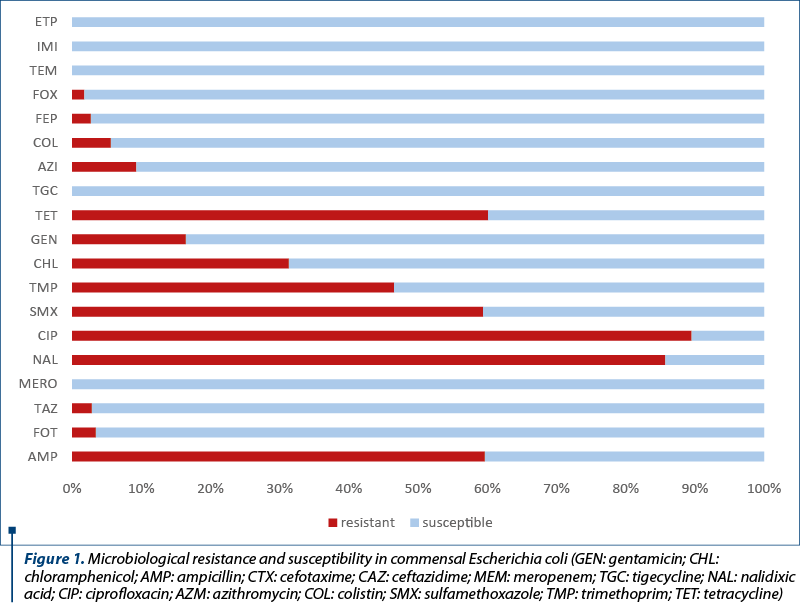
Antimicrobial resistance is defined as a top public health challenge in the 21st century. The excessive use of antimicrobials in human and veterinary medicine has accelerated the emergence and the spread of resistant microorganisms. In 2016, monitoring of antimicrobial resistance in commensal Escherichia coli (N=840) isolated from cecal samples of slaughtered broilers in Romania has been run according Decision 652EC/2013. Using the broth microdilution method and based on minimal inhibitory concentration, the microbiological resistance to each of the 19 tested antimicrobials was found, reaching the highest values for ciprofloxacin (89.52%), nalidixic acid (85.71%), tetracycline (60.12%), ampicillin (59.64%) and sulfamethoxazole (59.40%), whereas the lowest values were found for colistin (5.6%) and cephalosporins (1.79-3.45%). Confirmation tests for extended-spectrum β-lactamase producing E. coli were mandatory on all strains resistant to cefotaxime and/or ceftazidime. The high prevalence of antibiotic-resistant E. coli may be a consequence of antimicrobial consumption, usage preferences or management practices in food-producing animals. Thus, commensal E. coli could be a reservoir of resistance genes, with a major impact in their spread and possible threats for human and animal health.
Rezistenţa la antimicrobiene pentru Escherichia coli comensală izolată de la pui crescuţi pentru carne
Antimicrobial resistance in commensal Escherichia coli isolated from slaughtered broilers in Romania
First published: 31 octombrie 2018
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/Inf.55.3.2018.2037
Abstract
Rezumat
În secolul XXI, rezistenţa antimicrobiană este definită ca o provocare de top pentru sănătatea publică. Utilizarea excesivă a substanţelor antimicrobiene în medicina umană şi veterinară a accelerat apariţia şi răspândirea microorganismelor rezistente. În 2016, în conformitate cu Decizia 652CE/2013, s-a realizat monitorizarea rezistenţei la antimicrobiene a tulpinilor comensale de Escherichia coli izolate din probe de conţinut cecal prelevate de la puii de carne abatorizaţi în România. Utilizând metoda microdiluţiei în bulion, pe baza concentraţiei minime inhibitorii, rezistenţa la antimicrobiene pentru fiecare dintre cele 19 probe testate a atins cele mai mari valori pentru ciprofloxacină (89,52%), acid nalidixic (85,71%), tetraciclină (60,12%) şi ampicilină (59,64%) şi mai scăzute pentru colistin (5,6%) şi cefalosporine (1,79-3,45%). Pentru toate tulpinile rezistente la cefotaxim şi/sau la ceftazidim, au fost obligatorii testele de confirmare pentru E. coli producătoare de β-lactamaze cu spectru extins. Prevalenţa ridicată a tulpinilor comensale de E. coli rezistente la antimicrobiene ar putea fi consecinţa consumului crescut, utilizării preferenţiale sau a practicilor de management din unităţile de creştere a animalelor destinate consumului uman. Astfel, E. coli ar putea fi un rezervor al genelor de rezistenţă, cu un impact major în răspândirea lor, precum şi o posibilă ameninţare la adresa sănătăţii umane şi animale.
