The treatment of cervical intraepithelial neoplasia aims at removing the junction area, including abnormal tissues. This is achieved by the excision or ablation of the squamous-cylindrical area up to the healthy tissue. In the case of persistence of high-grade lesions after repeated excision and if the patient does not want to preserve fertility, hysterectomy may be indicated. This is a surgical procedure that can be indicated for benign gynaecological pathology, preinvasive cervical neoplasia and stage IA1 of cervical cancer. The objective of the study was to select patients regarding the management of cervical intraepithelial neoplasia, taking into account the grade of cervical lesion, the extension area, patients’ age and maternal desire, as well as the pathology associated with cervical lesions. Conization was the surgical choice in patients with cervical intraepithelial neoplasia, under 35 years of age, who wanted to conserve fertility and did not have other gynaecological lesions. Hysterectomy was the surgical treatment in patients over 35 years of age, with finished pregnancy planning and associated uterine pathology to intraepithelial cervical neoplasia.
Corelaţii clinice şi paraclinice în managementul neoplaziei intraepiteliale cervicale
Clinical and paraclinical correlations in the management of cervical intraepithelial neoplasia
First published: 08 martie 2019
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/Gine.23.1.2019.2220
Abstract
Rezumat
Tratamentul neoplaziei intraepiteliale cervicale vizează eliminarea zonei de joncţiune, inclusiv a ţesuturilor anormale. Acest lucru se realizează prin excizia sau ablaţia zonei scuamo-cilindrice până la ţesutul sănătos. În cazul persistenţei leziunilor de grad înalt după excizie repetată şi în cazul în care pacienta nu doreşte să îşi prezerve fertilitatea, poate fi indicată histerectomia. Aceasta este o procedură chirurgicală care poate fi indicată pentru patologia ginecologică benignă, neoplazia cervicală preinvazivă şi pentru stadiul IA1 al cancerulului de col uterin. Obiectivul studiului a fost selectarea pacientelor în funcţie de managementul neoplaziei intraepiteliale cervicale, luând în considerare gradul leziunii cervicale, zona de extensie, vârsta pacientelor şi dorinţa de menţinere a fertilităţii, precum şi patologia asociată cu leziunile cervicale. Conizaţia a fost procedura chirurgicală selectată la pacientele cu neoplazie intraepitelială cervicală, sub 35 de ani, care au dorit să-şi prezerve fertilitatea şi nu au avut alte leziuni ginecologice. Histerectomia a fost tratamentul chirurgical la pacientele cu vârsta de peste 35 de ani, fără dorinţă de procreare şi patologie uterină asociată cu neoplazia cervicală intraepitelială.
Introduction
The treatment of cervical intraepithelial neoplasia aims at removing the junction area, including abnormal tissues. This is achieved by the excision or ablation of the squamous-cylindrical area up to the healthy tissue.
Ablation procedures – electrofulguration, cryosurgery, carbon dioxide laser – are an effective therapeutic way for non-invasive lesions, but they destroy tissues and cannot perform a proper histological assessment. It is absolutely necessary to exclude the possibility of neoplasia by cytological, colposcopic and histological examinations before applying ablative procedures.
Excision is a surgical procedure that allows the complete histological evaluation of the piece, having both diagnostic and therapeutic character. It is indicated for patients with suspected invasive cancer lesions and endocervical adenocarcinoma, high-grade lesion in cytology, unsatisfactory colposcopy and cervical intraepithelial neoplasia (CIN) diagnosed at the fragmentary biopsy, requiring treatment, and for patients with recurrent atypical glandular cell cytology (AGC) of recurrent high-grade CIN. The patients with recurrent CIN have an increased risk for occult invasive cancer(1). The excision procedures – with electric loop LEEP, conization – increase the risk of complications, affecting the reproductive capacity: cervical canal stenosis, cervico-ischemic incompetence, premature birth, premature rupture of membranes(2). The patients with an interval of less than 2-3 months between conization and pregnancy are at risk of premature birth(3). The association with premature birth is uncertain, a number of studies demonstrating an increase in risk, while other studies did not support this hypothesis(4-8). On this basis, the treatment is preferred for the eradication of high-grade injuries.
