Uveal melanoma is the most common primary malignancy of the eye in adults. Little is known about its etiology. One set of uncommon risk factors includes predisposing diseases such as the dysplastic nevus syndrome, atypical ocular nevi, ocular and oculodermal melanocytosis. Another set of host risk factors are ancestry, light skin and iris pigmentation. In addition, a number of environmental and occupational factors are weakly or inconsistently associated with uveal melanoma. Overall mortality rate remains high, because of the development of metastatic disease which is highly resistant to systemic therapy. Improved understanding of the molecular pathogenesis of cancer has led to a new generation of therapeutic agents that interfere with a specific pathway critical in tumor development or progression. The aim of our clinical study is to establish the mechanisms through which the free radicals of oxygen are involved in the cellular cytotoxicity as a measurement of the treatment efficiency. We investigated 44 patients in the dynamic of more than two years of oncological treatment. In the serum, we monitored the oxidative stress index obtained by the fraction of lipid peroxidation reaction, the total thiol groups and the total antioxidant activity. The obtained data indicated an increase of the oxidative stress index in the first part of the treatment, followed by a stationary of the level even when the patients had a good treatment response. Our results suggest the installation of a systemic cytotoxicity due to the adaptative mechanisms of the tumoral presence.
Monitorizarea statusului oxidativ la pacienţii cu melanom uveal în timpul tratamentului oncologic
Monitoring oxidative status in patients with uveal melanoma during oncological treatment
First published: 14 decembrie 2019
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/OnHe.49.4.2019.2742
Abstract
Rezumat
Melanomul uveal este cea mai frecventă malignitate primară a ochiului la adulţi. Se cunosc puţine date despre etiologia sa. Factorii de risc includ predispoziţia organismului adult la diverse maladii, cum ar fi sindromul nevilor displazici, nevii oculari atipici şi melanocitoza oculară şi oculodermică. Un alt set de factori de risc îl reprezintă componenta genetică, tegumentele albe, puţin pigmentate şi pigmentarea irisului. În plus, o serie de factori de mediu şi ocupaţionali sunt parţial asociaţi cu melanomul uveal. Rata mortalităţii globale rămâne ridicată, din cauza progresiei şi a dezvoltării bolii metastazice, care este foarte rezistentă la terapia sistemică. Îmbunătăţirea cunoştinţelor legate de patogeneza moleculară a cancerului a dus la identificarea unei noi generaţii de agenţi terapeutici care interferează cu o cale specifică, critică în dezvoltarea sau progresia tumorii. Scopul acestui studiu clinic este de a stabili mecanismele prin care radicalii liberi de oxigen sunt implicaţi în citotoxicitatea celulară ca o măsură a eficienţei tratamentului. Am investigat 44 de pacienţi în dinamica tratamentului oncologic, urmăriţi timp de peste doi ani. S-au monitorizat din ser indicele de stres oxidativ obţinut prin fracţia reacţiei de peroxidare a lipidelor, alături de nivelul tiolilor totali şi de activitatea totală antioxidantă. Datele obţinute indică o creştere a indicelui de stres oxidativ în prima parte a tratamentului, urmată de o staţionare a nivelului, chiar şi atunci când pacienţii au un răspuns bun la tratament. Rezultatele noastre sugerează instalarea unei citotoxicităţi sistemice datorate mecanismelor adaptative ale prezenţei tumorale.