Introduction
The main undesirable side effect of antimicrobial use in both humans and animals is antimicrobial resistance (AMR)(1). Resistance is a natural and ancient phenomenon and results from the continuous positive selection of resistant bacterial clones, whether these are pathogenic, commensal or even environmental bacteria. AMR modifies the population structure of microbial communities, leading to accelerated evolutionary trends with consequences for human and animal health(2,3). In this context, the enhanced monitoring of AMR in bacteria from food-producing animals set out in European Commission Implementing Decision 2013/652/EU was successfully implemented in Romania during 2016(3,4).
Materials and method
The cecal samples (N=840) were randomly collected during 2016 from broilers, slaughtered in 29 slaughterhouses from 19 counties. The samples were processed in the labs no more than 48 hours after collection. The cecal content was directly streaked onto McConkey agar. Colonies showing typical E. coli morphology were confirmed biochemically using triple-sugar-iron agar, mobility-indol-urea medium, O-Nitrophenyl-b-D-galactopyranoside test. A single isolate per each sample was selected for antibiotic susceptibility testing (AST).
Minimum Inhibitory Concentrations (MICs) (mg/L) were determined using Sensititre plates (Trek Diagnostic System), respectively EUVSEC panel – ampicillin (AMP), cefotaxime (FOT), ceftazidime (TAZ), gentamicin (GEN), tetracycline (TET), sulfamethoxazole (SMX), trimethoprim (TMP), ciprofloxacin (CIP), nalidixic acid (NAL), chloramphenicol (CHL), azithromycin (AZI), tigecycline (TGC), colistin (COL), and meropenem (MERO). The E. coli strains which after testing with this panel of antimicrobials were resistant to cefotaxime or ceftazidime or meropenem have been tested with a second panel of antimicrobial, EUVSEC2 panel. This panel includes cefoxitin (FOX), cefepime (FEP) and clavulanate synergy test in combination with cefotaxime (FOT) and ceftazidime (TAZ) for the detection of extended-spectrum b-lactamase (ESBL) and AmpC enzymes production. In addition, the second panel also contains imipenem (IMI), meropenem (MERO) and ertapenem (ETP) to phenotypically verify the presumptive carbapenemase-producers(4). The quality control strain used was E. coli ATCC 25922. The obtained MICs values were interpreted according epidemiological cut-off values of European Committee on Antimicrobial Susceptibility Testing (http://eucast.org), criteria established by Decision 2013/652/EU and also by European Food Safety Authority (EFSA) criteria for presumptive phenotype(4,5).
Results and discussion
The prevalence of AMR and MIC distribution of commensal E. coli is shown in Table 1. Based on MIC obtained for 19 antimicrobials, the occurrence of resistance for the 840 strains isolated from cecal samples was extremely high to CIP (89.52%), NAL (85.71%), TET (60.12%), AMP (59.64%), SMX (59.40%), high to TMP (46.55%), CHL (31.31%), moderate to GEN (16.43%) and low to AZI (9.29%), COL (5.60%), and cephalosporins (FOT – 3.45%; TAZ – 2.86%; FEP 2.74%; FOX – 1.79%) (Figure 1).
