Diagnose and treatment of the gastric cancer represent a challenge for the medical oncologists working in county hospitals. Facing the lack of diagnosed early stages, the absence of oncological surgeons with expertise and more important the lack of tumor board and multidisciplinary teams, medical oncologists try to offer the best possible treatment with the means at their disposal. This article reviews the last international guidelines for the treatment of gastric cancer and highlights the differences between gold standard of care and daily clinical practice in county hospitals.
Tratamentul cancerului gastric în spitalele judeţene, între standardul de aur şi practica clinică
Gastric cancer treatment in county hospitals: between gold standard and clinical practice
First published: 24 martie 2015
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/OnHe.30.1.2015.4308
Abstract
Rezumat
Diagnosticarea şi tratamentul cancerului gastric reprezintă o provocare pentru medicii oncologi care lucrează în spitalele judeţene. Confruntându-se cu lipsa diagnostocării precoce a bolii, absenţa chirurgilor oncologici experimentaţi şi mai important lipsa echipelor focusate pe tumoare şi cele multidisciplinare, medicii oncologi încearcă să ofere cel mai bun tratament posibil, cu mijloacele de pe care le au la îndemâna lor. Acest articol trece în revistă indicaţiile internaţionale pentru tratamentul cancerului gastric şi evidenţiază diferenţele dintre standardul de aur de îngrijire şi practica clinică de zi cu zi în spitalele judeţene.
Gastric cancer represents a major health problem, being the fourth most common related death cancer worldwide. Despite a global decrease in its incidence, there is a relative increase in incidence of tumors of the gastroesophageal junction (GEJ) and gastric cardia. As our database reveal, only in 2014, in the Dâmboviţa county there were registered a number of 87 new patients with gastric cancer (29 being registered at the moment they died!). From the rest of 56 cases, 41 were men and 17 women. Even if it seems not to be a big number of patients, unfortunately most of them presented with advanced disease (stage III - 21 cases, stage IV - 28 cases). The median age at diagnostic was 68 years both for men and women, indicating the peak incidence in the seventh decade.
Gastric cancer is difficult to cure and has a poor overall prognosis, most patients survive less than 1 year. Approximately two thirds of patients present with advanced disease, with the except of Japan and Korea, due to surveillance programs. More than 90% of gastric cancers are adenocarcinoma with 3 histological subtypes: intestinal, diffuse and mixt.
For patients with early-stage gastric cancer, surgery is the primary treatment. T1N0 non-ulcerated tumors benefit from endoscopic mucosal resection (EMR)(1). This practice is more frequent in Japan where the screening programs allow to diagnose early gastric cancers. Surgical resection with total or subtotal gastrectomy is performed depending on the site and extent of the tumor, complete resection with 4 cm or more adequate margins being considered as a standard goal. Extensive limphadenectomy (D2) - which removes also the retroperitoneal lymphnodes in addition to gastrectomy and perigastric lymphnodes (D1) - showed lower local and regional recurrence rates and lower gastric cancer related deaths compared to D1 resection(2), but its contribution to the survival benefit remains unclear. There is a general consensus that removal of 15 or more lymphnodes is beneficial for staging purposes and D1 resection is mandatory in order to improve overall survival. Anyway, D2 is recommend to be performed only in specialized, high-volume centers with appropriate expertise and postoperative care(1,3).
Globally, this gold standard of treatment is hard to be achieved in county hospitals, due to the lack of oncological surgeons, limitations of endoscopy tools, difficult access to investigations in real time and last, but not least, lack of the tumor boards. International guidelines recommend perioperative (preoperative and postoperative) chemotherapy as an alternative option for patients with resectable gastroesophageal cancer, based on the results of MAGIC trial(4), which revealed a significantly improvement both in PFS and in OS (5-year survival rates were 36% among patients who received perioperative chemotherapy and 23% in the surgery group). The chemotherapy delivered in the MAGIC trial was ECF (epirubicin, cisplatin, fluorouracil), which can be successfully replaced with ECX (epirubicin, cisplatin, capecitabine), with capecitabine avoiding the continuous infusion of fluorouracil. Other alternatives can be used, like EOX, with oxaliplatin replacing cisplatin, by extrapolating the results of the REAL-2 phase III trial(5), which compared ECF with ECX, EOF (epirubicin, oxaliplatin and fluorouracil) and EOX (epirubicin, oxaliplatin, capecitabine) in advanced gastric cancer. The results of this trial showed a small benefit for median OS in favor of EOX (11.2 months - compared to 9.9 months in the ECF and ECX arms and 9.3 months in the EOF arm), but PFS and response rate were the same in all arms. Again, in county hospitals, all the factors mentioned above limit the administration of this gold standard for resectable gastric cancers (preoperative chemotherapy, surgical resection, postoperative chemotherapy), for the medical oncologist being more frustrating as the chemotherapy schedules based on non-infusional fluorouracil are easy to deliver.
