Objective. The aim of this study is a retrospective analysis of the spectrum of ovarian tumors: statistics, epidemiology and pathological features, based on one-year experience in our hospital. Materials and method. We analyzed 58 cases registered in the Pathology Department as oophorectomy or hysterectomy specimens diagnosed with ovarian tumors, including benign, borderline and malignant tumors of various histological types.
Results. Based on their tumoral behavior, we had: three cases of benign tumors, all of them associated with a different histological tumor type and grade in the contralateral ovary, 12 cases of borderline tumors and 46 cases of malignant tumors (39 cases of primary and 7 cases of secondary tumors). The most frequent histologic type was represented by high-grade serous carcinoma (37.9%). 85.7% of the secondary tumors were endometrial carcinomas extensions. Rare primary ovarian tumors were represented by: adult granulosa cell tumor, clear cell carcinoma, mixed serous-mucinous carcinoma and undifferentiated pleomorphic sarcoma (1.72% for each type). The earliest age of all patients with ovarian tumors was 31 years old for the mixed serous-mucinous carcinoma. Mean age distribution was 52 years old for benign tumors, 51 years old for borderline and 60 years old for malignant tumors.
Conclusions. Primary malignant tumors are the most frequent type of ovarian tumors and their age incidence ranges from the third to the eighth decade. The majority of secondary ovarian tumors are of endometrial origin.
Studiu clinico-patologic al tumorilor ovariene - experienţa de un an într-un centru medical
A clinical-pathological study of ovarian tumors - one-year center experience
First published: 18 decembrie 2017
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/Gine.18.4.2017.1329
Abstract
Rezumat
Obiectiv. Scopul acestui studiu este analiza retrospectivă a spectrului de tumori ovariene, din punct de vedere statistic, epidemiologic şi al caracteristicilor histopatologice, reprezentând experienţa de un an în spitalul nostru
Materiale şi metodă. Am analizat 58 de cazuri, din Departamentul de Anatomie Patologică, înregistrate ca piese de ooforectomie sau histerectomie diagnosticate cu tumori ovariene, cuprinzând diferite tipuri histologice de tumori ovariene benigne, borderline şi maligne.
Rezultate. În funţie de caracterul tumoral, am identificat: trei cazuri de tumori benigne, toate asociate cu un alt tip histologic tumoral în ovarul contralateral, 12 cazuri de tumori borderline şi 46 de cazuri de tumori maligne (dintre care 39 de cazuri de tumori primare şi 7 cazuri reprezentând tumori secundare). Cel mai frecvent tip histologic a fost reprezentat de carcinoame seroase cu grad înalt de malignitate (37,9%). 85,7% din tumorile secundare identificate au reprezentat extensii tumorale locoregionale de carcinoame endometriale. Tumorile ovariene primare rare au fost: tumora cu celule de granuloasă - tipul adult, carcinomul cu celule clare, carcinomul mixt seromucinos, sarcomul pleomorf nediferenţiat (fiecare cu un procent de 1,72%). Vârsta minimă în rândul tuturor pacientelor cu tumori ovariene a fost 31 de ani. Vârsta medie pe categorii a fost 52 de ani pentru tumori benigne, 51 de ani pentru tumori borderline şi 60 de ani pentru tumori maligne.
Concluzii. Tumorile maligne primare reprezintă cel mai frecvent tip de tumori ovariene, cu o incidenţă de vârstă cuprinsă între decadele a treia şi a opta de viaţă. Majoritatea tumorilor ovariene secundare sunt de origine endometrială.
Introduction
A female’s risk at birth of having an ovarian tumor sometimes in her life is 6-7%(1). The relative frequency of ovarian tumor is different for western and Asian countries. Two third of ovarian tumors occur in women of reproductive age group(1). There are numerous types of ovarian tumors and among them, about 80% are benign, and these occur mostly in young women between 20 and 45 years old. Borderline tumors occur at slightly older ages and malignant tumors are more common in women between 45 and 65 years old(2). Ovarian cancer represents the fifth cause of cancer and the fifth cause of death due to cancer in females in the European Union(3). The most common malignant ovarian tumors are serous carcinomas, and these account for 40% of all ovarian cancer. Based on their origin, there are various types of tumors, but the most frequent are: surface epithelial tumors (65-70%), germ cell tumors (15-20%), sex cord-stromal tumors (5-10%) and secondary tumors (5%)(2).
