Endocardita micotică neonatală
Neonatal fungal endocarditis
Abstract
Cardiac involvement in disseminated fungal infection is a rare condition among neonates with an increasing occurrence rate over the past years. Candida species represent the most common pathogen (66%) of fungal endocarditis for this class of patients. The disease is usually found in infants with an underlying immunodeficiency such as premature infants who receive corticoid therapy and neonate patients with preexisting cardiac pathologies. Fungal endocarditis has a high mortality rate due to the diagnostic difficulties represented by non-specific clinical manifestations, a low incidence of positive blood cultures, limited use of antifungal drugs and restrictions of surgical management. The purpose of this article is to evaluate the acquirements regarding this pathology that are stated in literature and to present the therapeutic options available at this point.Keywords
neonatetherapeutic optionsmortalityCandidaRezumat
Implicarea cardiacă în infecţiile micotice diseminate este o condiţie rară printre nou-născuţi, cu o rată a incidenţei în creştere în ultimii ani. Speciile de Candida sunt cel mai frecvent patogen responsabil de endocardită micotică la această grupă de pacienţi (66%). Boala este de obicei întâlnită la pacienţi cu imunodeficienţă, cum ar fi nou-născuţi prematuri care primesc terapie corticoidă sau nou-născuţi cu o patologie cardiacă preexistentă. Endocardita micotică are o rată de mortalitate ridicată, din cauza dificultăţilor diagnostice reprezentate de manifestările clinice nespecifice, a incidenţei scăzute a hemoculturilor pozitive, a utilizării limitate de medicamente anifungice la această grupă de pacienţi şi a restricţiilor de management chirurgical. Scopul articolului este de a evalua progresele menţionate în literatură în domeniul acestei patologii şi de a prezenta opţiunile terapeutice disponibile în prezent.Cuvinte Cheie
nou-născutopţiuni terapeuticemortalitateCandidaIntroduction
Fungal endocarditis is a rare infection among neonates, with an average occurrence rate of 1.1% of all infectious endocarditis cases in children.
Even though in the past years the prognosis has improved, the mortality rate is 75-90%, and the morbidity rate also remains high, with diagnosis in the majority of cases made postmortem(1,2). Fungal endocarditis occurs in critically ill patients treated in intensive care units and often appears as a result of systemic fungal infection. In two thirds of the cases, the pathogens involved are Candida species(1,3,4).
Candidiasis
In the last years, systemic candidiasis has had a significant increase in neonatal intensive care patients. Candida is responsible for 2.6% to 16.7% of late-onset sepsis among infants with very low birth weight (weight ≤1500 g) and up to 20% for infants with extremely low birth weight (weight ≤1000 g)(4,5).
The most often involved species in neonate pathology are C. albicans, followed by C. parapsilosis, in comparison with adults in whom the predominant isolated species are C. albicans, C. parapsilosis and C. glabrata(4,6). Colonization with Candida is mostly acquired by vertical transmission from the maternal vaginal mucosa during birth, therefore the existence of Candida vaginitis is an important risk factor for an increased colonization. Rarely, the infection can ascend from the vagina and transplacental transmission is possible. Also, candidal mastitis results in transmission of yeast from mother’s skin to the infant’s oral cavity. Infections with Candida species are often nosocomial-acquired, especially referring to C. parapsilosis, that can be carried by the hands of the health care providers(4).
The penetration of skin or mucosal epithelium and hematogenous spread to internal organs depend on the yeast virulence factors and certain host risk factor as follows: prematurity, very low/extremely low birth weight, Apgar score <5 at 5 minutes, prolonged broad-spectrum antimicrobial therapy (particularly with third generation cephalosporin), central intravascular catheters, total parenteral hyperalimentation >5 days, intravenous lipid emulsion >7 days, intubation, exposure to H2 blockers, abdominal and cardiac surgery, spontaneous intestinal perforation, neutropenia, hyperglycemia, steroid therapy, prolonged hospitalization >7 days(3,4,7).