For patients with stage ≥T1N1 (IB) gastric cancer who have not received perioperative chemotherapy, ESMO and NCCN guidelines recommend adjuvant chemo-radiotherapy following R0 resection - no residual disease at surgical margins - for patients with T1-T2 node positive tumors, selected patients with T2N0 tumors and with high-risk features, patients with T3-T4 tumors, or postoperative chemotherapy following R0 resection and D2 lymph node dissection (for patients with T2N0, T1-T2 node positive tumors or patients with T3-T4 tumors). Those recommendations are very difficult to be achieved in county hospitals, first of all due to the lack of radiotherapy devices and secondly, because D2 resection is rarely performed in those hospitals. So again, gold standard remains somehow “mission impossible”.
It is to be noted that adjuvant chemotherapy following complete resection is indicated only for patients with D2 resection, based on the results of the CLASSIC trial(6) (conducted in several Asian countries). This trial evaluated postoperative chemotherapy with capecitabine and oxaliplatin in patients with stage II-III B gastric cancer; the interim analysis of the trial showed that postoperative chemotherapy improved DFS compare to surgery alone (the 3-year DFS rates were 74% versus 59%). For patients with D1 or D0 lymph node dissection the benefit of postoperative chemotherapy has not been documented in randomized clinical trials, so the use of adjuvant chemotherapy in this cases is outside clinical guidelines.
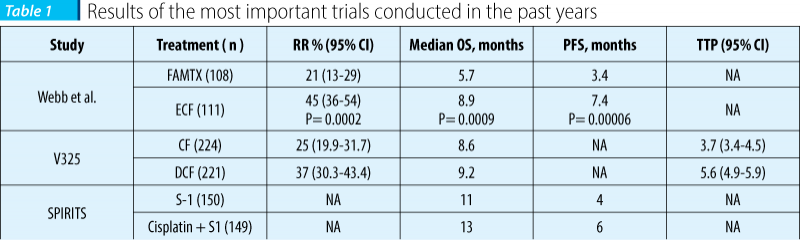
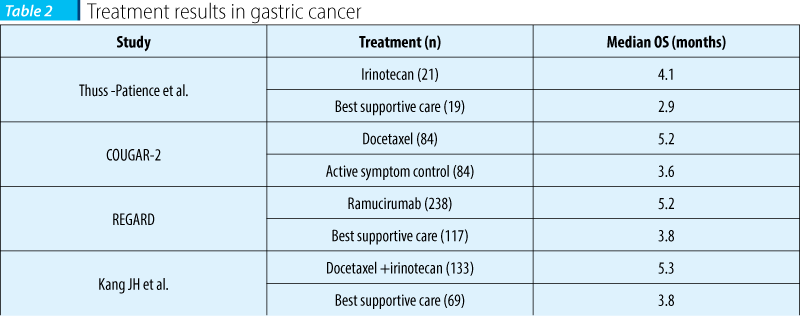
Patients with locally advanced or metastatic gastric cancer benefit from chemotherapy, which can provide symptoms palliation, improve survival and offer a better quality of life compared to best supportive care. There have been used various chemotherapeutical agents, with older agents (mitomycin, etoposide, cisplatin and fluorouracil) and newer agents (docetaxel, irinotecan, paclitaxel), everyone of them demonstrating a response rate as single agent: mitomycin C-30%, epirubicin - 19%, doxorubicin - 17%, cisplatin - 19%, fluorouracil - 21%, etoposide (oral) - 21%, UFT - 27%, capecitabine - 19%, paclitaxel - 17-23%, docetaxel - 17-29%, irinotecan - 18%, S1 (a pro drug) - 45%(7). Because monotherapy had an overall response rate of only 20%, beginning with the 80s, there have been conducted several trials using combination chemotherapies, which established the benefit of combination of chemotherapy versus monochemotherapy in terms of OS(8). The preferred chemotherapy regimens recommended by international guidelines for first-line chemotherapy are those based on platinum-fluoropyrimidine doublets, but findings from meta-analysis showed significant benefit when an antracycline is added to this doublet. Table 1 shows the results of the most important trials conducted in the past years, which established the ECF (epirubicin, cisplatin, fluorouracil) and DCF (docetaxel, cisplatin, flourouracil) as the regimens with best response in terms of median overall survival (8.9 months and 9.2 months respectively), with the mention that S1 remains an investigational agent and demonstrated statistically superior overall survival for patients with diffuse type gastric cancer (as a subset analysis of the FLAGS trial showed)(9,10,11,12). There is also to be noted that both DCF and ECF are regimens that require continuous administration of fluorouracil, a condition which make them difficult to apply in county hospitals, where the number of special chemotherapeutical devices is small. Fortunately, the results of the REAL 2 trial (which demonstrated the non-inferiority of using capecitabine and oxaliplatin instead of