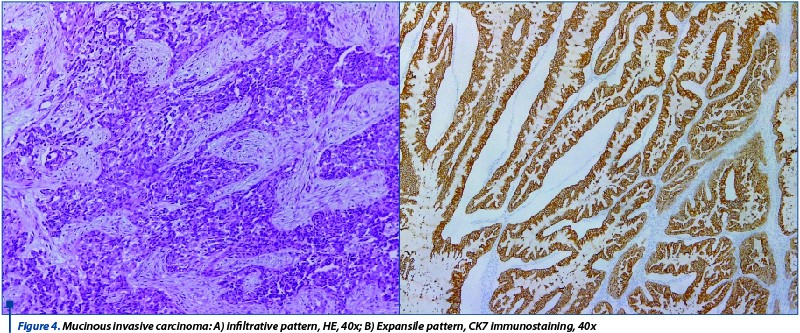
A third histopathological category of ovarian epithelial tumors were the mucinous tumors, which represented 2.81% of the total number of cases and 31.8% of the carcinomas. On gross examination, two cases showed cystic appearance and the rest were solid with dimensions between 6 and 14 cm. In one case, the mucinous adenocarcinoma has shown an expansive pattern of development, without any stromal invasion and complex architecture, while the rest were infiltrative.
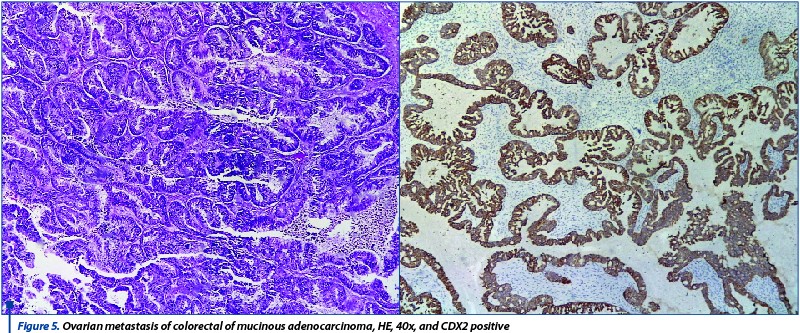
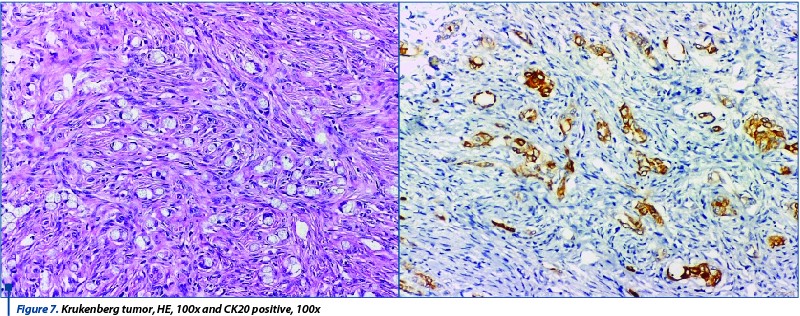
Of the 7 cases, one was present at the level of both ovaries, thus being classified as FIGO IIIA. The 61-year-old patient who was diagnosed with this tumor had epiploic metastasis at admission. Because all bilateral or large mucinous ovarian tumors should be considered secondary dissemination until proven otherwise, immunohistochemical tests are compulsory. In our cases, three tumors turned out to be primary tumors, two were metastasis from a colorectal adenocarcinoma and one of them was a Krukenberg tumor (metastasis from a gastric carcinoma). The combination used for the differential diagnosis of mucinous tumors was CK7/CK20 and CEA, CDX2. Secondary tumors did not show positivity for CK7, but were positive for CEA and CDX2, while the primary ones had mirroring reactions (Figure 5 and Figure 6).
