Pulmonary embolism is one of the emergency situations that require a lot of preparation and a good clinical sense to avoid those scenarios in which this pathology is overdiagnosed, with an increased consumption of financial resources and an unjustified exposure of the patient to radiation, or omitted, with the occurrence of complications or even death. In order to facilitate the diagnosis algorithm of the pulmonary embolism, several management guidelines have been proposed, without being able to establish with certainty which is the suitable moment when the laboratory analyses or the imaging investigations should be used for the correct diagnosis of the embolism. As pulmonary embolism is one of the leading causes of mortality among pregnant women in developed countries, with a mortality of approximately 15-30%, the correct and rapid diagnosis of this pathology is a priority. Although there are several laboratory investigations that can guide the clinician to the diagnosis of pulmonary embolism, many of them must be interpreted in the context of the modified biological profile encountered during pregnancy. Also, the frequent need to use imaging investigations should be carefully evaluated to reduce the risks of radiation exposure of the foetus and breast tissue, or the administration of the contrast agent. However, the correct diagnosis and treatment of pulmonary embolism should not be delayed.
Diagnosticarea şi managementul emboliei pulmonare în rândul pacientelor gravide
Diagnosis and management of pulmonary embolism among pregnant patients
First published: 20 septembrie 2019
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/Gine.25.3.2019.2494
Abstract
Rezumat
Embolia pulmonară este una dintre situaţiile de urgenţă care necesită multă pregătire şi un simţ clinic foarte bine dezvoltat pentru a evita scenariile în care această patologie este supradiagnosticată, cu un consum crescut de resurse financiare şi expunerea nejustificată a pacientului la radiaţii, sau omisă, cu apariţia complicaţiilor sau chiar a decesului. Pentru a uşura algoritmul de diagnostic al emboliei pulmonare, s-a încercat introducerea mai multor ghiduri de conduită terapeutică, fără a reuşi să se stabilească cu certitudine care este momentul propice în care trebuie să se utilizeze analizele de laborator sau investigaţiile imagistice pentru diagnosticarea corectă a emboliei. Deoarece embolia pulmonară este una dintre cauzele principale de mortalitåşşate în rândul gravidelor din ţările cu un nivel ridicat de trai, cu o mortalitate de aproximativ 15-30%, diagnosticarea corectă şi rapidă a acestei patologii reprezintă o prioritate. Deşi există mai måulte investigaţii de laborator care pot orienta clinicianul către diagnosticul de embolie pulmonară, multe dintre ele trebuie interpretate în contextul profilului biologic modificat pe parcursul sarcinii. De asemenea, frecventa necesitate de a utiliza investigaţiile imagistice trebuie evaluată cu multă atenţie pentru a reduce riscurile ce apar în urma expunerii la radiaţii a fătului şi a ţesutului mamar ori a administrării substanţei de contrast. Cu toate acestea, diagnosticarea şi tratamentul corect al emboliei pulmonare nu trebuie să fie întârziate.
Introduction
Pregnancy is a risk factor that predisposes to a high incidence of deep vein thrombosis or pulmonary embolism which is one of the main causes of mortality among pregnant women in developed countries(1), increasing from 15% to 30%(2). This pathology can be explained by the rising level of fibrinogen, the lowering capacity of fibrinolysis and the rise in venous stasis in the lower limbs due to compression on the pelvic veins from the gravid uterus(3). All these changes entail a risk of venous thromboembolism(4) four times greater than in non-pregnant women, and a threefold higher risk of deep vein thrombosis than the rest of the population(4), with an equal distribution throughout all trimesters of pregnancy(5). The incidence of pulmonary embolism among pregnant women is five times greater than in the rest of the female population of the same age(6), two-thirds of the deep vein thrombosis cases cited in studies were diagnosed antepartum, the majority of them being localized to the left inferior limb(5), those of pulmonary embolism having been diagnosed predominately postpartum(4,7). Most thromboembolic events which occur during pregnancy are associated with genetic or acquired thrombophilia(8). The physiological hypercoagulability during pregnancy, the reduced blood pressure in both femoral veins after the second trimester, thrombophilia, antiphospholipid syndrome, lupus, cardiac disease, sickle cell anaemia, obesity and advanced maternal age are only some of the predisposing factors for deep vein thrombosis. C-section and postpartum infections increase the risk for thrombosis twofold and fourfold, respectively(9).