fluorouracil and cisplatin) offers a good alternative of treatment to ECF and permit to achieve the best therapy for the patient, with minimal adverse effects. At the same time, because DCF regimen is associated with increased mielosuppresion and infections, a recent clinical trial (phase II) found that DOF (docetaxel, oxaliplatin, fluorouracil) was associated with improved TTP, response rate and median overall survival (7.7 months, 47% and 15 months) compared to DO (docetaxel and oxaliplatin) - 4.5 months, 23% and 9 months - and DOX (docetaxel, oxaliplatin and capecitabine) - 5.6 months, 26% and 11 months. But again, fluorouracil is given by continuous infusion(13). Alternative to first-line chemotherapy are regimens with paclitaxel or with irinotecan, IF (irinotecan and fluorouracil) being an option for patients who are not fit for cisplatin-based chemotherapy. For patients who progress after first-line chemotherapy and have a good performance status, a second-line chemotherapy is recommended; options include irinotecan, docetaxel, paclitaxel and recently ramucirumab. Table 1 summaries the present data to support the use of second-line chemotherapy(14,15,16,17): with the exception of ramucirumab, all others agents are available in county hospitals, thus the standard of care for those patients is achieved. Of note, ramucirumab was recently approved by the FDA and EMA for second-line treatment as a single agent and also in combination with paclitaxel, based on the results of RAINBOW trial(18).
One last comment regarding the gold standard of gastric cancer in county hospitals is to be made: for patients with locally advanced, recurrent or metastatic HER-2 positive gastric cancer, international guidelines recommend the use of Trastuzumab in combination with chemotherapy (fluorouracil or capecitabine and cisplatin). As the results of the ToGA trial showed, adding Trastuzumab to chemotherapy for patients with HER-2 positive tumors improved median overall survival (13.8 months for Trastuzumab arm versus 11.1 months for the chemotherapy only arm), with a 26% benefit for overall survival (HR=0.74)(19). The only impediment in achieving the standard of care is the lack of Trastuzumab reimbursement.
There is much to be done in county hospitals in order to achieve the best treatment for patients with gastric cancer and to fill the gap between gold standard and daily clinical practice. A small number of medical oncologists working in county hospitals were asked to respond to following questions:
- Does a tumor board exist in your hospital?
- Did the patients with gastric cancer operated in your hospital benefit of D1/D2 resection?
- Are your patients with gastric cancer treated with chemotherapy regimens based on continuous infusion?
- Did you used perioperative chemotherapy for resectable gastric cancer?
The majority of medical oncologists recognized the lack of tumor boards in their hospital and if the board exists is not functional as it should. All of the them highlight that patients operated in their hospitals do not benefit from D1/D2 resection, but those operated in surgical excellence centers present removal of a minimum 15 lymphnodes. Only one medical oncologist use continuous infusion of fluoropyrimidine and none of the responders used perioperative chemotherapy, because all the resectable gastric patients were already operated, without the possibility of perioperative chemotherapy delivery.
In conclusion, there are some measures that have to be implemented in the future: the establishment of tumor boards, multidisciplinary teams, better and earlier diagnose of gastric cancer, acquisitions of medical devices and, last but not least, the access to new approved drugs.