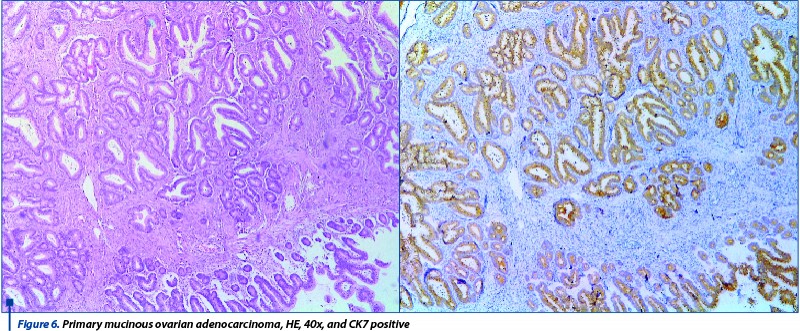
The Krukenberg tumor showed a specific pattern, with signet ring infiltrating tumoral cells, Ck20, CDx2 and CEA positive, but the diagnosis cannot be relied solely on histological and immunohistochemical pattern and it had to be confirmed by the clinical context.
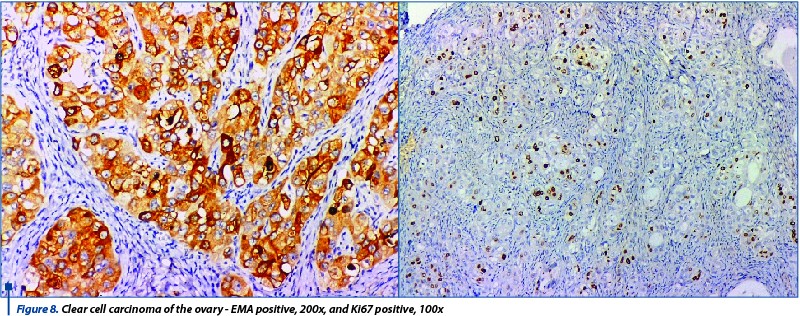
Clear cell carcinomas are rare tumors and we only found two cases that showed very different histological pattern: one had hobnail cell that protruded into the lumina, while the other showed clear cytoplasm. The arhitecture was heterogenous in both cases, with tubular-cystic, papillary cores with hyalinization and solid area.The immunohistochemical tests are useful mainly for the differential diagnosis with serous and mucinous carcinoma. The tumors showed positivity for EMA, Napsin and were negative for ER and WT1. Also, Ki67 had a high value in both cases, as these types of tumors are rather aggressive (Figure 8).
Discussions
Malignant ovarian neoplasms are the seventh most common form of cancer diagnosed in the female population. Out of these, the epithelial subtype represents the majority of cases and approximately 30% of all genital carcinomas(6).
The 5-year survival of patients diagnosed with this pathology is 32%, due to signs and symptoms reduced in intensity and number. Thus, about 70% of ovarian epithelial cancers are found in advanced stages(7).
Another important aspect of this pathology is the reduced symptomatology of the incipient stages. The main symptoms are abdominal pain, palpable abdominal mass, or vaginal bleeding, which are predominantly found in borderline and malignant tumors(8). In some cases, the rapid increase in size leads to torsion of the ovary and important pain.
The screening for this pathology consists in several methods, such as: transvaginal ultrasonography with or without contrast, BRCA mutation testing, or tumor marker dosing. Taking into account the prevalence of BRCA mutation in ovarian tumors (women with this mutation have a risk of developing ovarian cancer by the age of 70 between 30% and 70%), the implementation of this analysis has a favorable cost-effectiveness ratio(9).
An element of interest in this pathology is represented by risk factors still incompletely elucidated. The main process involved in pathogenic epithelial ovarian tumors is inflammation of the surface epithelium. The inflammatory changes occurring at the time of ovulation and hyaline body formation cause alterations of epithelial cell DNA(10).
A positive correct diagnosis of the histological subtype is essential for therapy and follow-up. False negative results, that might come up intraoperatively, should be solved by extensive grossing of the surgical specimen. In our study, only one case showed false negative result. Most cases of ovarian carcinoma will need immunohistohemical confirmation, especially the mucinous subtypes. Ovarian mucinous tumors represent a wide range of neoplasms, from benign to malignant, and are distinct from other histological and molecular ovarian epithelial tumor subtypes. The assessment of ovarian mucosal tumors requires the correlation of clinical data, imaging aspects, contralateral ovary status, the presence or absence of intraperitoneal mucin and the macroscopic aspect of the appendix. Although in most cases a positive diagnosis of a benign lesion does not pose diagnostic problems, the differential diagnosis between a primary ovarian tumor and a metastasis is difficult to perform intraoperatively or in the classic histological examination. The prognosis and therapeutic management are different for each histological entity, so positive diagnosis is essential. Malignant tumors of the ovary are characterized by positivity to CK7 and P53, and negative for CK20(11).
