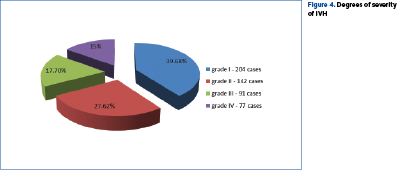
Aims. The authors intend to asses the risk factors, the grades of severity and the complications associated with peri-/intraventricular hemorrhage and to establish by means of standardized clinical criteria and specific investigations (cerebral ultrasonography, electroencephalogram, cerebral CT and MRI) the long-term prognosis. Materials and method. A 5-year retrospective study (2012-2017) performed in the “L. Ţurcanu” Preterms and Neonatology Department, Emergency Hospital for Children, Timişoara, on preterm newborns with clinical and ultrasonographic signs of peri-/intraventricular hemorrhage. Serial cerebral ultrasonography was the elective method in establishing the diagnosis. The exclusion criteria were: cerebral malformations, metabolic disorders, central nervous system infections, and genetic disorders. Results. The main incriminated risk factors were: acidosis, respiratory distress syndrome, hypoxia and hypercarbia. The incidence of peri-/intraventricular hemorrhage was 17.03%. The incidence related to gestational age was higher among very low birth weight preterm infants (64.21%) and extremely-low-birth-weight preterm infants (78.5%). The distribution of cases based on the degree of severity was as follows: grade I – 39.68%; grade II – 27.62%; grade III – 17.70%; grade IV – 15%. Posthemorrhagic obstructive hydrocephalus was the most common complication, in 29.50% of cases (26.04% of preterms with third-degree intraventricular hemorrhage, and 58.33% of those with fourth-degree intraventricular hemorrhage). Conclusions. Although ultrasonography, a rapid and simple diagnostic method, has certain advantages compared to other types of imagistic methods in current medical practice, cerebral MRI or CT is necessary in cases which need ventriculoperitoneal shunt. Moderate-to-severe disabilities such as cerebral palsy, vision or hearing loss and learning disabilities, language, cognitive/behavioural/attentional deficits were present, depending on the size and evolution of the hemorrhagic lesion.
Complicaţii clinice şi ecografice în hemoragia peri-/intraventriculară la nou-născuţii prematuri
Clinical and ultrasound complications in peri-/intraventricular hemorrhage of premature newborns
First published: 18 aprilie 2019
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/ObsGin.67.1.2019.2290
Abstract
Rezumat
Scopul. Autorii analizează factorii de risc, gradele de severitate şi complicaţiile asociate cu hemoragia peri-/intraventriculară şi stabilesc prin intermediul unor criterii clinice standardizate şi investigaţii specifice (ecografie transfontanelară, electroencefalogramă, computer-tomograf şi RMN cerebral) prognosticul pe termen lung. Materiale şi metodă. S-a efectuat un studiu retrospectiv pe o perioadă de 5 ani (2012-2017) în cadrul Clinicii de neonatologie şi prematuri a Spitalului Clinic de Urgenţă pentru Copii „L. Ţurcanu” din Timişoara pe un lot de nou-născuţi prematuri cu semne clinice şi ecografice de hemoragie intracraniană. Metoda de elecţie pentru stabilirea diagnosticului a fost ecografia transfontanelară. Criteriile de excludere au fost malformaţiile cerebrale, tulburările metabolice, infecţiile sistemului nervos central şi anomaliile genetice. Rezultate. Principalii factori de risc incriminaţi au fost acidoza, sindromul de detresă respiratorie, hipoxia şi hipercarbia. Incidenţa hemoragiei peri-/intraventriculare a fost de 17,03%. Incidenţa asociată vârstei de gestaţie a fost mai mare la prematurii cu greutate foarte mică (64,21%) şi extrem de mică (78,5%). Distribuţia cazurilor pe baza gradului de gravitate a fost următoarea: gradul I – 39,68%; gradul II – 27,62%; gradul III – 17,70%; gradul IV – 15%. Hidrocefalia obstructivă posthemoragică a fost cea mai frecventă complicaţie, în 29,50% din cazuri. Concluzii. Deşi ultrasonografia este o metodă rapidă şi simplă de diagnosticare, cu anumite avantaje comparativ cu alte tipuri de metode imagistice în practica medicală curentă, RMN-ul cerebral sau CT-ul este necesar în cazurile care necesită şunt ventriculoperitoneal. Au fost prezente complicaţii moderate până la severe, cum ar fi paralizia cerebrală, pierderea vederii sau a auzului şi tulburări de învăţare, de limbaj, deficite cognitive/comportamentale/atenţionale, în funcţie de mărimea şi evoluţia leziunii hemoragice.