In the case of persistence of high-grade lesions after repeated excisions and if the patient does not want to preserve fertility, hysterectomy may be indicated. Simple hysterectomy involves the removal of the uterus and cervix, but does not involve the excision of parameters and paracolpos. It is a surgical procedure that can be indicated for benign gynaecological pathology, preinvasive cervical neoplasia and stage IA1 of cervical cancer.
In CIN 1, which occurs especially in teenagers, CIN 2 or CIN 2/3, the treatment is applied if the lesion persists for at least two years in teenagers and young women. CIN 2 in adult women and CIN 3 in women irrespective of age require the excision or ablation(2,9-11).
Adenocarcinoma in situ is diagnosed at younger ages(12). The therapeutic objective is to excise all affected tissues after eliminating the diagnosis of invasiveness. It is indicated the cold scalpel conization to keep the edges and to facilitate the histological interpretation. The risk of residual adenocarcinoma is approximately 80%, and it is indicated the repetition of the conization(10). Hysterectomy is indicated after the maternity is ended(12,13).
The objective of the study was to select patients for the management of cervical intraepithelial neoplasia, taking into account the grade of cervical lesion, the surface extension, the patient’s age and maternity desire, as well as the pathology associated with cervical lesions.
Method
The study was conducted on a total of 44 patients, between January 2017 and May 2018. The patients were aged between 21 and 50 years old, and they were divided into two groups: 21-35 years old and 36-50 years old.
Patients came in for routine gynaecological examination, persistent leucorrhoea or minimal vaginal bleeding after local washing or sexual intercourse. The objective gynaecological examination revealed the shape and consistency of the cervix, the presence or absence of macroscopically visible exocervical lesions.
Investigations such as cervico-vaginal cytology, HPV testing and genotyping, colposcopy and fragmentary biopsy or conization were performed to determine the extent of cervical lesions.
Detailed anamnesis, gynaecological clinical examination, biopsy curettage and ultrasound revealed in 17 patients uterine-associated lesions such as uterine fibromatosis and endometrial hyperplasia.
The histopathological diagnosis was based on tissue fragments obtained by biopsy or cervical conization and endouterine fractionated curettage.
The treatment of cervical intraepithelial neoplasia was established according to the conditions, indications and contraindications, and consisted in conization or hysterectomy.
Results
From the distribution analysis by age group, we found that the incidence of cervical intraepithelial neoplasia was higher in the 36-50 age group (25 cases; 57%) compared to the 21-35 age group (19 cases; 43%) – Table 1.
The results of the cervicovaginal cytology test were positive for epithelial cell abnormalities in the Bethesda classification, such as atypia of undetermined squamous cells (ASC-US) in 8 cases (18%), atypia of squamous cells with a high-grade lesion (ASC-H) in 5 cases (11%), low-grade squamous intraepithelial lesions (L-SIL) in 10 cases (23%), high-grade squamous intraepithelial lesions (H-SIL) in 17 cases (39%), atypical glandular cells of undetermined significance (AG-US) in one case (2%), and cells suggestive of squamous carcinoma in three cases (7%) – Table 2 and Figure 1.
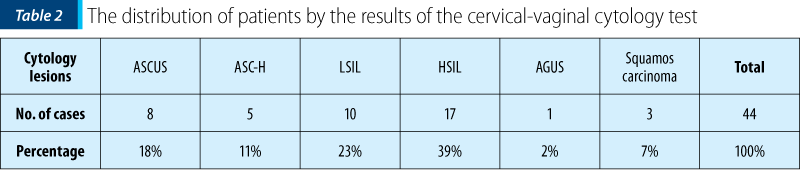
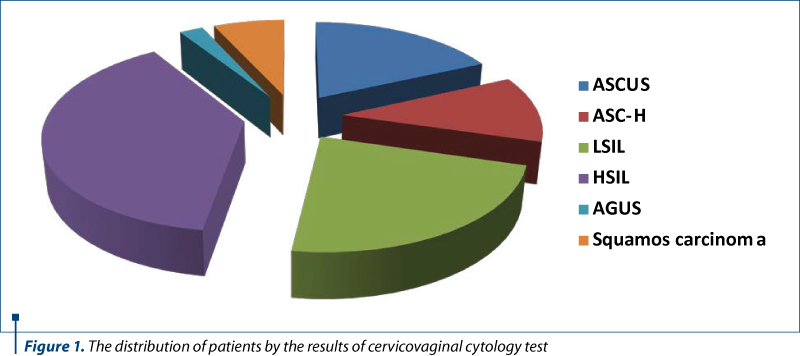
The colposcopic examination could not exclude a high-grade lesion or invasive cancer in 10 patients (23%).