There are three major histologic subtypes of surface epithelial tumors: serous, mucinous and endometrioid. Serous tumors represent 50% of epithelial tumors and about 30% of all ovarian tumors. Serous tumors can be benign or borderline (70%) and malignant (30%). These types of tumors are associated with KRAS, BRAF, p53, BRCA1 and BRCA2 mutations(2,4). Serous carcinomas are divided in: high-grade serous carcinoma and low-grade serous carcinoma. The low-grade variant represents only 5% of serous carcinomas(4).
Mucinous tumors are less frequent than serous tumors; they can also be benign, borderline or malignant, but mucinous carcinoma account for 3-4% of all primary ovarian carcinomas(4). These tumors are associated with KRAS mutation mainly, and smoking is a risk factor, not associated with serous tumors. Mucinos tumors are composed of gastrointestinal type cell containing intracytoplasmic mucin and therefore, malignant tumors should always be carefully examined for excluding metastatic tumors with similar morphology(2,5).
Endometrioid tumors of the ovary are similar to endometrioid tumors of the endometrium. Endometrioid carcinomas account for 20% of all ovarian cancers, but 15-30% of them are accompanied by endometrioid carcinoma of the uterus and is a matter of debate whether they are independent tumors or metastatic spread from one another(2,6). They are associated with mutations of PTEN, KRAS, b-catenin and microsatellite instability(2).
Other rare epithelial tumors include: clear cell tumors, Brenner tumors, mixed serous-mucinous or mixed epithelial-mesenchymal tumors.
Germ cell tumors represent approximately 15-20% of all ovarian tumors and are composed of a number of different histological types derived from primitive germ cells of the embryonic gonad(2). In children and adolescents, more than 60% of ovarian neoplasms are of germ cells origin, and one third of them are malignant. In adults, they are mostly benign (95%) and consist of mature cystic teraromas (dermoid cysts)(4,5). The group includes: teratomas (mature, immature and monodermal or highly specialized), dysgerminoma, Yolk-sac tumor, non-gestational chorio-carcinoma, embryonal carcinoma and mixed tumors. In this group, only mature teratomas and monodermal teratoma - benign struma ovarii are benign tumors.
Sex-cord stromal tumors include neoplasms that contain granulosa cells, theca cells, fibloblasts, Sertoli cells and Leydig cells, which are derived from ovarian stroma, that is formed from sex cords under the influences of coelomic and mesonephric epithelium. The most frequent tumors of this category is adult granulosa cell tumor (95%)(5,7). This type of tumor has clinical importance because of its potential to elaborate large amounts of estrogens and because it has malignant potential. Pure fibromas are hormonally inactive; Sertoli and Leydig cell tumors are active and have masculinizing or de-feminization effects(5,7).
Metastases account for 8% of malignant ovarian neoplasm(7). Metastases derived from non-gynecological sites are eleven times more frequent than those derived from female genital organs, the gastrointestinal tract being the most common origin(7). Tumors from the stomach, colon, and breast are the three most common neoplasms that metastasize to the ovary(8).
Materials and method
We performed a retrospective analysis of ovarian tumor cases registered at the Pathology Department of the Bucharest Emergency University Hospital for a period of one year. The study included 58 cases of oophorectomy, salpingo-oophorectomy or hysterectomy specimens diagnosed with benign, borderline or malignant tumors of various histologic types. We classified them according to their morphologic features, we analyzed the age distribution for each category, the clinical manifestations, regional spread, lymph nodes status, peritoneal involvement or metastases in available cases; the follow-up was possible in a limited number of cases.