Introduction
Uveal melanoma is the most common primary intraocular malignancy in adults and occurs with a frequency of 6 to 7 cases per 1 million adults(1). Although cutaneous and uveal melanomas arise from neural crest progenitors, they differ significantly in their epidemiologic, cytogenetic, metastatic and immunologic characteristics(2-4). Overall mortality rate remains high because of the development of metastatic disease which is highly resistant to systemic therapy. Improved understanding of the molecular pathogenesis of cancer has led to a new generation of therapeutic agents that interfere with a specific pathway critical in tumor development or progression(5). Although no specific genes have been linked to the pathogenesis of uveal melanoma which differ from that of cutaneous melanoma, progress has been made in identifying potential targets involved in uveal melanoma apoptosis, proliferation, invasion, metastasis and angiogenesis(6). Despite diagnostic accuracy and the introduction of new conservative treatment modalities over the past decades, the survival rate in patients affected by uveal melanoma has not been substantially modified(7). Furthermore, the identification of risk factors for the development of this tumor has proved to be unsuccessful so far(8). Although clinical, epidemiologic and biological observations support the hypothesis that UV light is an etiologic agent for cutaneous melanoma in white populations, no irrefutable evidence has appeared in favor of a role for sunlight in uveal melanoma(9,10). However, in both cutaneous and uveal melanomas, triggering factors related to the generation of free radical species – and subsequent oxidative stress – can be considered(11). Oxidative stress results from an imbalance between prooxidant agents and antioxidant systems. Wavelengths in the UVA range (320-400 nm) are not directly absorbed by DNA, but are capable of generating reactive oxygen species (ROS), which are considered to be effective in inducing cutaneous melanoma, at least in animal models(12). On the other hand, uveal vascular changes may significantly increase the amount of ROS generated. Therefore, the evaluation of the cellular antioxidant system could be crucial in understanding the pathway of tumorigenesis(13).
The aim of our clinical study is to establish the mechanisms through which the free radicals of oxygen are involved in the cellular cytotoxicity as a measurement of the treatment efficiency.
Materials and method
We investigated 44 patients who were diagnosed with uveal melanoma (UM) during the period of their own treatment. Due to the heterogeneity of the patients (age, sex, disease stage at initial presentation), they were grouped in batches depending on the treatment, as follows: Group 1 (n=14) – patients subjected to radiotherapy; Group 2 (n=18) – patients whose only form of therapy was enucleation; Group 3 – patients who after surgery received polichemotherapy and immunotherapy (n=12). The patients were informed about the study, about the moments of the harvest, and they agreed by registered consent to participate in this study, approved by the ethics committee of the institute. The data obtained did not influence the treatment, being used strictly for research.
All patients were investigated clinically and biochemically in dynamic therapy (at the beginning and at the end of primary medical acts, and regular control intervals over two years). Until now, there were 7 determinations of biochemical parameters of oxidative stress. These are the monitoring markers of the lipid peroxidation reaction by measuring the malondialdehyde final reaction product(14), the determination of the total albuminic thiol groups (using the Shoshinsky method(15)) as a measure of oxidative degradation of circulating proteins, and total antioxidants were measured biochemically by a method that utilizes the ability of the serum to reduce iron. At low pH, the Felll-tripyridyl-s-triazine complex (Felll-TPTZ) is reduced to the ferrous form with the formation of a new complex that develops an intense blue color, measurable, with a maximum absorption at 593 nm. Any reaction with a positive redox potential under the aforementioned conditions can lead to the reduction of the Felll-TPTZ complex. Using an excess of Fe, the limiting factor of the Felll-TPTZ complex and the color formation represent the reducing ability of the sample(16).
Results and discussion
Primary UM tumors are usually treated with surgery (plaque brachytherapy, enucleation of the tumor-bearing eye), radiation therapy and/or thermotherapy. Despite these treatments, about 50% of all UM cases progress to metastatic disease within 15 years of diagnosis. Most metastases of UM (80-90%) occurs in the liver. Enucleation was performed for large tumors, if it appeared unlikely to recover visual function, for tumors with optic nerve invasion or extraocular extension, and for tumors with a flat large infiltration of the choroid. Both sexes are affected in equal numbers.
The values of lipid peroxidation in the patients investigated in the dynamics of the treatment are presented in Figure 1.