Bibliografie
2. Songun et al. - 2010- Surgical treatment of gastric cancer:15 years follow-up results of the randomized nationwide Dutch D1D2 trial.; Lancet Oncol 11;439-449.
3. ESMO pocket guidelines - upper gastrointestinal cancers, 2014.
4. Cunningham et al. - 2006 - Perioperative chemotherapy versus surgery alone for resectable gastroesophageal cancer.; NEJM 355:11-20.
5. Cunningham et al. - 2008- Capecitabine and oxaliplatin for advanced esophagogastric cancer. NEJM 358:36-46.
6. Bang Y-J et al. - 2012 -Adjuvant capecitabine and oxaliplatin for gastric cancer after D2 gastrectomy (CLASSIC) - The Lancet 2012: 379,315-321.
7. Shah MA et al. - 2010 -J Natl Compr Canc Netw, 8; 437-447.
8. Wagner AD et al. -2006- JCO, 24;2903-2909.
9. Webb A, Cunningham D et al. -1997- Randomized trial comparing epirubicin, cisplatin and fluorouracil versus fluorouracil, doxorubicin and methotrexate in advanced esophagogastric cancer., JCO, 15: 261-267.
10. Van Cutsem E et al. -2006 - Phase III study of docetaxel and cisplatin plus fluorouracil compared with fluorouracil and cisplatin as first line for advanced gastric cancer. A report of the V325 Study group., JCO, 24:4991-4997.
11. Koizumi W et al. -2008- S-1 plus cisplatin versus S-1 alone for first line treatment of advanced gastric cancer (SPIRITS trial)- Lancet Oncol, 9:215-221.
12. Ajani JA et al. -2010- Multicenter phase III trial comparison of cisplatin/S-1 with cisplatin and infusional fluorouracil in advanced gastric or gastroesophageal adenocarcinoma study.: the FLAGS trial, JCO,28: 1547-1553.
13. Van Cutsen E et al. -2011- Randomized phase II study (GATE study) of docetaxel plus oxaliplatin with or without fluorouracil or capecitabine in metastatic or locally recurrent gastric cancer, JCO, 29 (15 suppl), abstract 4018.
14. Thuss- Patience PC et al. - 2009 - ASCO 2009, abstract 4540.
15. Cook N et al. - 2013 -COUGAR 02: Docetaxel versus BSC in relapsed esophagogastric adenocarcinoma -ASCO 2013, abstract 4023.
16. Fuchs CS et al. - 2014 - Ramucirumab monotherapy for previously treated advanced gastric or gastro-esophageal junction adenocarcinoma (REGARD) - Lancet 2014, 383: 31-3.
17. Kang JH et al. - 2012- Best supportive care plus salvage chemotherapy versus best supportive care alone in gastric cancer, a phase III Korean study, JCO 30: 1513-1518.
18. Wilke H et al. - 2014- Ramucirumab plus paclitaxel versus placebo plus paclitaxel in patients previously treated advanced gastric or gastroesophageal junction adenocarcinoma (RAINBOW), Lancet Oncol 2014, 15: 1224-1235.
19. Bang YJ, Van Cutsem E et al. -2010 - Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2 - positive advanced gastric or gastroesophageal junction cancer ( ToGA), Lancet 2010, 376:687- 697.
Articole din ediţiile anterioare
Asocierea dintre inhibitorii de tirozin-kinază şi chimioterapie în cancerul pulmonar fără celule mici
Această recenzie încearcă să analizeze câteva studii şi metaanalize ale inhibitorilor de tirozin-kinază (TKI) în combinaţie cu chimioterapia. Une ...
Regimul FOLFIRINOX în tratamentul adjuvant şi metastatic al cancerului pancreatic în era oncologiei de precizie
Cancerul pancreatic rămâne o boală agresivă, cu un prognostic nefavorabil, deşi s-au făcut progrese în ultimii ani în ceea ce priveşte diagnostic...
Preliminary results of markers as predictors for the tissue response to radiochemotherapy in rectal cancer
Radiochimioterapia preoperatorie (p-CRT) este o tehnică utilizată pe scară largă pentru tratarea cancerului rectal local avansat, a cancerului esof...
Cancerul pancreatic
Cancerul pancreatic este pe locul al doisprezecelea ca incidenţă în lume. Acest cancer are o mortalitate ridicată, iar supravieţuirea globală este ...