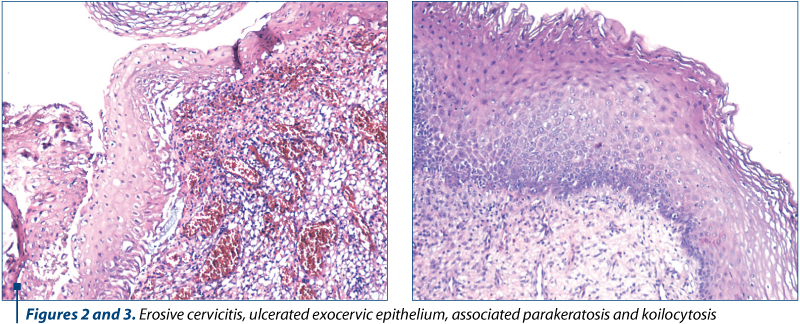
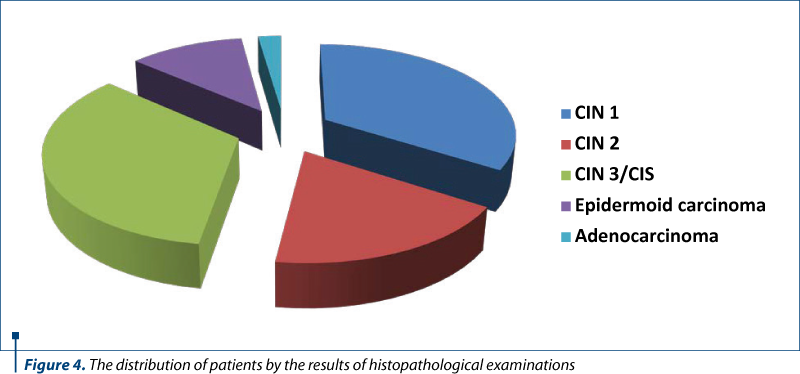
The results of the histopathological examinations of biopsy and conization pieces were: 15 cases (34%) of CIN 1, including HPV infection lesions (Figure 2); eight cases (18%) of CIN 2; 15 cases (34%) of CIN 3/CIS; five cases (12%) of invasive epidermoid carcinoma and one case (2%) of adenocarcinoma (Table 3 and Figure 3).
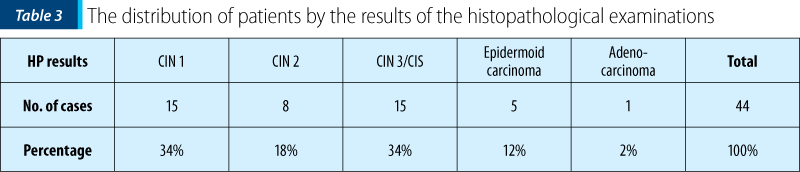
The 17 patients (39%) diagnosed with uterine fibromatosis or endometrial hyperplasia associated with cervical lesions, except for two cases of CIN 1, were over 40 years of age and had finished pregnancy planning. The cervical lesions in this group of patients were low-grade CIN (two cases), high grade CIN (15 cases – three cases with CIN 2 and 12 cases with CIN 3). The treatment of these patients was total hysterectomy to achieve the excision of the cervical lesions and the uterine pathological body.
The adenocarcinoma patient and the five epidermoid carcinoma patients also benefited from radical treatment – total hysterectomy with bilateral adnexectomy and lymphadenectomy (6 cases; 13%). Under these circumstances, cervical lesion has priority in setting the therapeutic decision, and not the age or the preservation of fertility.