Dyspnoea, tachycardia, heart palpitations, and lower limb edema are frequent changes that occur during pregnancy and which can be mistakenly interpreted as symptoms of pulmonary embolism or deep vein thrombosis. Despite the fact that there are many scores which can rule out this diagnosis based on clinical and paraclinical parameters, few of them include the specific modifications that take place during pregnancy. Omitting this population group is justified by the impossibility of correct interpretation of the parameters which constitute the diagnostic scores. One example would be the use of D-dimer in ruling out pulmonary embolism. A negative value invalidates the diagnosis, whilst a high value cannot confirm the diagnosis. During pregnancy, the value of D-dimer increases with gestational age, meaning it can no longer be used for diagnostic purposes(10,11). A study by Chan et al., including 149 pregnant women with suspected deep vein thrombosis, concludes that a negative D-dimer value had a negative predictive value of 100%, with a sensitivity of 100% and a specificity of 60%(12). Another example consists of physiological changes in the acid-base equilibrium, with respiratory alkalosis explained by the high level of free progesterone(13). This is a sign that appears frequently in pulmonary embolism cases(14).
Diagnosis
The first step in diagnosing pulmonary embolism during pregnancy is pulmonary X-ray and lower extremity compression ultrasound, all performed to rule out deep vein thrombosis. If both investigations turn out negative, contrast-enhanced pulmonary CT angiography will be used as a diagnostic tool if the patient is not allergic to iodine and does not have renal insufficiency. If CT angiography produces an ambiguous result, it may be repeated, or ventilation/perfusion scintigraphy can be used instead. If none of these investigations are capable of producing a clear diagnosis, a time-of-flight magnetic resonance sequence can be used to exclude a possible massive pulmonary embolism(15). Magnetic resonance sensitivity rises between 90% and 100%, and has a specificity between 62% and77%, without negative effects on the mother or the baby, but with less availability and a longer time required to produce the results, compared to CT angiography(16).
Even though it has a low sensitivity and specificity in diagnosing pulmonary embolism, chest X-ray is still one of the most used investigations, because it can rule out other diseases which can have clinical similarities, and because it is necessary for the ventilation/perfusion scintigraphy.
Compression ultrasound has a 97% sensitivity and a 94% specificity(17) in diagnosing deep vein thrombosis, being sufficient for the doctor to prescribe anticoagulation treatment without any other imagistic investigations(17). Ultrasound assesses the compressibility of the veins, as well. When evaluating pregnant women, compression ultrasound should be used first, because it has no radiation effect, and because deep vein thrombosis has high incidence in this group(18-20). Ultrasound has reduced capacity in diagnosing deep vein thrombosis localized to iliac veins because of the local anatomy. However, ultrasound can evaluate the reduced blood flow and low compressibility of the femoral veins, modifications which suggest iliac veins thrombosis(17). When evaluating a patient for deep vein thrombosis, if ultrasound is negative, the patient should be further evaluated. Magnetic resonance venography is one of the imagistic investigations which deeply evaluate venous plexuses located in the pelvic area and inferior vena cava. It has a higher accuracy and sensitivity, but a lower availability(21).
The use of imaging in the diagnosis of pulmonary embolism is justified when clinical manifestations appear, but the potential risks related to exposure of the mother and the foetus to ionizing radiation should not be neglected. Although the radiation emitted during pulmonary angiography and ventilation/perfusion scintigraphy is low, even lower in case of pulmonary radiography, the decision to perform multiple imaging investigations must take into consideration the risk/benefit ratio and the fact that radiation exposure is cumulative(22,23).
The possible effects of radiation on the foetus depend on the gestational age and the duration of exposure(24). At a radiation dose of more than 15 cGy, pregnancy termination is considered, according to its gestational age, because the negative effects are most often present(25).