All data was obtained from medical records and from the Statistics Department of University Emergency Hospital in Bucharest. The statistical analysis was performed using SPSS™ and Microsoft Excel 2010™.
Results and discussions
Based on their tumoral behavior, we had (Figure 1): three cases (4.92%) of benign tumors, all of them associated with a different histological tumor type and grade in the contralateral ovary (two serous cystadenomas, both associated with serous borderline tumors and one endometriotic cyst associated with mucinous borderline tumor of the contralateral ovary); 12 cases (19.67%) of borderline tumors (5 cases [41.66%] of serous type, 5 cases [41.66%] of mucinous type and two cases [16.66%] of endometrioid borderline tumor), 46 cases (75.41%) of malignant tumors (39 cases of primary [84.78%] and 7 cases of secondary tumors [15.21%]). In our study we found a smaller prevalence of benign tumors of only 4.92%, compared to 80% found in literature(1,2).
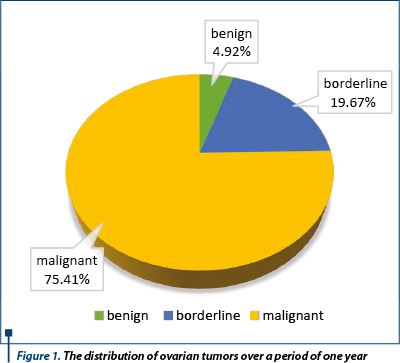
The most frequent histologic type among all ovarian tumors was represented by high-grade serous carcinoma (37.9%). According to the data we found in the specialty literature, high-grade serous carcinoma represents approximately 40% of ovarian cancers and 9% of all ovarian tumors(2,9).
Mucinous carcinoma accounted for 17.94% of all primary ovarian carcinomas, much higher than in other studies(5,10).
Endometrioid tumors of borderline type are rare neoplasms and patients with these tumors have associated endometriosis in 63% of cases(4). Studies of molecular alteration have suggested that this tumors do actually progress from endometriosis(4,11).
85.7% of the secondary tumors were endometrial carcinomas extensions, with six cases of endometrioid endometrial carcinoma and one case of uterine adenosquamos carcinoma. We had one case of poorly differentiated carcinoma of unknown origin. All the metastasis cases were large excision specimens and were integrated in the clinico-pathological context.
The gynecological origin is usually less frequent than the gastrointestinal origin, which is the first cause of ovarian metastasis(7,8,12).
Rare primary ovarian tumors were represented by: adult granulosa cell tumor (Figure 2), clear cell carcinoma, mixed serous-mucinous carcinoma, and undifferentiated pleomorphic sarcoma (1.72% for each type).
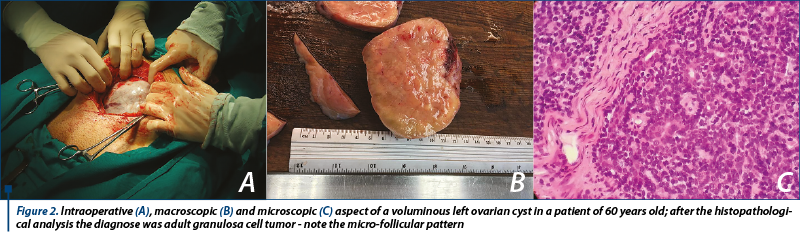
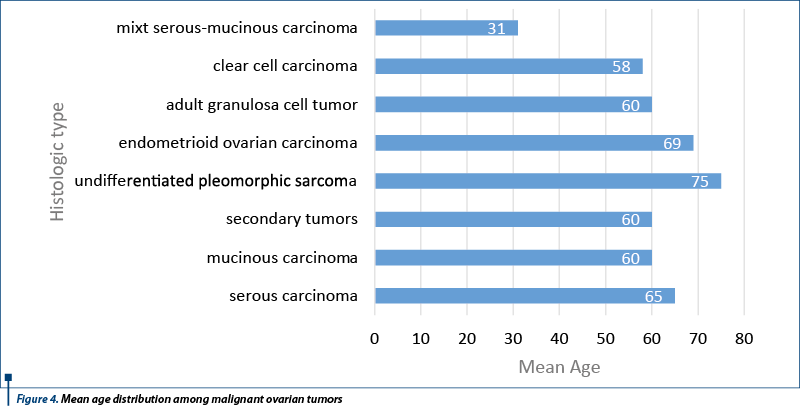
The earliest age of a patient with ovarian tumor was 31 years old for the mixed serous-mucinous carcinoma, a younger age than mean age for this type of tumor, but cases of patients within 16 to 79 years had been previously reported(13,14). Mean age distribution was: 52 years old for benign tumors, with a range of 45 to 67 years old; 51 years old for borderline tumors, with a range of 32 to 73 years old; 60 years old for malignant tumors, with a range of 31 to 83 years old (Figure 3). Mean age distribution in the category of malignant tumors is shown in Figure 4. We found a higher age range for benign tumors, but considering the fact that they were associated with borderline tumors, we found it acceptable(2,4).