Conclusions
In conclusion, epithelial ovarian neoplasia affects an increased number of women, mainly of reproductive age. The most affected age categories are the third and fourth decades, with a maximum incidence in the fourth decade. Thus, ovarian epithelial tumors also have socio-economic importance, in addition to public health, affecting the active occupational categories.
Reducing the morbidity and mortality of this pathology through effective screening programs becomes a priority of the health system. As therapy has progressed towards a more targeted approach, there have been changes and advances in the understanding of ovarian carcinoma.
Also, the ovarian tumors of the epithelial type present a large histological variety, each exhibiting several developmental patterns and requiring additional immunohistochemical tests.
Bibliografie
2. Crum CP, Nucci MR, Lee KR (eds). Diagnostic Gynecologic and Obstetric Pathology. 2nd Ed, NY, Elsevier, 2011.
3. PDQ Screening and Prevention Editorial Board. Ovarian, Fallopian Tube and Primary Peritoneal Cancer Screening (PDQ®): Health Professional Version. 2017 Feb 3. In: PDQ Cancer Information Summaries. Bethesda (MD): National Cancer Institute (US); 2002. Available from: https://www.ncbi.nlm.nih.gov/books/NBK65898.
4. Kurman RJ, Shih IM. The origin and pathogenesis of epithelial ovarian cancer: a proposed unifying theory. Am J Surg Pathol 2010; 34:433-43.
5. Mutter G, Prat J. Pathology of the Female Reproductive Tract, 3rd Edition, Churchill Livingstone, 2014.
6. Reid BM, Permuth JB, Sellers TA. Epidemiology of ovarian cancer: a review. Cancer Biol Med, 2017; 14(1):9-32.
7. Goff BA, Mandel LS, Drescher CW, Urban N, Gough S, Schurman KM, et al. Development of an ovarian cancer symptom index: possibilities for earlier detection. Cancer, 2007; 109(2):221-7.
8. Lurie G, Thompson PJ, McDuffie KE, Carney ME, Goodman MT. Prediagnostic symptoms of ovarian carcinoma: A case-control study. Gynecologic oncology, 2009; 114(2):231-6.
9. Eccleston A, Bentley A, Dyer M, et al. A Cost-Effectiveness Evaluation of Germline BRCA1 and BRCA2 Testing in UK Women with Ovarian Cancer. Value in Health, 2017; 20(4):567-76.
10. Gates MA, Rosner BA, Hecht JL, Tworoger SS. Risk factors for epithelial ovarian cancer by histologic subtype. Am J Epidemiol, 2010; 171(1):45-53.
11. Kriplani D, Patel MM. Immunohistochemistry: A diagnostic aid in differentiating primary epithelial ovarian tumors and tumors metastatic to the ovary. South Asian J Cancer. 2013; 2(4):254-8.
Articole din ediţiile anterioare
Cancere genitale sincrone
Studiul de faţă prezintă o evaluare histopatologică a cancerelor sincrone primare ginecologice. Material şi metodă. Au fost identificate 17 pacient...
Carcinom endometrial mixt endometrioid şi neuroendocrin
Neuroendocrine tumors are rarely found in the genital tract, making up approximately 2% of all gynecological tumors(1). In most cases, they appear ...
Complicaţiile perinatale şi neonatale la pacientele cu răspuns ovarian scăzut în sarcinile obţinute prin proceduri de reproducere umană asistată
In vitro fertilization (IVF) technologies with a controlled ovarian hyperstimulation approach have classified patients into three different groups ...
Tumorile cordoanelor sexuale stromale ale ovarului: tumoarea de granuloasă. Prezentare de caz şi revederea literaturii
Tumorile cordoanelor sexuale stromale ale ovarului reprezintă o patologie rară, constituind mai puţin de 2% din totalul cancerelor primare ov...