To avoid neurological damage, the foetus should be exposed at a dose of less than 10 cGy(26,27), but the minimum fetal risk was considered to be at a dose of less than 5 cGy(28,29). Regarding the mother’s exposure, Chen et al. describe in their study that glandular proliferation of breast tissue during pregnancy causes increased radio-sensitivity, which raises the risk of carcinogenesis(30). The highest dose of radiation to breast tissue is during pulmonary angiography.
Pulmonary angiography is the investigation of choice in the diagnosis of pulmonary embolism if the ultrasound was not conclusive, having a sensitivity between 83% and 100% and a specificity between 89% and 97%(31-33). Among pregnant women, these values are slightly lower due to the changes in the pharmacodynamics of the contrast agent(34). In addition to the rapid diagnosis, pulmonary angiography also evaluates the ratio between the right and the left ventricles, which is an important prognostic factor of survival, according to Ghaye et al.(35). Also, by performing pulmonary angiography, other pathologies can be diagnosed, which may cause the actual symptomatology. The use of contrast substance is not strictly forbidden – it is included in category B of recommendations and administered only in cases where it is absolutely necessary(36). One of the possible side effects of the administration of contrast substance on the foetus is the reduction in thyroid function, which is why it is advisable to monitor the thyroid function of the newborn in the first weeks of life(37).
The radiation dose of the breast tissue is much higher than that absorbed during pulmonary scintigraphy, this dose being reduced by the use of chest protective shields. However, the long-term risk of breast cancer is 13% higher in case of angiography compared to scintigraphy(38).
The diagnostic limitations of pulmonary angiography lie in the mistaken interpretation of lymph nodes arranged at the level of the pulmonary segments as thrombi, the inadequate opacification of the superior pulmonary lobe arteries, and the difficult highlighting of the right middle pulmonary lobe, the segments of the lingula, and the thrombi located at the level of the sub-segmental arterioles(16).
Ventilation/perfusion scintigraphy is the method of diagnosing pulmonary embolism used by some clinicians after excluding pulmonary angiography, due to its low specificity of only 10%(36). However, it is sometimes considered before CT angiography, due to lower radiation doses, the lack of contrast substance and low incorrect diagnostic rate among pregnant women.
According to a study by Seyed et al., the radiation dose to which the pregnant woman is exposed during pulmonary scintigraphy is lower than in case of angiography, suggesting scintigraphy as a first-line investigation in the diagnosis of pulmonary embolism(14).
Even though the diagnosis of this pathology involves radiation exposure of the foetus and mother, the doctor should not avoid using imaging diagnostic methods(39). The choice between angiography and pulmonary scintigraphy as a diagnostic method is controversial because, while both involve exposure to ionizing radiation, the doses are different, but so are the chances of diagnosis. Pulmonary angiography is superior to pulmonary scintigraphy in terms of specificity and sensitivity, but involves a higher dose of radiation. The clinician’s decision to use one of the two diagnostic methods is not supported by any guidelines or studies clearly describing the benefits and risks of each method and mentioning an algorithm according to which one of the variants can be chosen on a case-by-case basis.
To reduce radiation exposure of pregnant women and to ease the diagnosis of pulmonary embolism based on the clinical picture, O’Conner et al., in a retrospective study, applied the modified Wells score (Table 1)(40) to 103 women who had pulmonary angiographies during pregnancy or immediately postpartum for suspected pulmonary embolism. Only five of the patients showed clear signs of pulmonary embolism on angiography. After applying the modified Wells score (value >2) to this population group, the positive predictive value was 36%, with a sensitivity of 100% and a specificity of 90%. None of the patients with a score value less than 2 had a positive result on angiography, resulting a negative predictive value of 100%. If the score had been applied to these patients, especially to those with a low risk of pulmonary embolism, according to the score results, 31.3% of the angiographies would have no longer be needed. To generalize the use of this score, more studies are needed to support and strengthen these results(41).
