Corelaţii anatomoclinice şi ecografice în hipertensiunea arterială indusă de sarcină
Anatomical-clinical and ultrasound correlations in pregnancy-induced hypertension
Abstract
Pregnancy-induced hypertension is diagnosed by an increase in systolic blood pressure above 140 mmHg and diastolic blood pressure above 90 mmHg, after 20 weeks of gestational age, in a normotensive pregnant woman before pregnancy. The preeclamptic syndrome is specific to gestation, can affect all organs, and is characterized by the association of hypertension, proteinuria and oedemas. The aim of this paper is that, through early clinical-paraclinical and especially ultrasound monitoring, to achieve an adequate completion of pregnancy and childbirth, with at least three fundamental objectives: minimal maternal and foetal trauma; the birth of a child with the highest chances of survival, with an immediate and distant good functional prognosis; the complete restoration of the mother’s health condition, allowing an optimal life, with a good, reproductive and maternal prognosis in the future. Early clinical-paraclinical and especially ultrasound monitoring ensure an adequate completion of the pregnancy to term and childbirth. The foetal ultrasound, the foetal biophysical score and the multivascular Doppler velocimetry are of major importance in determining the obstetrical attitude – vaginal birth or foetal removal by caesarean section, in correlation with foetal intrauterine vitality, weight and Apgar score. The foetal, vital and functional prognosis, immediate and distant, was good. The complete restoration of the mother’s health allows an optimal life, with a good reproductive and maternal prognosis in the future.Keywords
pregnancy-induced hypertensionpreeclampsiamaternal-foetal ultrasoundDoppler velocimetryRezumat
Hipertensiunea arterială indusă de sarcină este diagnosticată prin creşterea tensiunii arteriale sistolice peste 140 mmHg şi a celei diastolice peste 90 mmHg, după 20 de săptămâni de vârstă gestaţională, la o gravidă normotensivă anterior sarcinii. Sindromul preeclamptic este specific gestaţiei, poate afecta toate organele şi se caracterizează prin asocierea hipertensiunii arteriale cu proteinurie şi edeme. Scopul acestei lucrări este ca, prin monitorizarea precoce clinico-paraclinică şi mai ales ecografică, să se realizeze o finalizare adecvată a sarcinii spre termen şi a naşterii, cu cel puţin trei obiective fundamentale: minime traumatisme materne şi fetale; naşterea unui copil cu şanse cât mai mari de supravieţuire, cu prognostic funcţional imediat şi îndepărtat bun; refacerea cât mai completă a stării de sănătate a mamei, care să permită o viaţă optimă, cu un prognostic bun, reproductiv şi matern, în viitor. Monitorizarea precoce clinico-paraclinică şi mai ales ecografică asigură o finalizare adecvată a sarcinii spre termen şi a naşterii. Studiul ecografic fetal, scorul biofizic fetal şi velocimetria Doppler multivasculară au o importanţă majoră în stabilirea atitudinii obstetricale – naştere pe cale vaginală sau extragerea fătului prin operaţie cezariană, în corelaţie cu vitalitatea intrauterină a fătului, greutatea şi scorul Apgar. Prognosticul fetal, vital şi funcţional, imediat şi îndepărtat, a fost bun. Refacerea cât mai completă a stării de sănătate a mamei permite o viaţă optimă, cu un prognostic bun, reproductiv şi matern, în viitor.Cuvinte Cheie
hipertensiune arterială indusă de sarcinăpreeclampsieecografie materno-fetalăvelocimetrie DopplerIntroduction
Pregnancy-induced hypertension (PIH) complicates 5-10% of pregnancies and is a major cause of maternal morbidity and mortality, along with haemorrhages and infections. Gestational hypertension is followed by preeclampsia (PE) in almost half of the cases, preeclampsia being identified in 3.9% of the total number of pregnancies(1). PIH determines 16% of the maternal mortality cases(2). More than half of these deaths due to gestational hypertension could be prevented(3,4). The incidence is higher in pregnant women with a history of PIH, in overweight and obese women, in pregnant women with twin or multiple pregnancies, and in pregnant women with molar pregnancy, with thrombophilia(5-7).