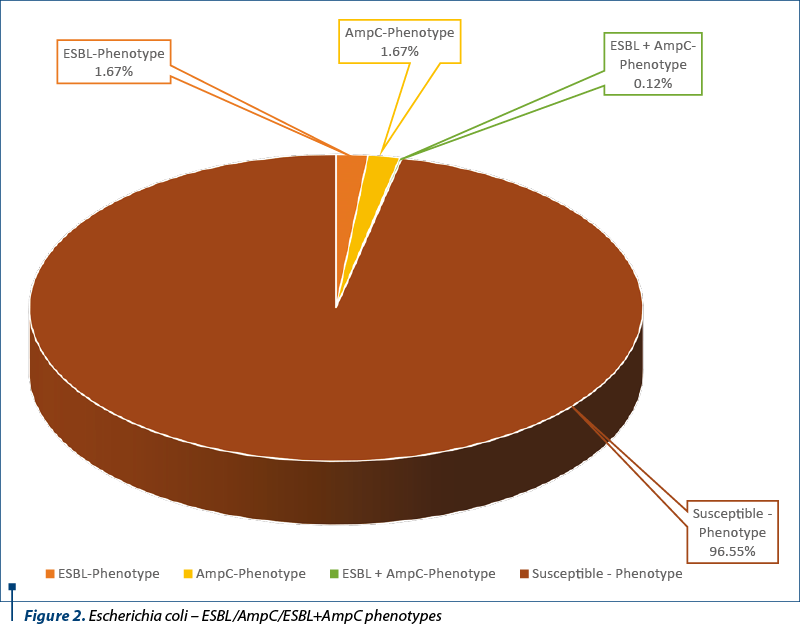
The commensal E. coli strains resistant to cefotaxime or/and ceftazidime were tested for clavulanate synergy test and FOX for the detection of ESBL and/or AmpC production. The global prevalence of commensal ESBL/AmpC producing E. coli strains isolated from the broilers ceca on MacConkey agar was 3.45%, respectively 29 strains from 840 samples. Presumptive ESBL (14/840) and AmpC-positive (14/840) strains were more frequent (1.67%) compared with presumptive ESBL+AmpC-type cephalosporinase producers (1/840 – 0.12%) (Figure 2).
The occurrence of isolates exhibiting combined resistance to cefotaxime and ciprofloxacin, that are critically important antimicrobials for public health, has high relevance in monitoring(2). Thus, 2.86% (24/840) of the isolates from broilers were resistant to both cefotaxime and ciprofloxacin.
Conclusions
The resistance to quinolones (nalidixic acid and ciprofloxacin) was the most common resistance trait in E. coli isolates from broilers in 2016. Poultry could be also a reservoir of third-generation cephalosporin-resistant bacteria, but specific selective screening is needed to increase the rate of isolation. The prevalence of colistin resistance was at a low level, but taking into account that could be probably a cause of existence of mobile genetic elements in commensal bacterial population further molecular investigations are necessary.
Acknowledgments. The work was funded by the National Sanitary Veterinary and Food Safety Authority of Romania and European Commission, on the basis of Grant Decision SI2.728669/12.04.2016 (Ares(2016)1724197). A part of the laboratory investigations was performed at the Sanitary Veterinary and Food Safety Directorates of Alba, Brăila, Giurgiu, Mureş, Prahova and Satu Mare counties. The authors are grateful to Camelia Sârbu, Sorinel Voicu, Mihail Cartojan, Camelia Nuţiu, Alina Văcarel, and Claudiu Andor for their contribution to this paper.
Bibliografie
- Bengtsson B, Greko C. Antibiotic resistance – consequences for animal health, welfare, and food production. Ups J Med Sci. 2014; 119(2):96-102.
- Davies J, Davies D. Origins and Evolution of Antibiotic Resistance. Microbiology and Molecular Biology Reviews. 2010; 74(3):417-433.
- *** EFSA (European Food Safety Authority) and ECDC (European Centre for Disease Prevention and Control), 2018. The European Union summary report on antimicrobial resistance in zoonotic and indicator bacteria from humans, animals and food in 2016. EFSA Journal. 2018; 16(2):5182.
- *** Commission Implementing Decision 2013/652/EU of 12 November 2013 on the monitoring and reporting of antimicrobial resistance in zoonotic and commensal bacteria. OJ L 303, 14.11.2013, pag. 26–39.
- *** EFSA (European Food Safety Authority) and ECDC (European Centre for Disease Prevention and Control), 2017. The European Union summary report on antimicrobial resistance in zoonotic and indicator bacteria from humans, animals and food in 2015. EFSA Journal. 2017; 15(2):4694, pag 43.
Articole din ediţiile anterioare
Sepsis cu Escherichia coli dobândit în spital – analiză de caz
Pacienţii cu hemopatii maligne au un risc crescut de a contracta infecţii asociate asistenţei medicale, care le pot fi fatale. Acesta este în speci...
Infecţii intestinale ale epocii moderne: actualităţi privind formele severe de diaree provocate de Clostridium difficile și Escherichia coli enterohemoragic
În cele trei articole precedente pe tema infecțiilor intestinale ale epocii moderne, s-au discutat aspecte legate de diareea călătorului, detaliind...