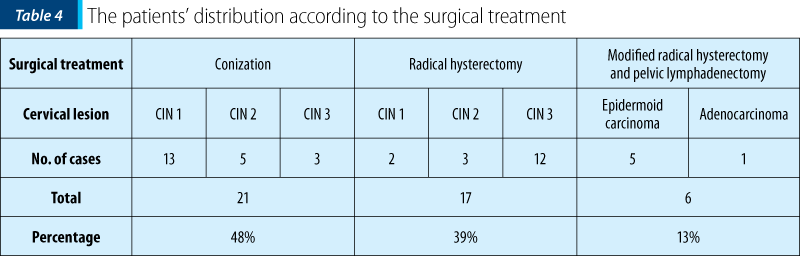
The surgical treatment in 13 patients with CIN 1, five patients with CIN 2 and three patients with CIN 3 consisted in conization (21 cases; 48%). These patients were predominantly under the age of 35 or wanted to maintain fertility, so they had conservative treatment.
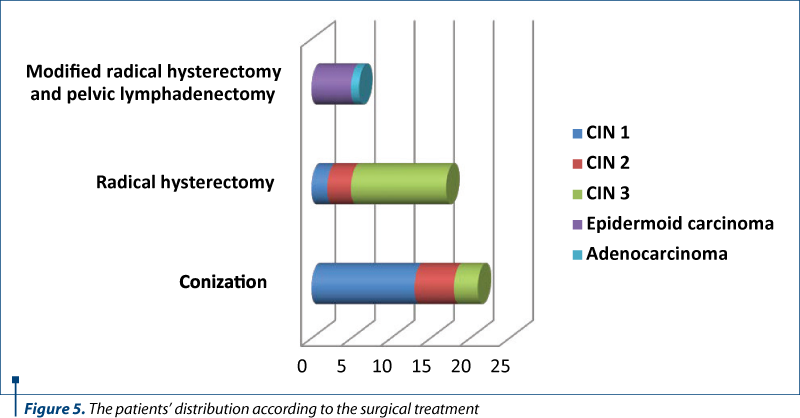
The surgical treatment was conservative in 21 patients (48%) – conization; radical in 17 patients (39%) – total hysterectomy, and in 6 cases (13%) it was performed total hysterectomy with bilateral adnexectomy and lymphadenectomy (Table 4 and Figure 4).
Discussion
The surgical treatment in patients younger than 35 years old who wanted to conserve fertility benefited from conization, both as a biopsy method and as a treatment, although conization increases the risk of adverse effects on the reproductive function. The histopathological result of the conization parts revealed cervical lesions, such as CIN 1, CIN 2 and CIN 3. Post-surgical monitoring is required. Patients with CIN and negative resection margins are monitored by cytology examination once every 6 months until two consecutive negative results are obtained and then routinely supervised; one can test the presence of HPV DNA between 6 and 12 months after the treatment followed by colposcopy if HPV DNA is detected, because it is a marker of the persistence of the disease(14-16). The cytological screening is recommended to be performed for at least 20 years because there is an increased risk of cervical neoplasia after the diagnosis of high-grade CIN. If the excision margins are positive for CIN 2 or CIN 3, cytology and biopsy are recommended between 4 and 6 months(17).
Although hysterectomy is an unacceptable procedure for CIN 1, 2 and 3(18) lesions, the surgical treatment of patients over 35 years of age who had uterine polifibromatosis, endometrial hyperplasia and who had completed pregnancy planning after making biopsies and establishing the diagnosis benefited from hysterectomy. The histopathological result of surgical parts after hysterectomy revealed uterine leiomyofibromatosis, adenomyosis, simple endometrial hyperplasia with or without atypia associated with CIN 1, CIN 2, CIN 3/CIS type lesions (Figure 5).
Although hysterectomy provides the lowest disease recurrence, in the case of a CIN hysterectomy with negative resection margins, hysterectomy is not completely protective for recurrences. Under these circumstances, there is a risk for a local recurrence, and for this reason monitoring by cytological screening of the vagina is required(19).