Pregnancy-induced hypertension is diagnosed by an increase in systolic blood pressure above 140 mmHg and diastolic blood pressure above 90 mmHg, after 20 weeks of gestational age, in a normotensive pregnant woman before pregnancy. Pregnant women with an increase in systolic blood pressure of 30 mmHg or diastolic blood pressure of 15 mmHg should be closely monitored, as they may develop eclamptic convulsions even at blood pressure values below 149/90 mmHg(8). A sudden increase in average blood pressure that occurs late in pregnancy may cause risks similar to those of preeclampsia, even though the blood pressure values are less than 140/90 mmHg(9).
The preeclamptic syndrome is specific to pregnancy and can affect all organs. It is characterized by the association of hypertension and proteinuria over 300 mg/24 hours, protein/creatinine ratio greater than 0.3 or thrombocytopenia below 100,000/mm3, creatinine above 1.1 mg/dl, indicating renal failure, increased transaminases (ALAT, ASAT), that suggests liver damage, headache, visual disturbances, convulsions, that objectify neurological suffering, and pulmonary oedema(10).
Pregnant women normally develop a refractory status to vasopressors(11). Pregnant women with early preeclampsia show an increased vascular reactivity to norepinephrine and angiotensin II(12,13). Increased sensitivity to angiotensin II precedes the onset of gestational hypertension(14).
The genotypic and phenotypic expression of preeclamptic syndrome is influenced by several factors: maternal and paternal genotype, associated diseases, genomic ethnicity, interactions between genes, epigenetic phenomenon and gene-environment interactions.
The presence of chorionic villi is essential for the appearance of this pathology.
The extent of abnormal trophoblastic invasion is thought to correlate with the severity of the hypertensive disorder(15,16).
The cascade of events that cause preeclamptic syndrome is dominated by abnormalities that cause damage to the endothelial vascular system, a damage that results in vasospasm, plasma transudation and ischemic and thrombotic sequelae.
The aim of this paper is that, through early clinical-paraclinical and especially ultrasound monitoring, to achieve an adequate completion of pregnancy to term and childbirth, with at least three fundamental objectives: minimal maternal and foetal trauma, the birth of a child with the highest chances of survival, with an immediate and distant good functional prognosis, the complete restoration of the mother’s health condition, allowing an optimal life, with a good reproductive and maternal prognosis in the future.
Method
The study was performed on a group of 24 pregnant women with pregnancy-induced hypertension and five patients with severe forms of preeclampsia. The patients were hospitalized in the maternity ward of the County Emergency Clinical Hospital of Craiova. The parturients’ age ranged from 17 to 40 years old. The written consent of the patients was obtained in the observation sheets.
The positive diagnosis at hospitalization was established based on the general and obstetrical clinical examination. The pregnant women had a gestational age between 24 and 37 weeks of gestation; 16 pregnant women had a gestational age between 24 and 32 weeks, and 13 pregnant women had a gestational age between 33 and 37 weeks.
The symptoms at admission were polymorphic, the pregnant women presenting: painful uterine contractions, marked excess weight in the last two weeks, generalized oedemas, headache, high blood pressure.
The paraclinical investigations performed were: complete blood count, blood group, Rh, glycaemia, urea, creatinine, uric acid, liver transaminases, blood coagulation, serum and urinary ionogram, proteinuria, complete urine examination.
To these tests there were added foetal electrocardiographic monitoring, uterine arteries, middle cerebral arteries and umbilical artery Doppler velocimetry, obstetrical ultrasound for foetal biophysical profile and the condition of foetal appendages – placenta, amniotic fluid.
The blood pressure was also measured, and electrocardiography, cardiology examination, neurology, ophthalmology and nephrology examinations were performed.
The Gant test or the roll-over positional test was performed in correlation with the average blood pressure over 105 mmHg. The blood pressure (BP) values taken from the pregnant woman in the elongated/left lateral decubitus position were compared, the BP was taken every 5 minutes until it stabilized for at least 15 minutes. Then, the BP was measured in dorsal decubitus position or with the pregnant woman standing 1 and 5 minutes after changing position. Any value greater than 20 mmHg of diastolic pressure was considered abnormal.
Results
The 24 pregnant women with PIH did not show any suggestive clinical changes. The BP was between 140-150 mmHg/80-90 mmHg. The haematological and urinary investigations were within normal limits.