The clinical manifestations of candidiasis can vary depending whether the disease is disseminated or not, and complications can be extensive. Infants often present sepsis features like lethargy, feeding intolerance, apnea and respiratory distress, cardiovascular instability and hyperbilirubinemia. Fever occurs rarely, even in critical cases(4).
Regarding dissemination, histological examinations found microabscesses in the kidney, retina, brain, liver, spleen, peritoneum, heart, lungs and even joints(4). The histopathology of the cardiac valves reveals fungal colonies that form vegetations variable in size (Figures 1 and 2).
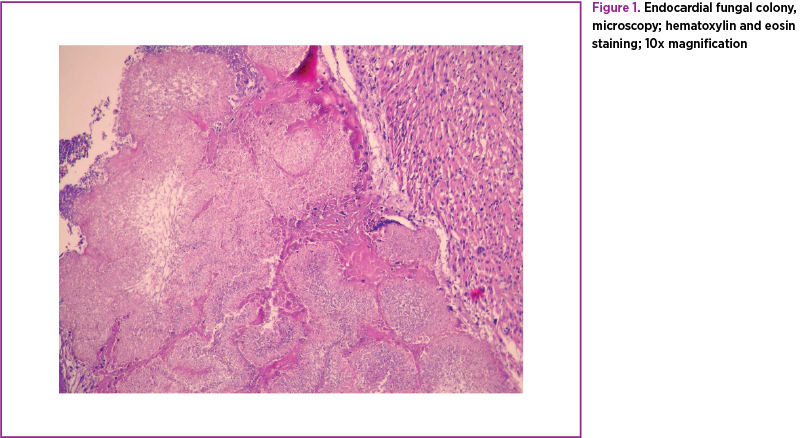
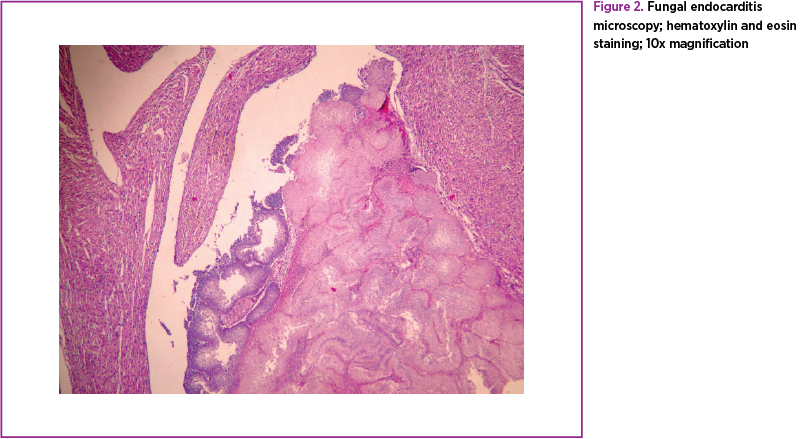
Endocarditis
Cardiac involvement has been observed in approximately 15.2% of neonates with candidemia. Fungal endocarditis rarely affects native valves. Several studies have stated that it usually occurs under the form of a right-sided intracardiac thrombus, in the presence of a valvular injury, due to the existence of a central venous catheter. Candida species are commensal organisms that colonize the skin and mucosal surfaces (mostly gastrointestinal) and adhere to catheter surfaces(1,6).
Fungal endocarditis may also appear as a complication of cardiac surgery or a systemic fungal infection in the high-risk group of patients, previously mentioned. The risk for endocarditis is significantly risen by the use of broad-spectrum antibiotics, parenteral hyperalimentation, congenital heart disease or any condition associated with immunodeficiency(1,3).
Physical examination often reveals non-specific features like bradycardia, hypotension and acute respiratory distress requiring intubation. Occasionally, fungal endocarditis presents with fever, weight loss, splenomegaly, splinter hemorrhages, petechiae, typical for bacterial endocarditis, although dermatologic lesions like Roth spots, Osler nodes and Janeway spots occur in less than 5% of the cases(1,3,8). Only rarely a new heart murmur is present or a change in a preexisting heart murmur can be detected(1). Dysphagia, hoarseness or even superior vena cava syndrome may be the unique manifestation of the disease.