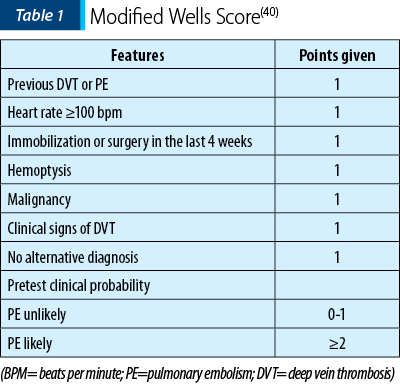
Another way to effectively diagnose patients with pulmonary embolism was proposed by Righini et al., in one of their multicenter, multinational studies, which included 395 pregnant women over the age of 18 years old, who had suggestive symptoms for pulmonary embolism. The first criterion for the selection of patients was to determine the probability of embolism, by performing the revised Geneva score (Table 2)(40). Patients with a high pretest probability underwent a compression ultrasound performed on both lower limbs. Patients with moderate or low pretest probability performed the D-dimer test. If it was positive, the patients then performed Doppler ultrasound, and if the result was negative, the diagnosis of pulmonary embolism was excluded.
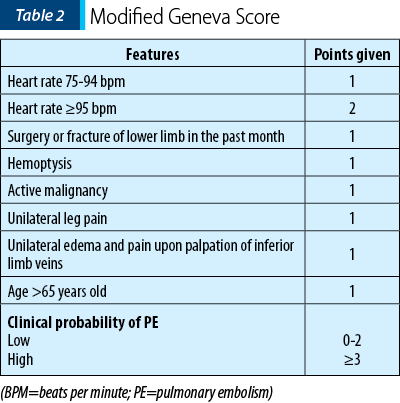
In patients diagnosed with vein thrombosis following ultrasound, anticoagulant therapy was instituted. If the diagnosis could not be clearly established after the ultrasound, the patients were directed to perform CT pulmonary angiography. Those with a positive result received anticoagulant treatment, and patients whose angiography was inconclusive received ventilation/perfusion scintigraphy (ventilation/perfusion [V/Q] scan) to establish the diagnosis of pulmonary embolism and initiate curative treatment.
Of the total number of pregnant women included in the study, 28 (7.1%) were diagnosed with pulmonary embolism (seven of them by performing Doppler ultrasound, 19 following pulmonary angiography, two by performing ventilation/perfusion scintigraphy), and in 367 cases the diagnosis was excluded. Following a review performed three months after excluding the diagnosis of pulmonary embolism, none of the patients had changes or acute symptoms of a thromboembolic episode(42).
Therapy
The treatment of choice for pulmonary embolism in pregnant patients is low molecular weight heparin, 1 mg/kg body weight every 12 hours(43). The therapeutic agent does not have the ability to cross the fetal-placental barrier, and its excretion into breast milk is minimal and thus can be used during the lactation period(44). In some cases, skin allergies manifested by pruritus, hives-like rashes, and in rare cases epidermal necrosis or bleeding have been reported as adverse reactions(43). Another disadvantage of low molecular weight heparin therapy is the possibility of an epidural hematoma following anesthesia(45), thrombocytopenia induced by heparin administration, or osteoporotic fractures(43).
Unfractionated heparin is used if the patient has renal impairment, or if emergency surgery is required. The most common side effects are bleeding, more frequent than in the case of low molecular weight heparin, allergic reactions, and osteoporotic changes with a risk of reduced bone density, between 2% and 36%(43).
The use of oral anticoagulants is prohibited during pregnancy because they cross the fetal-placental barrier, and their consumption is associated with congenital malformations, fetal or neonatal bleeding and pregnancy loss.
Once the diagnose of deep vein thrombosis is established, the patient will undergo the treatment with low molecular weight heparin at a therapeutic dose for one month. Later, the dose can be reduced by a quarter, as a maintenance dose, which will have to be maintained throughout the entire pregnancy and the postpartum period. If low molecular weight heparin is not available, it may be replaced by unfractionated heparin, the control of which is more difficult to achieve. Patients may also wear antithrombotic compressive stockings with a pressure of 30-40 mmHg to reduce the risk of post-thrombotic syndrome(46), which has an incidence of nearly 60%(47).
The use of thrombolytic treatment should be avoided in pregnant patients because of the increased risk of bleeding. This therapeutic resource should be used only in the cases of critically ill patients, with an increased risk of reembolization or death(48).