Introduction
Intraventricular hemorrhage (IVH) in neonates is usually defined as the occurrence of bleeding in lateral and third or fourth ventricles, characterized by hyperattenuating fluid, typically seen as layering within the ventricles in imaging studies(1). IVH is the most common form of intracranial hemorrhage in preterm infants, especially those with gestational age under 32 weeks and birth weight under 1500 g(2).
The etiology of IVH is multifactorial and is primarily attributed to the intrinsic fragility of the germinal matrix vasculature and the disturbance in the cerebral blood flow. The importance of IHV resides in the correlation with neurodevelopmental outcome. The incidence of IVH, inversely proportional to gestational age, has gradually declined in the last decades, ranging between 15% and 25%(3). Risk factors, besides prematurity, include: intrapartum asphyxia, maternal chorioamnionitis(4), respiratory distress syndrome, pneumothorax, neutropenia(5), hypoxemia, hypo-/hypercapnia(6), severe acidosis, rapid infusions of bicarbonate and necrotizing enterocolitis(7). The most frequent and serious complication is posthemorrhagic hydrocephalus, which appears in 75% of the survivors of severe forms (III and IV degrees)(8). Other complications, such as multicystic encephalomalacia, periventricular leukomalacia(9), cerebral palsy(10), learning disabilities, language and behavior disorders(11), represent causes of neonatal and pediatric morbidity – neurological sequelae.
Cranial ultrasonography is the elective diagnostic investigation, hemorrhagic lesions having as ultrasonographic correspondent increased echogenicity in the periventricular region. From the echographic point of view, the dilatation of trigon and occipital horns, before frontal horns, could be the only sign of hydrocephalus development. Initially, these areas are the only one dilated in small hemorrhage, while in large hemorrhages the entire ventricular system is dilated.
Aim
The authors intend to asses the risk factors, the grades of severity and the complications associated with IVH and to establish by means of standardized clinical criteria and specific investigations (cranial ultrasonography – CU; EEG; cerebral CT and MRI) the long-term prognosis.
Materials and method
A 5-year retrospective study (2012-2017), performed in the Preterms and Neonatology Department, “L. Ţurcanu” Emergency Hospital for Children, Timişoara, on preterm newborns with clinical and ultrasonographic signs of IHV. Serial cranial ultrasonography was the elective method in establishing the diagnosis and the follow-up of the disease. The exclusion criteria were: cerebral malformations, metabolic disorders, central nervous system infections and genetic disorders.
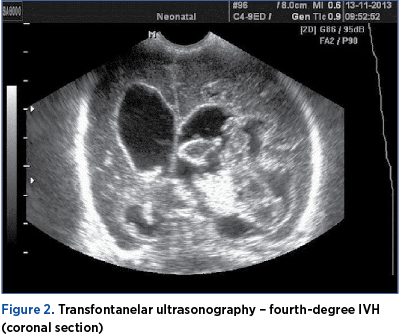
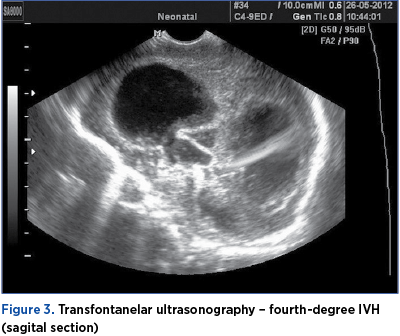
Results
The study included 514 cases out of 3017 preterm infants admitted, the incidence of the disease being 17.03%. The prevalence of IVH was higher among very-low-birth-weight (VLBW; 64.21%) and extremly-low-birth-weight (ELBW; 78.5%) preterms, as opposed to the rest (25.19%). Medium gestational age was 29.7 weeks, and medium birth weight was 1100 g.