The cardiology examination performed by patients showed an increase in systolic blood pressure over 140 mmHg and diastolic blood pressure over 90 mmHg. Increased systolic blood pressure by more than 30 mmHg and increased diastolic blood pressure by more than 15 mmHg were considered as pregnancy-induced hypertension. The electrocardiography did not show changes in the terminal phase or other pathologies.
Ophthalmologic and neurological examinations did not describe organic or functional lesions at these levels.
The patient’s monitoring recorded blood pressure values over 140/90 mmHg, pulse 70-95 beats/min, present diuresis, absent proteinuria, and foetal heartbeats with values of 138-146 beats/minute.
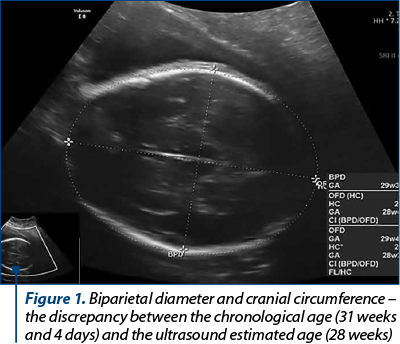
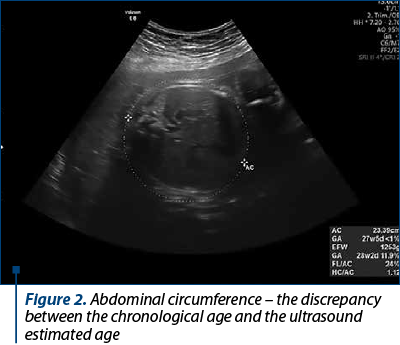
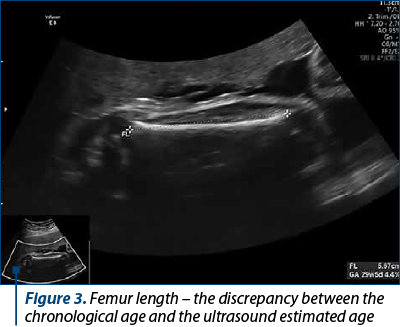

Foetal ultrasound monitoring showed a slight restriction of intrauterine growth, foetal biophysical profile 8. The uterine artery, the umbilical artery and middle cerebral artery Doppler velocimetry showed no significant pathological changes.
In the five cases of severe preeclampsia, at the objective clinical examination we found oedema of the eyelids, face, hand and lower limbs. The oedemas were suggestive for the diagnosis of preeclampsia, given that they occurred within the last 48 hours.
These were associated with an increase in systolic and diastolic blood pressure of over 160/110 mmHg, with oliguria and proteinuria.
The parturients presented intense headache, diplopia, and decreased visual acuity due to amaurosis.
The haematological investigations showed a decrease in fibrinogen below 400 mg/dl, a decrease in platelets below 100,000/mm3, an increase in fibrin degradation products over 16 micrograms/ml, an increase in serum creatinine over 1.1 mg/dl, an increase in uricemia over 5.7 mg/dl, increased prothrombin time over 20 seconds, and an increase in liver transaminases (ALAT/GPT, ASAT/GOT) over 70 U/L.
The urinary investigations showed proteinuria over 0.3 g/l urine/24 hours.
The cardiology examination showed an increase in systolic and diastolic blood pressure above 160/110 mmHg, without shortness of breath or other changes in the heart rate.
The ophthalmologic examination revealed retinal arteriole spasm, retinal oedema, and retinal haemorrhage.
The neurological examination showed headache, agitation, visual disturbances (scotomas, diplopia), speech and hearing disorders, cerebral vomiting, and vertigo.
The evolution of preeclampsia being nonlinear, the maternal-foetal condition can suddenly worsen; the non-stress test was repeated 2-3 times a day.
The obstetric ultrasound in the five parturients with severe preeclampsia showed restriction of intrauterine foetal growth, calcifications at the level of the maternal and foetal basal plate, and foetal biophysical profile between 6 and 8 (Figures 1-4).