The therapeutic recommendations in pregnant patients diagnosed with deep vein thrombosis are different near term. One may attempt to install a venous filter in the inferior vena cava, cancel the effect of the anticoagulant or stop its administration and trigger the birth(49). Stopping the administration of anticoagulant treatment is not indicated without the initial installation of a venous filter, due to the increased mortality during this time lapse(50). To prevent spontaneous or caesarean delivery during the therapeutic window of anticoagulant therapy, it is recommended to induce labor or to administer unfractionated heparin instead of the low molecular weight, because its effect can be easily antagonized with protamine sulphate.
When the acute episode has ended, in patients undergoing anticoagulant treatment with therapeutic or prophylactic doses, with gestational age near term, vaginal birth or caesarean section may be attempted, without the need to install a filter in the inferior vena cava. Spinal anesthesia can be administered 12 hours after a low molecular weight heparin prophylactic dose, or 24 hours after a therapeutic dose(51).
Anticoagulant treatment can be resumed 12 hours after vaginal birth, or 24 hours after caesarean section(52), and the administration is continued during the first 6 weeks postpartum.
Prophylaxis
The prophylaxis of deep vein thrombosis recurrence in patients without other risk factors is achieved by mobilizing the patient as quickly as possible, and using antithrombotic compressive stockings(53). The presence of other risk factors for the recurrence of deep vein thrombosis (obesity, thrombophilia, immobilization, caesarean section) should be taken into consideration when establishing the treatment. The most important risk factor for the development of deep vein thrombosis is another episode of this pathology in the patient’s history(52). The chances of recurrence of a thromboembolic event in the case of a patient who had a similar episode during pregnancy are between 4% and 12%(52). Between 15% and 25% of the thromboembolic events which occur during pregnancy are actually recurrences, the risk of recurrence decreasing if the patient received anticoagulant treatment during the first episode(52). In order to prevent these episodes, prophylactic anticoagulant treatment may be initiated during the next pregnancy, stopped in the proximity of birth, and re-administered in the postpartum period, up to six weeks(54). Similar prophylaxis also applies to patients who have a diagnosis of thrombophilia with an increased risk of thrombosis, even though they have no history of thromboembolism(55).
Conclusions
The diagnosis and management in cases of pulmonary embolism among pregnant women are difficult to achieve, due to the lack of studies performed in this population group; they are also complicated by the physiological changes which occur during pregnancy. Although the incidence of this pathology is not very high, the morbidity and mortality rates are nevertheless increased. The clinician’s role is always to consider the possibility of this pathology, and at the slightest suspicion, to use all the necessary resources to establish a clear diagnosis, without hesitation. In order to facilitate the entire diagnostic process, it is critical to introduce pregnant women in as many clinical studies as possible, thus establishing the clinical criteria that will assure a rapid and correct selection of patients who need to be extensively investigated. It is also essential to establish biomarkers that can differentiate between physiological changes in pregnancy and the risk of thromboembolism.
Another aspect that should not be neglected is the need to clearly establish the situations when the imaging investigations are necessary, and which of them would be of first intention, taking into account the risks of radiation and the accuracy of the diagnosis. It is of interest to identify patients with thrombophilia (genetic or acquired) early, in order to reduce the recurrence of thromboembolic episodes, and to be able to establish the early prophylaxis during future pregnancies.
Conflict of interests: The authors declare no conflict of interests.
Bibliografie
- Panting-Kemp A, Geller SE, Nguyen T, Simonson L, Nuwayhid B, Castro L. Maternal deaths in an urban perinatal network 1992-1998. Am J Obstet Gynecol. 2000; 183(5):1207-12.
- Anderson DR, Barnes D. The use of leg venous ultrasonography for the diagnosis of pulmonary embolism. Semin Nucl Med. 2008; 38(6):412–7.
- Moser KM, Fedullo PF, LitteJohn JK, Crawford R. Frequent asymptomatic pulmonary embolism in patients with deep venous thrombosis. JAMA. 1997; 271(3):223-5.
- Heit JA, Kobbervig CE, James AH, Petterson TM, Bailey KR, Melton J. Trends in the incidence of venous thromboembolism during pregnancy or postpartum: a 30-year population-based study. Ann Intern Med. 2005; 143(10):697–706.
- Ray JG, Chan WS. Deep vein thrombosis during pregnancy and the puerperium: a meta-analysis of the period of risk and the leg of presentation. Obstet Gynecol Surv. 1999; 54(4):265–71.