We had two cases of tumoral recurrence with multiple metastases of previous high-grade serous carcinoma in patients of 62 and 70 years old, respectively, and one deceased patient of 75 years old, with undifferentiated pleomorphic sarcoma. Primary sarcomas account for of 2-3% of all female genital tract cancers, and ovaries are unusual sites for sarcomas. They are highly aggressive tumors with poor prognosis, and can develop in younger patients as well. Kurtoglu et al. reported the case of a 46-year-old women with undifferentiated pleomorphic sarcoma with focally rhabdomyosarcomatous differentiation of the ovary, with recurrent disease and fail of chemotherapy treatment(15).
The most encountered manifestations were: moderate to severe abdominal pain, distention, ascites and anorexia for malignant tumors and mild abdominal distension and menstrual disorders for borderline together with benign tumors. The symptoms are usually found in large tumors(16,17).
The treatment of ovarian cancer with molecular targeting therapy, platinum and taxane containing chemotherapy and other specific drugs has improved the prognosis over time(18). Currently, the standard primary therapy for advanced disease involves a combination of maximal cytoreductive surgery and chemotherapy with carboplatin plus paclitaxel or with carboplatin alone. Despite initial high response rates, a large proportion of patients relapse, resulting in a therapeutic challenge(19,20). Because these patients are not curable, the goal of therapy becomes the improvement in both quality and length of life. Single-agent paclitaxel, topotecan, or pegylated liposomal doxorubicin have demonstrated activity in this patient population and are reasonable treatment options(19,21).
Conclusions
In our study, primary malignant tumors are the most frequent type of ovarian tumors and their age incidence ranges from the third to the eighth decade. We found higher prevalence rates for high-grade serous carcinomas. The majority of secondary ovarian tumors we studied were of endometrial origin, which may suggest that gynecological primary situs can be considered of greater potential of spreading to the ovaries than previously thought.
Bibliografie
2. Kumar V, Abbas AK, FaustoN, Aster J. Robbins and Cotran. Pathologic basis of Disease; 8th Ed Philadelphia Saunders Elsevier, 2010:1041.
3. Ferlay J, Steliarova-Foucher E, Loertet-Tieulent et al. Cancer incidence and mortality patterns in Europe: Estimates for 40 countries in 2012. Eur J Cancer, 2013;49:1374-403.
4. Kurman RJ, Carcangiu ML, Herrington CS, Young RH. WHO Classification of Tumours of Female Reproductive Organs. 4th Ed Lyon, IARC;2014:8-86
5. Kurman RJ, Ellenson LH, Ronnett BM. Blaustein’s Pathology of the Female Genital Tract. Sixth Edition. London, Springer Science Business Media, 2011;680-992.
6. Grammatoglou X, Skafida E, Glava C et al. Synchronous endometrioid carcinoma of the uterine corpus and ovary. A case report and review of the literature. Eur J Gynaecol Oncol. 2009;30(4):437-9.