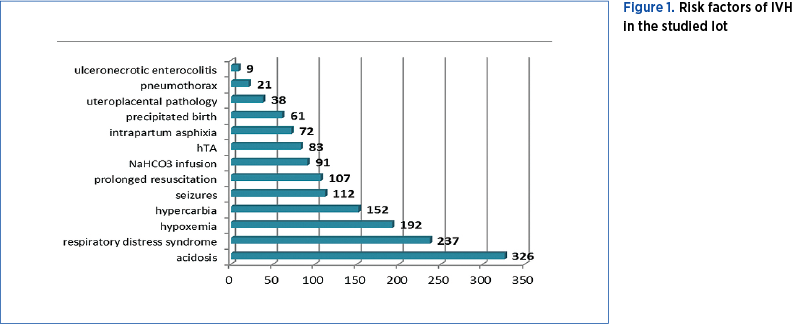
The intrapartum risk factors in 14% of cases were acute hypoxia, complicated labor and precipitate delivery, while postnatal factors included: acidosis, hypoxia, hypercarbia and respiratory distress syndrome.
The majority of the cases were asymptomatic (43%), being diagnosed by routine cranial ultrasound examinations. The saltatory pattern, present in 36% of cases, was associated with progressive neurologic deterioration, abnormal posture and eye movements, and respiratory distress. The rest of 21% of cases had catastrophic clinical findings: bulging anterior fontanelle, dehiscent cranial sutures, projectile vomiting, fixation of pupils, nystagmus, seizures, decerebrate posture, apneic spells, cardiac rhythm disorders.
Cranial ultrasonography was the elective diagnostic investigation, hemorrhagic lesions having as ultrasonographic correspondent increased echogenicity in the periventricular region.
Regarding the severity, most cases presented with echographic signs of first- and second-degree hemorrhage.
In our study group, the cerebral CT and MRI were used in 48 cases with active posthemorrhagic hydrocephalus that demanded ventriculoperitoneal shunt.
The outcome of IVH was depended on the severity of damage:
-
Grade I: complete resorbtion (46.52%), germinal matrix cyst (52.46%), medium ventriculomegaly (1.02%).
-
Grade II: complete resorbtion (38.11%), germinal matrix cyst (34.59%), mild (14.2%), medium (9.01%), and severe ventriculomegaly (11.3%).
-
Grade III: medium ventriculomegaly (36.45%), posthemorrhagic hydrocephalus (26.04%).
-
Grade IV: multicystic encephalomalacia (18.05%), porencephaly (21.66%), posthemorrhagic hydrocephalus (58.33%).
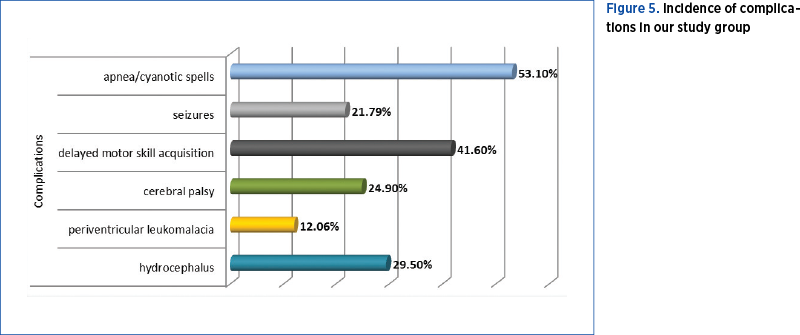
The most common complications of IVH were obstructive hydrocephalus, periventricular leukomalacia, cerebral palsy, learning disabilities, language and behavior disorders, and delayed motor skill acquisitions.
Discussion
IVH remains a serious problem despite significant medical progress in the last decade, because of increasing survival of extremely-low-birth-weight preterms, and because of the potential complications of this disease. The incidence of IVH was relatively low compared to the literature reviews (15-25%)(12). Among the risk factors, the most common were acidosis (pH<7.2), acute respiratory failure (SpO2<80%), neonatal seizures, prolonged neonatal resuscitation, and rapid infusion of bicarbonate.