Conclusions
The decision on the management of cervical intraepithelial neoplasia took into account the degree of cervical lesion, the extent to the surface, the age of the patients, and the desire for maternity, as well as the pathology associated with the cervical lesions.
Conization was the surgical choice in patients with cervical intraepithelial neoplasia, under 35 years of age, who wanted to conserve fertility and did not have other gynaecological injuries.
Hysterectomy was the surgical treatment in patients over 35 years of age, with finished pregnancy planning and uterine pathology associated to intraepithelial cervical neoplasia.
Conflict of interests: The authors declare no conflict of interests.
Bibliografie
- Paraskevaidis E, Jandial L, Mann E. Pattern of treatment failure following laser for cervical intraepithelial neoplasia: implications for follow-up protocol. Obstet Gynecol. 1991; 78, 80.
- Wright TC Jr, Massad S, Dunton CJ. 2006 consensus guidelines for the management of women with cervical intraepithelial neoplasia or adenocarcinoma in situ. Am J Obstet Gynecol. 2007c; 197(4), 340.
- Himes KP, Simhan HN. Time from cervical conization to pregnancy and preterm birth. Obstet Gynecol. 2007; 109(2Pt1), 314.
- Jakobsson M, Gissler M, Paavonen J. Loop electrosurgical excision procedure and the risk for preterm birth. Obstet Gynecol. 2009; 114, 504.
- Kyrgiou M, Koliopoulos G, Martin-Hirsch P. Obstetrics outcomes after conservative treatment for intraepithelial or early invasive cervical lesions: systematic review and meta-analysis. Lancet. 2006; 367, 489.
- Sadler L, Saftlas A, Wang W. Treatment for cervical intraepithelial neoplasia and risk for preterm delivery. JAMA. 2004; 291, 2100.
- Samson SL, Bentley JR, Fahey TJ. The effect of loop electrosurgical excision procedure on future pregnancy outcome. Obstet Gynecol. 2005; 105, 325.
- Werner CL, Lo JY, Heffernan S. Loop electrosurgical excision procedure and risk of preterm birth. Obstet Gynecol. 2010; 115, 605.
- American College of Obstetricians and Gynecologists. Cervical cancer in adolescents: screening, evaluation and management. Committee opinion No 463, August 2010a.
- Erdelean I, Anastasiu DA, Grigoraş D, Sas I, Dema A, Anastasiu D. Correlation between preoperative diagnosis and histopathological changes in preneoplastic cervical lesions. Gineco.eu. 2015; 11(1), 22-3.
- Dragosloveanu C, Vlădăreanu R. “See and treat” strategy for HSIL: correlation between colposcopy and histopathology. Gineco.eu. 2014; 10(2), 66-9.
- Krivak TC, Rose GS, McBroom JW. Cervical adenocarcinoma in situ: a systematic review of therapeutic options and predictors of persistent or recurrent disease. Obstet Gynecol Surv. 2001; 56(9), 567-75.
- Poynor EA, Barakat RR, Hoskins WJ. Management and follow-up of patients with adenocarcinoma in situ of the uterine cervix. Gynecol Oncol. 1995; 57(2), 158-64.
- Wright TC Jr, Massad S, Dunton CJ. 2006 consensus guidelines for the management of women with abnormal cervical cancer screening tests. Am J Obstet Gynecol. 2007b; 197(4), 346.
- Saad H, Brăila AD, Velea R, Brăila M, Lungulescu C. The importance of primary screening in early detection of cervical dysplastic lesions. Gineco.eu. 2018; 14(2), 62-6.
- Bălulescu I, Badea M. Interobserver variability in the interpretation of cervical smears, a must for developing an internal laboratory quality control system. Gineco.eu. 2013; 9(4), 178-83.
- American College of Obstetricians and Gynecologists. Cervical cytology screening. Practice Bulletin. December 2009, No. 109.
- Wright TC Jr. Cervical cancer screening in the 21st century: is it time to retire the Pap smear? Clin Obstet Gynecol. 2007a; 50(2), 313.
- Saslow D, Runowicz CD, Solomon D. American Cancer Society guideline for the early detection of cervical neoplasia and cancer. CA Cancer J Clin. 2002; 52(6), 342-62.