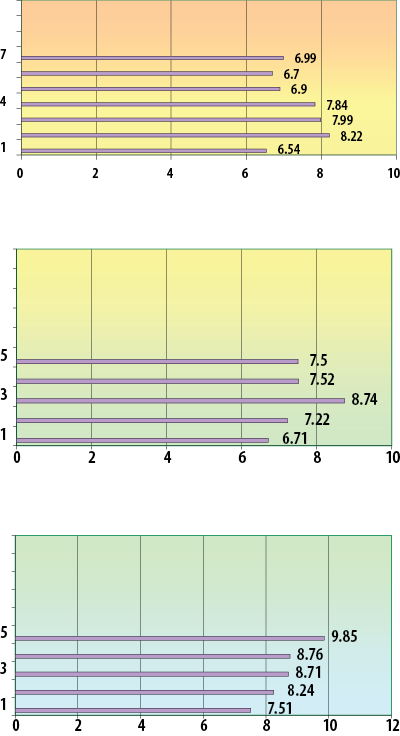
The lipid peroxidation reaction measures the extent of the oxidative attack and especially of the reactive oxygen species on the lipids. From the obtained data, we can notice the increased values in all patients when the treatment was initiated, suggesting that the tumor is an inductive factor of radical reactive species, given by the intense metabolism and perhaps by a specific metabolic reprogramming of the tumor cells(17). During the treatment, this destructive attack manifested at the molecular level on the lipids increases continuously due to chemotherapy, decreases after enucleation and decreases at the end of radiotherapy. The intensity of the toxicity given by the destruction of lipids by their peroxidation is proportional to the applied treatment and its efficiency. In case of radiotherapy, the decrease occurs one month after the completion of the therapeutic irradiation, after enucleation the intensity of the reaction also decreases, in the dynamics of chemotherapy, as expected, the intensity of the molecular damage, given by the efficiency of the treatment, increases. Regarding the group treated strictly by enucleation, the increase of the level of free radicals is observed even after the first determination. Two hypotheses are outlined: the first hypothesis is that of triggering a cascade reaction producing free circulating radicals, which continues after the surgical removal of the tumor (at least the primary one, clinically detectable); the second hypothesis is that oxidative stress and the level of free radicals, at least at the first determination, are increased by the surgical trauma itself.
Radiation therapy is itself an inducer of oxidative stress. There is a peculiarity of this type of treatment, which affects in large numbers the normal cells of the body also, associated with a level of free radicals in excess of that induced by the presence of the tumor. After stopping the mechanism attributed to the therapy, the substrate is depleted, and the main source of free radicals remains the lipids of the tumor cells.
Further, the oxidative destruction reactions manifested in circulating proteins were measured by determining the final product, total thiols. The values obtained in the dynamics of the treatments are presented in Figure 2.
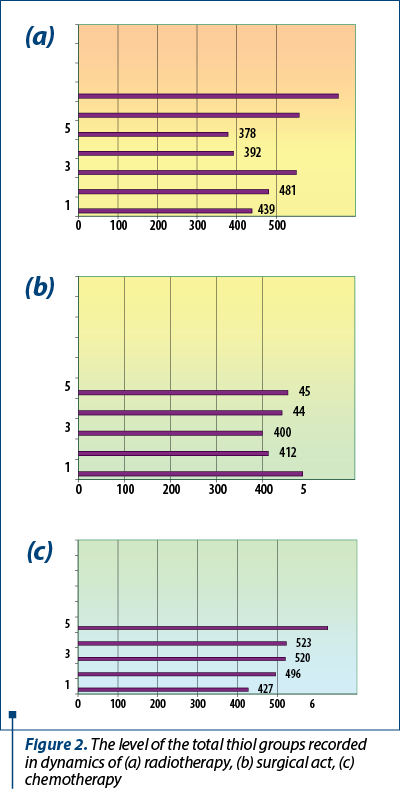
The data recorded by us suggest an increase in the intensity of oxidative degradation of proteins in the dynamics of chemotherapy, as in the case of the oxidative attack manifested on lipids. This fact demonstrates a destructive molecular toxicity given to the treatment, clearly suggesting the efficacy of chemotherapy in uveal melanoma. The results obtained in the dynamics of radiotherapy and of the operative act demonstrate the installation of an intense oxidative stress, irreversible, but later, assuming that the primary target of the oxidative attack are the lipids, only then the proteins are affected.
To highlight the installation of oxidative stress, measurements were made from the same biological samples and in the same dynamics, of the level of total antioxidants, as an indication of the antioxidant fighting capacity at the molecular level. The values obtained are displayed in Figure 3.