- Matthews S. Imaging pulmonary embolism in pregnancy: what is the most appropriate imaging protocol?. Br J Radiol. 2006; 79(941):441–4.
- Nicolescu Veselu I, Solomon OA, Filipescu GA, Vlădăreanu R, Vlădăreanu S. Pulmonary thromboembolism associated with pregnancy - physiological procoagulant status. Ginecologia.ro. 2017; 5(16):28-31.
- Nelson SM, Greer IA. Thrombophilia and the risk for venous thromboembolism during pregnancy, delivery and puerperium. Obstet Gynecol Clin North Am. 2006; 33(3):413–27.
- James AH, Jamison MG, Brancazio LR, Myers ER. Venous thromboembolism during pregnancy and the postpartum period: incidence, risk factors, and mortality. Am J Obstet Gynecol. 2006; 194(5):1311–15.
- Nijkeuter M, Ginsberg JS, Huisman MV. Diagnosis of deep vein thrombosis and pulmonary embolism in pregnancy: a systematic review. J Thromb Haemost. 2006; 4(3):496–500.
- Nijkeuter M, Huisman MV. Diagnosing pulmonary embolism in pregnancy: is there a role for D-dimer as a stand-alone test?. Crit Care Med. 2006; 34(10):2701-2.
- Chan WS, Chunilal S, Lee A, Crowther M, Rodger M, Ginsberg JS. A red blood cell agglutination D-dimer test to exclude deep venous thrombosis in pregnancy. Ann Intern Med. 2007; 147(3):165–70.
- Zvâncă ME, Petca A, Boţ M. Progesterone, vitamins, metformin during pregnancy – “myth” or evidence-based medicine?. Ginecologia.ro. 2016; 4(14):14-7.
- Astani SA, Davis LC, Harkness BA, Supanich MP, Dalal I. Detection of pulmonary embolism during pregnancy: comparing radiation doses of CTPA and pulmonary scintigraphy. Nucl Med Commun. 2014; 35(7):704-11.
- Durán-Mendicuti A, Sodickson A. Imaging evaluation of the pregnant patient with suspected pulmonary embolism. Int J Obstet Anesth. 2011; 20(1):51-9.
- Matsumoto AH, Tegtmeyer CJ. Contemporary diagnostic approaches to acute pulmonary emboli. Radiol Clin North Am. 1995; 33(1):167–83.
- Kearon C, Julian JA, Math M, Newman TE, Ginsberg JS. Noninvasive diagnosis of deep venous thrombosis. Ann Intern Med. 1998; 128(8):663–77.
- Patel SJ, Reede DL, Katz DS, Subramaniam R, Amorosa JK. Imaging the pregnant patient for nonobstetric conditions: algorithms and radiation dose considerations. Radiographics. 2007; 27(6):1705–22.
- Pahade JK, Litmanovich D, Pedrosa I, Romero J, Bankier AA, Boiselle PM. Imaging pregnant patients with suspected pulmonary embolism: what the radiologist needs to know. Radiographics. 2009; 29(3):639–54.
- Remy-Jardin M, Pistolesi M, Goodman LR, Gefter WB, Gottschalk A, Mayo JR, Sostman HD. Management of suspected acute pulmonary embolism in the era of CT angiography: a statement from the Fleischner Society. Radiology. 2007; 245(2):315–29.
- Evans AJ, Sostman HD, Witty LA, Paulson EK, Spritzer CE, Hertzberg BS, Delong DM. Detection of deep venous thrombosis: prospective comparison of MR imaging and sonography. J Magn Reson Imaging. 1996; 6(1):44–51.
- Sodickson A, Baeyens PF, Andriole KP, Prevedello LM, Nawfel RD, Hanson R, Khorasani R. Recurrent CT, cumulative radiation exposure, and associated radiation-induced cancer risks from CT of adults. Radiology. 2009; 251(1):175–84.
- Griffey RT, Sodickson A. Cumulative radiation exposure and cancer risk estimates in emergency department patients undergoing repeat or multiple CT. Am J Roentgenol. 2009; 192(4):887–92.