Laboratory findings
According to some authors, positive blood cultures in fungal endocarditis are obtained in only 25% of cases and more than 50% of Candida endocarditis cases have negative blood cultures(1,9). However, in fungal endocarditis, if found positive, blood cultures may persist this way despite therapy, especially with Candida infection(1). A blood culture should be routinely performed, particularly in premature infants, low birth weight infants and those with high risk(9). When infection is suspected, blood culture must be taken and repeated in 48 hours(5). When blood cultures are positive for more than 5 days, persistent candidemia occurs and the risk for endocarditis highly increases, but not for other complications(3).
When blood cultures do not provide enough help for diagnosis, culture of sputum, urine, cerebrospinal or synovial fluid, bone marrow or even lymph node can become the only evidence of disseminated fungal infection(1). Urinalysis demonstrate hematuria or/and proteinuria and some authors sustain that a routine microscopic urinary examination for Candida is strongly recommended in premature infants(1,9).
A complete blood count can reveal leukocytosis, but not necessarily, with a left shift and thrombocytopenia is often seen in fungal infections of the neonates. An elevated C-reactive protein may also be found. In many cases, laboratory findings are difficult to distinguish from bacterial sepsis, but persistent thrombocytopenia, hypoglycemia and hypotension must be seen with concern(1,5).
There are some tests available for Candida determination, such as anti-Candida antibody, beta-glucan antibody, d-arabinitol concentration and PCR, but they are not very popular in common practice(5).
Imaging findings
If a chest radiography is performed, it can reveal fluid in the pleural cavity, pulmonary infiltrates or cardiomegaly(1).
When a transesophageal echocardiography can be performed, the chance of revealing a cardiac thrombus or a valvular vegetation is higher than through a transthoracic echocardiography. Fungal endocarditis may also present with pericardial effusion or associated myocarditis or pericarditis. Abscesses of the myocardium may also be found using ultrasonography(1).
A cardiac catheterization can be performed in order to reveal vegetation, thrombi or underlying cardiac lesions, but is not widely used because of the frequency of severe complications such as vascular disruption, hemorrhage, vascular occlusion, bradyarrhythmia or tachyarrhythmia(1).
Treatment
The treatment of fungal endocarditis consists of aggressive antifungal therapy, but may not always be enough, so the removal of infected nidus plays a major role in managing these cases(1).
Despite its toxicity, liposomal amphotericin B is usually the first line of therapy due to the fact that it is the most studied drug. Flucytosine is used together with amphotericin B and has a synergistic effect in severe fungal infections. There is insufficient data about fluconazole’s efficiency, so it remains a second-line drug(1,3,5).
There is limited data in literature about the use of Caspofungin, Variconazole, Micafungin, Anidulafungin or Posaconazole for the treatment of fungal endocarditis, so the use of these drugs is not largely accepted(3,5).
There is not a general agreement on the duration of antifungal therapy, but in common practice the drugs are administered for 2-6 weeks, depending on the severity and dissemination of the disease, but also on the occurrence of the side effects of the antifungal drugs, like nephrotoxicity, hepatotoxicity and electrolyte embalances(5).
In some articles, it is stated that infants with extremely low birth weight could benefit from prophylaxis with intravenous fluconazole for a period of 2-6 weeks, starting with the first day of life(5).
Right atrial mycetoma has been reported in 9% of the cases and the management has to include prolonged antifungal therapy along with a surgical procedure. Depending on its dimensions and location, the mycetoma may determine hemodynamic compromise and increases the risk of septic emboli, therefore, when possible, removal of thrombus, valve replacement or abscess resection should be done. Surgery is strongly indicated if there is an ongoing infection with poor response to medical therapy, complicated by embolic phenomena or cardiac failure(1,3). The use of prolonged antifungal treatment alone is an approach made for some critically ill neonates, when there are few alternatives left. If surgical thrombectomy cannot be taken into consideration, thrombolytic therapy with urokinase in addition to antifungal drugs proved to be a satisfactory alternative, mostly for preterm neonates(1,10).