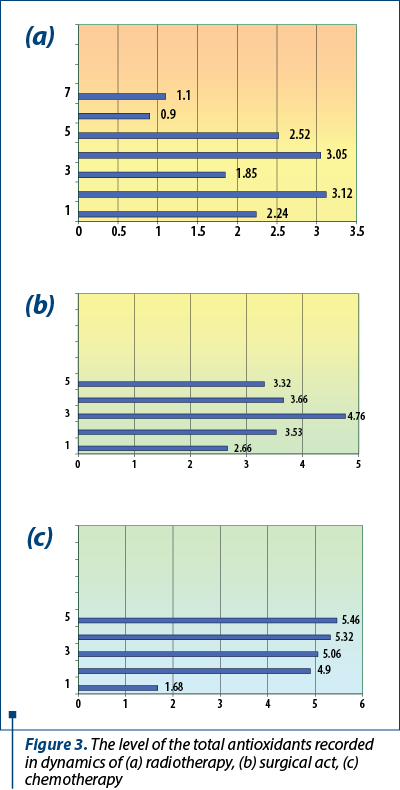
Antioxidant levels increase with the intensification of oxidation reactions, as a result of the attempt at the cellular level to maintain the oxidation/reduction balance in relative equilibrium. In the uveal melanoma, the oxidative level is much increased, justifying to say that in this location there is a pronounced oxidative stress, more intense, with a low capacity of the endogenous antioxidant system to diminish this imbalance. Aerobic metabolism in uveal melanoma is pronounced, destructive, with excess production of reactive oxygen species and with minimal homeostasis capacity. At the same time, it is necessary, in order to establish a complete picture of the extent of oxidative stress, the measurement of several molecular parameters, such as the oxidative degradation of nucleic acids, glycoproteins or other essential macromolecules.
Conclusions
For all the investigated patients, a series of biochemical tests were carried out that aimed to monitor the oxygen metabolism and the installation of oxidative stress in the dynamics of the treatment. We should mention that, regardless of the treatment, its purpose is to reduce the tumor size, improve the relative health and quality of life of the patient.
In the context in which the values of the peroxides are higher than of the thiols, one can affirm with certainty that the primary target of the oxidative attack given by the reactive oxygen species is represented by the lipid molecules and only then the protein ones are attacked.
At the end of the antioxidant reactions, we obtained the instalation of the oxidative stress which means an unbalance between the reactive oxygen production to the detriment of the natural antioxidant system ability to reduce this. By increasing the level of total antioxidants, it is deduced the inability/overcoming of their capture and counteracting systems, especially in the last determinations, overall speaking, under the most probable conditions of the progression of the neoplastic disease. Several studies attest that oxidative stress resulting from this imbalance has a causative role, but is also a consequence of carcinogenesis, interfering with all phases of this process, such as initiation, promotion, progression, invasion and metastasis(18,19).
Conflict of interests: The authors declare no conflict of interests.
Bibliografie
- Jensen OM, Parkin DM, MacLennan R, Muir CS, Skeet RG. Cancer registration. Principles and methods. vol. 95. Lyon: IARC, 1991.
- Stang A, Hakulinen T. Eye melanoma in Finland: time trends and relative survival rates. Strahlenther Onkol. 2000; 176: S153.
- Vajdic CM, Kricker A, Giblin M, McKenzie J, Aitken J, Giles GG, Armstrong BK. Incidence of ocular melanoma in Australia from 1990 to 1998. Int J Cancer. 2003; 105: 117–22.
- Inskip PD, Devesa SS, Fraumeni JF Jr. Trends in the incidence of ocular melanoma in the United States, 1974-1998. Cancer Causes Control. 2003; 14: 251–7.
- Nathan P, Cohen V, Coupland S, Curtis K, Damato B, Evans J, Fenwick S, Kirkpatrick L, Li O, Marshall E, McGuirk K, Ottensmeier C, Pearce N, Salvi S, Stedman B, Szlosarek P, Turnbull N; United Kingdom Uveal Melanoma Guideline Development Working Group. Uveal Melanoma UK National Guidelines. European Journal of Cancer. 2015; 51, 2404–12.
- Onken MD, Li J, Cooper JA. Uveal Melanoma Cells Utilize a Novel Route for Transendothelial Migration. PLoS One. 2014 Dec 15; 9(12):e115472.