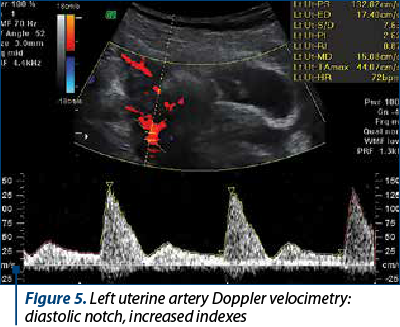
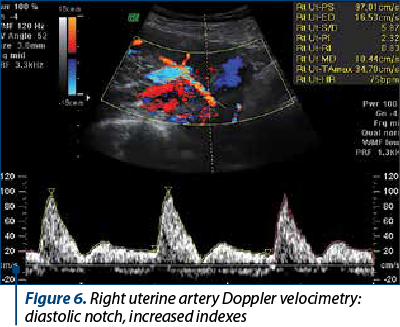
Doppler velocimetry in the uterine artery (Figures 5 and 6) showed an increase in the resistance index (RI)and the pulsatility index (PI) (RI 0.83-0.92; PI 2.32-2.78).
Doppler velocimetry in the umbilical artery (Figure 7) indicated an increased pulsatility and resistance index (PI 1.10-1.70; RI 0.75-0.91).
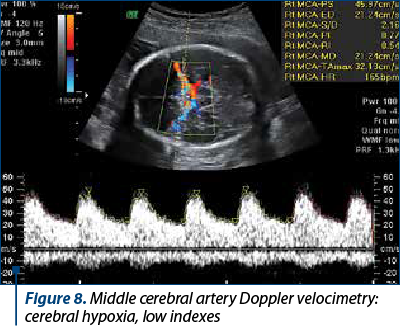
Middle cerebral artery Doppler velocimetry (Figure 8) showed reduced pulsatility index and decreased resistance index (PI 1.20-1.70; RI 0.53-0.67).
The cerebroplacental IP ratio between MCA and UA was supraunitary.
Of the 29 cases studied, 18 pregnant women gave birth vaginally, spontaneously and with forceps in 4 cases, and in 11 cases the foetuses were removed by caesarean section. The Apgar birth score was 8-9 in 22 cases, and 6-7 in 7 cases. The foetuses had a weight between 2600 and 3200 g.
Discussion
Late-onset PE is more common than early-onset PE (before 32 weeks of gestation), the comparative incidence being 2.7% in late-onset PE, compared to 0.3% in early-onset PE. Approximately 10-15% of maternal mortality is caused by complications of PIH, along with haemorrhage, cardiovascular disease and thromboembolism. The essential element in the appearance of PE seems to be the incomplete invasion of the trophoblast on the decidualized endometrium, more precisely at the level of the spiral arterioles. Compared to a normal pregnancy, in a pregnancy with PIH the invasion is made in the distal third of the spiral arterioles, the other two proximal thirds being approximately intact. The conflict between the trophoblast and the vascular endothelium disrupts the balance between the synthesis and discharge of vasoconstrictor substances (thromboxane A2 and endothelin ET1) to the detriment of the vasodilators (prostacyclin PG I2 and nitric oxide). This imbalance also affects the small vessels in the vasa vasorum that cover the spiral arteriole like a sleeve, nourishing its wall, the prolonged spasm induced by vasoconstrictors producing ischemia, necrosis and infarction of the walls of the spiral arterioles, which can rupture, a predominant aspect in a major complication of PIH, such as retroplacental hematoma and uteroplacental apoplexy/abruptio placentae(17,18). The vasospasm in the uteroplacental territory later extends to the renal, pulmonary, cardiac and cerebral ones(19, 20). The hypoperfusion of the intravenous space with oxygen, water, electrolytes and nutrients leads to chronic foetal distress, foetal hypotrophy and IUGR.
The vascular resistance in the uterine circulation decreases in the first part of a normal pregnancy due to the invasion of maternal uterine vessels with trophoblastic tissue, therefore the uterine artery Doppler ultrasound is useful in assessing pregnancies with maximum risk of uteroplacental insufficiency(21). The uterine artery Doppler examination shows a high rate of diastolic flow in a normal pregnancy. Increased flow resistance and the development of a diastolic notch are associated with the further development of pregnancy-induced hypertension, preeclampsia and intrauterine growth restriction(22). The uterine artery Doppler velocity is considered a sign of placental dysfunction, with the aim of assessing the risk of intrauterine foetal death and the risk of premature birth(23). The risk of foetal death before 32 weeks, when associated with preeclampsia, uteroplacental apoplexy or growth restriction, is significantly corroborated with high resistance of blood flow in the uterine artery(24).