- McCollough CH, Schueler BA, Atwell TD, Braun NN, Regner DM, Brown, DL, LeRoy AJ. Radiation exposure and pregnancy: when should we be concerned?. Radiographics. 2007; 27(4):909–17.
- Wieseler KM, Bhargava P, Kanal KM, Vaidya S, Stewart BK, Dighe MK. Imaging in pregnant patients: examination appropriateness. Radiographics. 2010; 30(5):1215–29.
- Streffer C, Shore R, Konermann G, Meadows A, Uma Devi P, Preston Withers J, Holm LE, Stather J, Mabuchi K, HR. Biological effects after prenatal irradiation (embryo and fetus). A report of the International Commission on Radiological Protection. Ann ICRP. 2003; 33(1-2):5-206.
- Wagner LK, Lester RG, Saldana LR. Exposure of the pregnant patient to diagnostic radiations: a guide to medical management. 2nd ed, Madison, WI: Medical Physics Publishing. 1997.
- US Nuclear Regulatory Commission. National council on radiation protection and measurements, medical radiation exposure of pregnant and potentially pregnant women. Washington DC, NCRP Report, 1997;54:9-11.
- ACOG Committee on Obstetric Practice. Guidelines for diagnostic imaging during pregnancy. ACOG Committee opinion no. 299. Obstet Gynecol. 2004; 104(3):647–51.
- Chen J, Lee RJ, Tsodikov A, Smith L, Gaffney DK. Does radiotherapy around the time of pregnancy for Hodgkin’s disease modify the risk of breast cancer?. Int J Radiat Oncol Biol Phys. 2004; 58(5):1474–79.
- Stein PD, Fowler SE, Goodman LR, Gottschalk A, Hales CA, Hull RD, Sostman HD. Multidetector computed tomography for acute pulmonary embolism. N Engl J Med. 2006; 354(22):2317–27.
- Qanadli SD, Hajjam ME, Mesurolle B, Barré O, Bruckert F, Joseph T, Lacombe P. Pulmonary embolism detection: prospective evaluation of dual-section helical CT versus selective pulmonary arteriography in 157 patients. Radiology. 2000; 217(2):447–55.
- Winer-Muram HT, Rydberg J, Johnson MS, Tarver RD, Williams MD, Shah H, Trerotola SO. Suspected acute pulmonary embolism: evaluation with multi-detector row CT versus digital subtraction pulmonary arteriography. Radiolog. 2004; 233(3):806–15.
- Karabulut N. Inferior vena caval blood flow affects the quality of CTPA during pregnancy. Eur Radiol. 2009; 19(7):1693.
- Ghaye B, Ghuysen A, Willems V, Lambermont B, Gerard P, D’Orio V, Dondelinger RF. Severe pulmonary embolism: pulmonary artery clot load scores and cardiovascular parameters as predictors of mortality. Radiology. 2006; 239(3):884–91.
- Webb JA, Thomsen HS, Morcos SK, Members of Contrast Media Safety Committee of European Society of Urogenital Radiology (ESUR). The use of iodinated and gadolinium contrast media during pregnancy and lactation. Eur Radiol. 2005; 15(6):1234–40.
- Tremblay E, Therasse E, Thomassin-Naggara I, Trop I. Quality initiatives: guidelines for use of medical imaging during pregnancy and lactation. Radiographics. 2012; 32(3):897–911.
- Winer-Muram HT, Boone JM, Brown HL, Jennings SG, Mabie WC, Lombardo GT. Pulmonary embolism in pregnant patients: fetal radiation dose with helical CT. Radiology. 2002; 224(2):487–92.
- McCollough CH, Schueler BA, Atwell TD, Braun NN, Regner DM, Brown DL, LeRoy AJ. Radiation exposure and pregnancy: when should we be concerned ?. Radiographics. 2007; 27(4):909–17.
- Grupul de Lucru pentru Diagnosticul şi Tratamentul Emboliei Pulmonare Acute al Societăţii Europene de Cardiologie. Ghidul european de diagnostic şi tratament al emboliei pulmonare acute (versiunea 2014). Rom J Cardiol. 2015; 25(1):75-133.