- Chattopahdyay C, Kim DW, Gombos D, Oba J, Qin Y, Williams M, Esmaeli B, Grimm E, Wargo J, Woodman S, Patel S. Uveal Melanoma: From Diagnosis to Treatment and the Science in Between. Cancer. 2016; 122(15): 2299–2312.
- Gallagher RP, Elwood JM, Rootman J, Spinelli JJ, Hill GB, Threlfall WJ, Birdsell JM. Risk factors for ocular melanomas: Western Canada Melanoma Study. J Natl Cancer Inst. 1985; 74: 775–8.
- Seddon JM, Gragoudas ES, Glynn RJ, Egan KM, Albert DM, Blitzer PH. Host factors, UV radiation, and risk of uveal melanoma. A case-control study. Arch Ophthalmol. 1990; 108: 1274–80.
- Tucker MA, Shields JA, Hartge P, Augsburger J, Hoover RN, Fraumeni JF Jr. Sunlight exposure as risk factor for intraocular malignant melanoma. N Engl J Med. 1985; 313: 789–92.
- Majmundar AJ, Wong WJ, Simon MC. Hypoxia-inducible factors and the response to hypoxic stress. Mol Cell. 2010 Oct 22; 40: 294–309.
- Holly EA, Aston DA, Char DH, Kristiansen JJ, Ahn DK. Uveal melanoma in relation to ultraviolet light exposure and host factors. Cancer Res. 1990; 50: 5773–7.
- Dörschmann P, Kopplin H, Roider J, Klettner A. Effects of Sulfated Fucans from Laminaria hyperborea Regarding VEGF Secretion, Cell Viability, and Oxidative Stress and Correlation with Molecular Weight. Mar Drugs. 2019 Sep 25;17(10). pii: E548. doi: 10.3390/md17100548.
- Nair U, Bartsch H, Nair J. Lipid peroxidation-induced DNA damage in cancer-prone inflammatory diseases: a review of published adduct types and levels in humans. Free Radic Biol Med. 2007; 43(8):1109-20.
- Albini A. Standardization of protein free SH groups in blood plasma. Boll Soc Ital Sperim. 1990, 18:1829-98.
- Halliwell B. Antioxidant characterization. Methodology and mechanisms. Biochemical Pharmacology. 1995; 49(10): 1341-8.
- Sciacovelli M, Frezza C. Metabolic reprogramming and epithelial-to-mesenchymal transition in cancer. FEBS J. 2017 Oct; 284(19): 3132–3144.
- Snezhkina AV, Kudryavtseva AV, Kardymon OL, Savvateeva MV, Melnikova NV, Krasnov GS, Dmitriev AA. ROS Generation and Antioxidant Defense Systems in Normal and Malignant Cells, Oxidative Medicine and Cellular Longevity. Oxidative Medicine and Cellular Longevity. Volume 2019; https://doi.org/10.1155/2019/6175804.
- Stang A, Ahrens W, Anastassiou G, Jöckel KH. Phenotypical characteristics, lifestyle, social class and uveal melanoma. Ophthalmic Epidemiol. 2003; 10: 293–302.
Articole din ediţiile anterioare
Rolul antioxidanţilor neenzimatici în diminuarea stresului oxidativ asociat creşterii şi dezvoltării tumorilor maligne
Speciile active ale oxigenului sunt produse ca o consecinţă a metabolismului aerobic normal. Dintre aceştia, radicalii liberi sunt de obicei remeta...
Tiolii albuminici - potențiali markeri ai statusului oxidativ în cancer
Stresul oxidativ a fost asociat, pentru o lungă perioadă de timp, cu fiziopatologia cancerului și a altor boli. Speciile reactive generate în exces...
Melanomul malign ocular – corelaţia evoluţiei clinice cu parametrii biologici şi imunologici
Melanomul malign, deşi relativ mai puţin frecvent comparativ cu restul neoplaziilor cutanate, este responsabil de cele mai multe fatalităţi prin c...
Comparing oxidative metabolism in patients with uveal melanoma versus cutaneous melanoma
Melanoamele maligne (MM) cutanate şi uveale prezintă caracteristici diferite de metastazare, fără un tratament sistemic standardizat pentru mela...