7. Nucci MR, Oliva E. Gynecologic Pathology. Churchill Livingstone Elsevier; 2009:367-610.
8. Yamanishi Y, Koshiyama M, Ohnaka M. et al. Pathways of metastases from primary organs to the ovaries. Obstet Gynecol Intal, 2011, ID 612817.
9. Lengyel E. Ovarian cancer development and metastasis. Am J Pathol, 2010;177(3): 1053–64.
10. Seidman JD, Horkayne-Szakaly, Haiba M, Boice CR, Kurman RJ, Ronnett BM. The histologic type and stage distribution of ovarian carcinomas of surface epithelial origin. Int J Gynecol Pathol, 2004;23(1):41-4.
11. Wei J, William J, Bulun S. Endometriosis and ovarian cancer: A Review of Clinical, Pathologic and Molecular Aspects. Int J Gynecol Pathol, 2011;30(6): 553–68.
12. Ulbright TM, Roth LM, Stehman FB. Secondary Ovarian Neoplasia. A Clinicopathologic study of 35 cases cancer 1984;53:1164-74.
13. Taylor J, McCluggage WG. Ovarian seromucinous carcinoma: report of a series of a newly categorized and uncommon neoplasm. Am J Surg Pathol, 2015;39(7):983-92.
14. Tang SX, Sun YH, Xu Y et al. Clinicopathologic study of seromucinous carcinoma of ovary. Zhonghua Bing Li Xue Za Zhi. 2016;45(11):774-9.
15. Kurtoglu E, Celik H, Kokcu A et al. Undifferentiated pleomorphic sarcoma with focally rhabdomyosarcomatous differentiation of the ovary. Eur J Gynaecol Oncol, 2016;37(3):401-3.
16. Cîrstoiu MM, Sajin M, Secară DC, Munteanu O, Cîrstoiu FC. Giant ovarian mucinous cystadenoma with borderline areas: a case report. Rom J Morph Embryo, 2014;55(4):1443-7.
17. Oprescu DN, Herghelegiu C, Moldoveanu A. Granulosa cell tumor of the ovary Ginecologia.ro, 2017;5(16):48-54.
18. Shimada T, Saito T, Shimokawa M et al. Improvement in the prognosis of ovarian cancer in the era before addition of molecular targeting therapy. Jpn J Clin Oncol,2017;24:1-5.
19. Fung-Kee-Fung M, Oliver T, Elit L et al. Optimal chemotherapy treatment for women with recurrent ovarian cancer. Curr Oncol, 2007;14(5):195-208.
20. Högberg T, Glimelius B, Nygren P et al. A systematic overview of chemotherapy effects in ovarian cancer. Acta Oncol, 2001;40(2-3):340-60.
21. Colombo N, Parma G, Bocciolone L et al. Medical therapy of advanced malignant epithelial tumours of the ovary. Forum (Genova), 2000;10(4):323-32.
Articole din ediţiile anterioare
De la hiperplazia limfoidă benignă a bazei limbii la hipotiroidism iatrogenic şi osteoporoză severă de menopauză
Aspectele anatomopatologice descrise în hiperplazia limfoidă benignă (HLB) sunt rar decelate la nivelul capului şi al gâtului. Prognosticul este ...
Fetal and neonatal diagnosis and management in a case of a neonate with congenital heart disease – case report
Boala cardiacă congenitală (BCC) este cea mai frecventă formă de anomalie congenitală. La făt, canalul arterial (CA) deviază sângele neoxigenat din...
Conduita terapeutică în tumorile borderline la femeia de vârstă fertilă
Tumorile ovariene borderline reprezintă un grup distinct de leziuni heterogene, diferite din punct de vedere histologic şi ca evoluţie de carcino...
Tumoră phyllodes gigantică
Tumorile phyllodes sunt tumori rare, reprezentând doar 1% din toate cazurile de tumori ale sânului. Acestea au tendinţa de a creşte rapid, disemi...