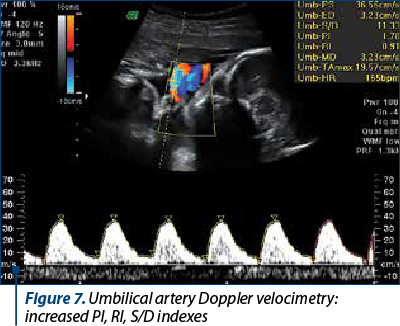
The umbilical artery Doppler examination is corroborated with information on the health of the foetus, as it provides information on blood flow throughout the cardiac cycle. In extreme cases of intrauterine growth restriction, the end-diastolic flow is absent or even reversed, this change being associated with more than 70% obliteration of the placental villi arterioles(25-27). The perinatal mortality rate in the absence of end-diastolic flow is approximately 10%, and for reversed end-diastolic flow, approximately 33%(28). The umbilical artery Doppler examination brings an important benefit in the management of complicated pregnancies by intrauterine growth restriction(29), as in pregnancies with intrauterine growth restriction and the absence of end-diastolic flow on the umbilical artery, along with monitoring, expectation until birth at 34 weeks is the preferred option, and in those with reversed end-diastolic flow the conduct is expected until birth at 32 weeks(30).
The middle cerebral artery Doppler examination is also useful in assessing intrauterine growth restriction. Foetal hypoxemia causes an increase in blood flow to the brain, heart and kidneys and, thus, an increase in the end-diastolic flow of the middle cerebral artery. This phenomenon of “brain protection” is associated with perinatal morbidity and mortality(31,32).
Conclusions
Early clinical-paraclinical and especially ultrasound monitoring ensures an adequate completion of the pregnancy to term and of the birth.
The foetal ultrasound, the foetal biophysical score and the multivascular Doppler velocimetry are of major importance in determining the obstetric attitude, vaginal birth or foetal removal by caesarean section, in correlation with foetal intrauterine vitality, weight and Apgar score.
The foetal, vital and functional prognosis, immediate and distant, was good.
The complete restoration of the mother’s health allows an optimal life, with a good reproductive and maternal prognosis in the future.
Conflict of interests: The authors declare no conflict of interests.
Bibliografie
-
Martin JN Jr, Owens My, Keiser SD. Standardised Mississippi Protocol treatment of 190 patients with HELLP syndrome slowing disease progression and preventing new major maternal morbidity. Hypertens Pregnancy. 2012;31(1):79-90.
-
Khan KS, Wojdyla D, Sai L. Who analysis of causes of maternal death: a systematic review. Lancet. 2006;367(9516):1066-74.
-
Mitran M, Velicu O, Bohîlţea R. Diagnosis and prevention of preeclampsia – literature review. Ginecologia.ro. 2017;5(17):10-2.
-
Berg CJ, Harper MA, Atkinson SM. Preventability of pregnancy related deaths. Obstet Gynecol. 2005;106(6):1228-34.
-
Arsene LV, Munteanu O, Voicu D. Assessment of the fetal-placental unit using clinical and ultrasound evaluation and inherited thrombophilia in pregnant patients. Ginecologia.ro. 2018;6(21):18-22.
-
Brăila AD, Neacşu A, Muşetescu AE. Biochemical markers in pregnancy associated with Sjogren syndrome and thrombophilia. Chim. 2018;69(8):2300-3.
-
Ţurcan N, Bohîlţea R, Ioniţă D. Preeclampsia prevention strategies. Pharmacokinetics and pharmacodynamics of low-molecular-weight heparin (LMWH) in pregnancy. Ginecologia.ro. 2020;8(28):18-22.
-
Alexander JM, McIntire DD, Leveno KJ. Selectiv magnesium sulfate prophylaxis for the prevention of eclampsia in women with gestational hypertension. Obstet Gynecol. 2006;108(4):826-32.
-
Vollaard E, Zeeman G, Alexander JA. “Delta eclampsia” – a hypertensive encephalopathy of pregnancy in “normotensive” women. Am J Obstet Gynecol. 2007;197(6):S140.
-
Hypertension in pregnancy. Report of the American College of Obstetricians and Gynecologist`s Task Force on Hypertension in Pregnancy. Obstet Gynecol. 122(5):1122-31.
-
Abdul-Karim R, Assali NS. Pressor response to angiotonin in pregnant and non-pregnant women. Am J Obstet Gynecol. 1961;82:246.