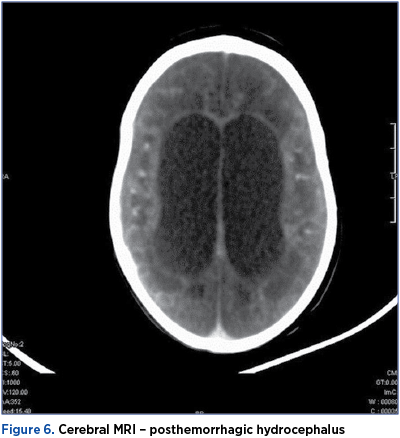
Posthemorrhagic hydrocephalus occurs by modifying the normal flow of cerebrospinal fluid (CSF) during the organization process and reaction to the blood in ventricles leaking secondarily through the fourth ventricle into pericerebellar leptomeninges. Two weeks after hemorrhage occurs, cranial ultrasonography can detect small protein containing particles floating about in the CSF (probably contributing to the obliteration of its distal flow)(13). Distal flow’s obstruction may be found in the lateral ventricle, near the foramen of Monro, in the cerebral aqueduct, near the foramina of the fourth ventricle, in the pericebellar cisterns and in the pericerebral arachnoid spaces, and in the arachnoid granulations of sinuses. Depending on the obstruction location, hydrocephalus can be unilateral, bilateral, internal, external, or mix hydrocephalus (of communicating type). The obstruction of aqueduct and foramina of the fourth ventricle, “isolated fourth ventricle”, is a rare complication after reseated (or revision) repeated shunt. The posthemorrhagic hydrocephalus occurs in 40-80% of all preterm infants with IVH grade III or IV(14), and 25% of preterm infants with posthemorrhagic hydrocefalus need an insertion of a VP shunt(15).
The ventriculoperitoneal shunt can be associated with severe complications like dysfunction or infections, that require the replacement of it. All these will influence the neurodevelopmental outcome. In literature, the prevalence of severe disease was 25% for third-degree IVH, and 15% for fourth-degree IVH(16). In our group of study, posthemorrhagic obstructive hydrocephalus was the most common complication, in 29.50% of cases (26.04% of preterms with third-degree IVH, and 58.33% of those with fourth-degree IVH).
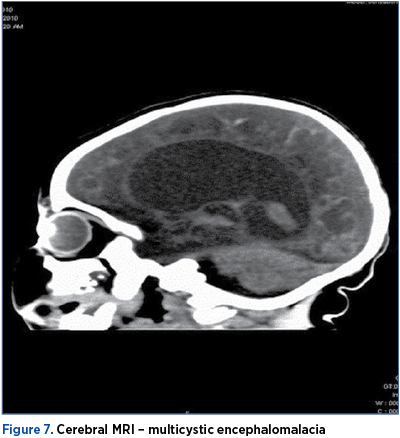
Periventricular leucomalakia (PL) is a cerebral white matter injury in the premature infant. In the USA, each year approximately 60,000 infants (1.5% of the 4,000,000 yearly live births) are born with a birth weight <1500 g. Based on MRI data, at least 50% exhibit some degree of cerebral white matter injury(15). This injury likely accounts for the predominance of neurological deficits observed in the approximately 90% of infants who survive. The most commons deficits in survivors include cerebral palsy in 5-10% and cognitive/behavioral/attentional deficits in 50%(17).
PL is often associated with IVH. This association is present due to similar pathogens and prematurity; 36% premature babies with IVH develop periventricular ecogenes such as PL.
In patients with germinal matrix hemorrhage – intraventricular hemorrhage (GMH-IVH), using MRI is not uncommon to find white matter lesions, with an important role in neurodevelopmental outcome. A.J. Brouwer et al., in 2014, reported that 4% of pacients associating GMH and cystic PLV developed cerebral palsy(12). The incidence of PL in our study group was lower compared to literature (only 12.06%, but 24.9% of our patients developed cerebral palsy).
In almost half of the cases, the ultrasonographic lesions had no clinical correspondent. The symptomatology of the cases with saltatory evolution had subacute onset, in 1-3 days, with hypotonia, nystagmus and posture disorders. The clinical picture of the catastrophic cases included respiratory disorders (apneic spells, increased oxygen demand), seizures, somnolence, decerebrate posture, bulging anterior fontanelle, projectile vomiting, protrusion and mastication movements, fixation of pupils, hypotension and bradycardia, sudden decrease of hemoglobin/hematocrit, and glucose metabolism disorders.
Cases with marked deterioration of general wellbeing disclosed ultrasonographic changes: the big hiperreflectogenic lesions from inside the lateral ventricles and the cerebral parenchyma were followed by major neuropathological lesions: multicystic encephalomalacia, porencephaly, cerebral atrophy and evolutive hydrocephalus.
Conclusions
Cranial ultrasonography was the method of choice in establishing the correct diagnosis.
The prevalence of IVH was 17.03%, being highest among VLBW and ELBW preterms, which presented severe forms of disease.