- O’Connor C, Moriarty J, Walsh J, Murray J, Coulter-Smith S, Boyd W. The application of a clinical risk stratification score may reduce unnecessary investigations for pulmonary embolism in pregnancy. J Matern Fetal Neonatal Med. 2011; 24(12):1461-64.
- Righini M, Robert-Ebadi H, Elias A, Sanchez O, Le Moigne E, Schmidt J. CT-PE-Pregnancy Group. Diagnosis of pulmonary embolism during pregnancy:a multicenter prospective management outcome study. Ann Intern Med. 2018; 169(11):766-73.
- Greer IA, Nelson-Piercy C. Low-molecular-weight heparins for thromboprophylaxis and treatment of venous thromboembolism in pregnancy: a systematic review of safety and efficacy. Blood. 2005; 106(2):401–7.
- Richter C, Sitzmann J, Lang P, Weitzel H, Huch A, Huch R. Excretion of low molecular weight heparin in human milk. Br J Clin Pharmacol. 2001; 52(6):708–10.
- Howell R, Fidler J, Letsky E, de Swiet M. The risks of antenatal subcutaneous heparin prophylaxis: a controlled trial. Br J Obstet Gynaecol. 1983; 90(12):1124–28.
- Brandjes DPM, Buller HR, Heijboer H, Huisman MV, de Rijk M, Jagt H, ten Cate JW. Randomised trial of effect of compression stockings in patients with symptomatic proximal-vein thrombosis. Lancet. 1997; 349(9054):759–62.
- McColl MD, Ellison J, Greer IA, Tait RC, Walker ID. Prevalence of the posthrombotic syndrome in young women with previous venous thromboembolism. Br J Haematol. 2000; 108(2):272–74.
- Leonhardt G, Gaul C, Nietsch HH, Buerke M, Schleussner E. Thrombolytic therapy in pregnancy. J Thromb Thrombolysis. 2006; 21(3):271–76.
- Jamjute P, Reed N, Hinwood D. Use of inferior vena cava filters in thromboembolic disease during labor: case report with a literature review. J Matern Fetal Neonatal Med. 2006; 19(11):741–44.
- Kearon C, Hirsh J. Management of anticoagulation before and after elective surgery. N Engl J Med. 1997; 336(21):1506–11.
- Horlocker TT, Wedel DJ, Benzon H, Brown DL, Enneking KF, Heit JA, Yuan CS. Regional anesthesia in the anticoagulated patient: defining the risks (the second ASRA Consensus Conference on Neuraxial Anesthesia and anticoagulation). Reg Anesth Pain Med. 2003; 28:172–97.
- James AH, Grotegut CA, Brancazio LR, Brown H. Thromboemoblism in pregnancy: recurrence and its prevention. Semin Perinatol. 2007; 31(3):167–75.
- Geerts WH, Bergqvist D, Pineo GF, Heit JA, Samama CM, Lassen MR, Colwell CW. Prevention of venous thromboembolism: American College of Chest Physicians evidence-based clinical practice guidelines. Chest. 2008; 133(6):381–453.
- Voicu DI, Bodean OM, Munteanu O, Arsene LV, Boieru C, Brătilă E, Cîrstoiu MM. Correlations between hereditary thrombophilias and recurrent abortions. Ginecologia.ro. 2018; 6(19):8-11.
- Bates SM, Greer IA, Pabinger I, Sofaer S, Hirsh J. Venous thromboembolism, thrombophilia antithrombotic therapy and pregnancy: American College of Chest Physicians evidence-based clinical practice guideline. Chest. 2008; 133(6):844S–86S.
Articole din ediţiile anterioare
Profilaxia trombozei post-partum - de cât timp este nevoie pentru a fi în siguranţă?
Perioada post-partum este perioada cu riscul cel mai ridicat pentru tromboză. Riscul poate fi influenţat de vârsta maternă şi de durata până la naş...
Managementul gravidei cu hepatită virală B cronică - review de literatură şi prezentarea experienţei Spitalului Clinic de Obstetrică-Ginecologie „Prof. Dr. Panait Sîrbu”, Bucureşti
Managementul hepatitei virale B cronice în timpul sarcinii rămâne o provocare și implică numeroase aspecte de îngrijire materno-fetale. Obiectivu...