-
Raab W, Schroeder G, Wagner R. Vascular reactivity and electrolytes in normal and toxemic pregnancy. J Clin Endocrinol. 1956;16:1196.
-
Talledo OE, Chesley LG, Zuspan FP. Renin-angiotensin system in normal and toxemic pregnancies, 3. Differential sensitivity to angiotensin II and norepinephrine in toxemia of pregnancy. Am J Obstet Gynecol. 1968;100:218-221.
-
Gant NF, Chand S, Worley RJ. A clinical test useful for predicting the development of acute hypertension in pregnancy. Am J Obstet Gynecol. 1974;120(1):1-7.
-
Mandazli R, Budak E, Calay Z. Correlation between placental bed biopsy findings, vascular cell adhesion molecule and fibronectin levels in preeclampsia. BJOG. 2000;107(4):514-8.
-
Bouariu A, Popescu M, Iancu G. Current opinions on PlGF and sFlt-1 as reliable markers for preeclampsia. Ginecologia.ro. 2019;7(26):24-7.
-
Brăila AD, Marinov Krastev B, Mihai-Zamfir E. Uteroplacental apoplexy associated with invasive cervical neoplasm. RJME. 2017;58(4):1465-70.
-
Brăila AD, Gluhovschi A, Neacşu A. Placental abruption: etiopathogenic aspects, diagnostic and therapeutic implications. RJME. 2018;59(1):187-95.
-
Mihai-Zamfir E, Brăila AD, Brăila M. Convulsive syndrome in eclampsia and epilepsy in parturition under 30 years of age. RJMP. 2018;3(59):216-20.
-
Brăila AD, Brăila M, Velea R. The severe form of preeclampsia. Particular situations, clinical and therapeutic implications. Filodiritto Editore. The 6th National Congress of SRUOG. 2018:62-6.
-
Abramowicz JS, Sheiner E. Ultrasound of the placenta: A systemic approach. Part 2: function assessement (Doppler). Placenta. 2008;29(11):921-9.
-
Zeeman GG, McIntire DD, Twickler DM. Maternal and fetal artery Doppler findings in women with chronic hypertension who subsequently develop superimposed pre-eclampsia. J Matern Fetal Neonatal Med. 2003;14(5):318 -23.
-
Ghidini A, Locatelli A. Monitoring of fetal well-being: role of uterine artery Doppler. Semin Perinat. 2008;32(4):258-62.
-
Smith GCS, Yu CKH, Papageorghiou AT. Maternal uterine artery Doppler flow velocimetry and the risk of stillbirth. Obstet Gynecol. 2007;109(1):144-51.
-
Kingdom JC, Burrell SJ, Kaufmann P. Pathology and clinical implications of abnormal umbilical artery Doppler waveforms. Ultrasound Obstet Gynecol. 1997;9(4):271-86.
-
Morrow RJ, Abramson SL, Bull SB. Effect of placental embolization of the umbilical artery velocity waveform in fetal sheep. Am J Obstet Gynecol. 1989;161(4):1055-60.
-
Todros T, Sciarrone A, Piccoli E. Umbilical Doppler waveforms and placental villous angiogenesis in pregnancies complicated by fetal growth restriction. Obstet Gynecol. 1999;93(4):499-503.
-
Zelop CM, Richardson DK, Heffner LJ. Outcomes of severely abnormal umbilical artery Doppler velocimetry in structurally normal singleton foetuses. Obstet Gynecol. 1996;87(3):434-8.
-
ACOG Practice bulletin no. 134: fetal growth restriction. Obstet Gynecol. 2013;121(5):1122-33.
-
Berkley E, Chauhan S, Abuhamad A. Society for Maternal-Fetal Medicine Clinical Guideline: Doppler assessment of the fetus with intrauterine growth restriction, Am J Obstet Gynecol. 2012;206(4):300-8.
-
Bahado-Singh RO, Covanci E, Jeffres A. The developer cerebroplacental ratio and perinatal outcome in intrauterine growth restriction. Am J Obstet Gynecol. 1999;180(3 Pt 1):750-6.
-
Cruz-Martinez R, Figueras F, Hernandez-Andrade E. Fetal brain Doppler to predict caesarean delivery for nonreassuring fetal status in term small-for-gestational-age fetuses. Obstet Gynecol. 2011;117(3):618–26.