Although ultrasonography, a rapid and simple diagnostic method, has certain advantages compared to other types of imagistic methods (dynamic evaluation, prompt recognition of complications, lower costs), in current medical practice, cerebral MRI or CT is necessary in cases that need ventriculoperitoneal shunt.
Moderate-to-severe disabilities, such as cerebral palsy, vision or hearing loss and learning disabilities, language, cognitive/behavioural/attentional deficits, were present depending on the size and evolution of the hemorrhagic lesion.
Conflict of interests: The authors declare no conflict of interests.
Bibliografie
2. Osborn DA, Evans N, Kluckow M. Hemodynamic and antecedent risk factors of early and late periventricular/intraventricular hemorrhage in premature infants. Pediatrics. 2003; 112:33-9.
3. Horbar JD, Badger GJ, Carpenter JH, et al. Trends in mortality and morbidity for very low birth weight infants, Pediatrics. 2002; 110(1 Pt 1):143-51.
4. Soraisham AS, Singhal N, McMillan DD, et al. A multicenter study on the clinical outcome of chorioamnionitis in preterm infants. Am J Obstet Gynecol. 2009; 200:372.e1.
5. Palta M, Sadek-Badawi M, Carlton DP. Association of BPD and IVH with early neutrophil and white counts in VLBW neonates with gestational age <32 weeks. J Perinatol. 2008; 28:604.
6. Fabres J, Carlo WA, Phillips V, et al. Both extremes of arterial carbon dioxide pressure and the magnitude of fluctuations in arterial carbon dioxide pressure are associated with severe intraventricular hemorrhage in preterm infants. Pediatrics. 2007; 119:299.
7. Jen HC, Graber JJ, Hill JL, et al. Surgical necrotizing enterocolitis and intraventricular hemorrhage in premature infants below 1000 g. J Pediatr Surg. 2006; 41:1425.
8. Boia M, Manea A, Boia ES, et al. Complications of Peri-/Intraventricular Hemorrhage. Jurnalul Pediatrului. 2005; Year VIII, Vol. VIII, Nr. 29-30, 9:11.
9. Kusters CD, Chen ML, Follett PL, et al. “Intraventricular” hemorrhage and cystic periventricular leukomalacia in preterm infants: how are they related? J Child Neurol. 2009; 24:1158.
10. Futagi Y, Toribe Y, Ogawa K, et al. Neurodevelopmental outcome in children with intraventricular hemorrhage. Pediatr Neurol. 2006; 34(3):219.
11. Luu TM, Ment LR, Schneider KC, et al. Lasting effects of preterm birth and neonatal brain hemorrhage at 12 years of age. Pediatrics. 2009 123: 1037.
12. Brouwer AJ, et al. Early and Late Complications of Germinal Matrix-Intraventricular Haemorrhage in the Preterm Infant: What Is New? Neonatology. 2014;106:296–303.
13. Murphy BP, et al. Posthaemorrhagic ventricular dilatation in the premature infant: natural history and predictors of outcome. Arch of Dis in Childhood, Fetal and Neonatal. 2002; 87, 37-41.
14. Groenendaal F, Termote JU, van der Heide-Jalving M, et al. Complications affecting preterm neonates from 1991 to 2006: what have we gained? Acta Paediatr. 2010; 99:354.
15. Khwaja O, Volpe JJ. Pathogenesis of cerebral white matter injury of prematurity: Arch Dis Child Fetal Neonatal. 2008 March; 93(2): F153–F161. doi:10.1136/adc.2006.108837.
16. McCrea HJ, Ment LR. The Diagnosis, Management and Postnatal Prevention of Intraventricular Hemorrhage in the Preterm Neonate. Clinics in Perinatology. 2008; 35(4):777-vii. doi:10.1016/j.clp.2008.07.014.
17. Nopoulos PC, Conrad AL, Bell EF, Strauss RG, et al. Long-term outcome of brain structure in premature infants: effects of liberal vs restricted red blood cell transfusions Arch Pediatr Adolesc Med. 2011; 165(5):443-50.
Articole din ediţiile anterioare
Formă severă de displazie bronhopulmonară – prezentare de caz
Displazia bronhopulmonară (DBP) este o afecţiune care apare în continuarea detresei respiratorii a prematurului. Boala afectează în special prematu...
Un caz sever de osteopenie de prematuritate
Osteopenia de prematuritate – denumită şi boala metabolică osoasă sau rahitismul prematurului – este o complicaţie tardivă după naşterea prematură,...