Medical therapy should also include removal of the central venous catheter in the presence of persistent fungemia(1,3,5). Furthermore, the decrease of immune suppression and providing inotrope support might bring additional improvement of the patient’s condition(1).
Conclusions
Neonatal fungal endocarditis is a rare condition with an increasing incidence because of the increasing number of disseminated fungal infections. The most frequent pathogen in neonates is Candida albicans, and it usually affects patients with different underlying cardiac anomalies, or with a poor general condition because of prematurity, low/extremely low birth weight, immunosuppression of different causes and use of prolonged broad-spectrum antimicrobial therapy.
The frequency and severity of Candida infection is inversely associated with the gestational age and birth weight of preterm infants.
The diagnostic is difficult because of the non-specific manifestations, rare typical dermatological findings, and a low occurrence of positive blood cultures.
The treatment consists in antifungal therapy, but other than amphotericin B there are few studies to prove the efficiency of antifungal drugs in the treatment of endocarditis. Even in association with surgical intervention or thrombolytic therapy, the outcomes are poor and mortality and morbidity remain high for this disease.
Bibliografie
1. Sandy N Shah. Pediatric Fungal Endocarditis, Medscape, Mar 09, 2015.
2. Millar BC, Jugo J, Moore JE. Fungal endocarditis in neonates and children, Pediatr Cardiol. 2005 Sep-Oct; 26(5):517-36.
3. J Sharma, A Nagraj, D Allapathi, B Rajegowda, and R Leggiadro. Fungal endocarditis in a premature infant complicated by a right atrial mycetoma and inferior vena cava thrombosis. Images Paediatr Cardiol. 2009 Oct-Dec; 11(4): 6–11.
4. Remington and Klein, Wilson, Nizet, Maldonado. Infectious diseases of the Fetus and Newborn Infant, 7th edition, chapter 33, pages 1055-1073.
5. Abdurrahman A. Ozdemir, Tugce K. and Aydın Varol. Fungal endocarditis in an extremely low birth weight infant. Case report, Arch Argent Pediatr 2016; 114(2): e117-e120/e117.
6. David A Kaufman. Fungal Infections in Preterm Infants, Medscape Jun 25, 2014.
7. Sabah Kalyoussef, DO. Pediatric Candidiasis Clinical Presentation; Medscape Feb 07, 2016.
8. CO Callaghan and P. McDouglas. Infective endocarditis in neonates. Royal Children's Hospital, Melbourne, Australia; Archives of Disease in Childhood, 1988, 63, 53-57.
9. Emilio Mayayo, Jose Moralejo, Juan Camps, and Josep Guarro. Fungal Endocarditis in Premature Infants: Case Report and Review, From the Hospital Universitari de Tarragona "Joan XXIII" and Unitat de Microbiologia, Facultat de Medicina, Universitat Rovira i Virgili, Tarragona, Spain.
10. PA Ankola, S Perveen and B Fish. Fungal endocarditis, Journal of Perinatology (2006) 26, 509–510. doi:10.1038/sj.jp.721.
Bolile rare și testarea genetică, în centrul unor proiecte naționale
Cristina Ghioca
UMFCD și Institutul de Cercetare-Dezvoltare în Genomică au lansat proiectele „ExpertRARE” și „Genetică Echitabilă”. Acestea vizează, între altele, oferirea de servicii de screening genetic pentru aproape 20.000 de persoane.
...Bolile rare și testarea genetică, în centrul unor proiecte naționale
Cristina Ghioca
UMFCD și Institutul de Cercetare-Dezvoltare în Genomică au lansat proiectele „ExpertRARE” și „Genetică Echitabilă”. Acestea vizează, între altele, oferirea de servicii de screening genetic pentru aproape 20.000 de persoane.
...

