Introduction. Several previous studies have identified an association between endometriosis and the development of ovarian carcinomas. This study aims to follow-up the prevalence of endometriosis and the histological features in ovarian tumors. Materials and method. The study group included 50 patients from the County Emergency Hospital of Galaţi diagnosed with different histological types of ovarian tumors of the surface epithelium during the period 2015-2018. The cases were reviewed in order to highlight the presence of endometriosis and the different histopathological changes. Results. In this study, the incidence of endometriosis was 14% (7/50). The average age of patients in the group with endometriosis associated with ovarian tumors was 59.4±11.35, respectively 59.5±13.4 in the group presenting only ovarian tumors. From the group of 50 patients, 29 (58%) were diagnosed with serous carcinoma, 8 (16%) with endometrioid carcinoma, 5 (10%) with mucinous carcinoma, 4 (8%) with serous borderline tumors, one (2%) with mucinous borderline tumor, one (2%) with clear cell carcinoma, one (2%) with undifferentiated carcinoma, and one (2%) with malignant Brenner tumor. From the seven cases of tumors associated with endometriosis, two presented areas with atypia, considered premalignant lesions. Conclusions. Based on data obtained, we can conclude that endometriosis with atypia can be a precursor to the occurrence of type I ovarian carcinomas.
Incidenţa endometriozei şi a endometriozei atipice în cazul tumorilor ovariene epiteliale
Incidence of endometriosis and atypical endometriosis in epithelial ovarian tumors
First published: 29 octombrie 2019
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/ObsGin.67.3.2019.2615
Abstract
Rezumat
Introducere. Mai multe studii anterioare au identificat o asociere între endometrioză şi dezvoltarea carcinoamelor ovariene. Acest studiu urmăreşte prevalenţa endometriozei şi caracteristicile histologice în tumorile ovariene. Materiale şi metodă. În lotul de studiu au fost incluse 50 de paciente diagnosticate cu diferite tipuri histologice de tumori ovariene ale epiteliului de suprafaţă în perioada 2015-2018 la Spitalul Judeţean de Urgenţă din Galaţi. Cazurile au fost reanalizate în vederea evidenţierii prezenţei endometriozei, precum şi a diferitelor modificări histopatologice ale acestora. Rezultate. Incidenţa endometriozei în acest studiu a fost de 14% (7/50). Vârsta medie a pacientelor a fost 59,4±11,35 ani în lotul pacientelor care au prezentat endometrioză asociată şi 59,5±13,4 ani în lotul pacientelor care au prezentat doar tumori ovariene. Din cele 50 de paciente, 29 (58%) au fost diagnosticate cu carcinom seros, 8 (16%) cu carcinom endometrial, 5 (10%) cu carcinom mucinos, 4 (8%) cu tumoră seroasă borderline, una (2%) cu borderline mucinos, una (2%) cu carcinom cu celule clare, una (2%) cu carcinom nediferenţiat şi una (2%) cu tumoră Brenner malignă. Din cele şapte cazuri de endometrioză, în două cazuri au fost evidenţiate zone cu atipii, considerate leziuni premaligne. Concluzii. Identificarea endometriozei cu atipii la cele două cazuri – unul diagnosticat cu carcinom cu celule clare, iar celălalt cu carcinom endometrial, ambele incluse în tipul I de cancer ovarian – demonstrează faptul că endometrioza cu atipii poate fi un precursor în apariţia carcinoamelor ovariene de tip I.
Introduction
Endometriosis is a chronic estrogen-dependent modification affecting about 3-10% of active females and 2-5% of postmenopausal women. It is assumed that approximately 25-80% of the women who can’t get pregnant are suffering from this disease(1-3).
Endometriosis is a gynecological condition, being characterized from a histopathological point of view by the presence of glandular structures at the level of the endometrium and stroma in ectopic locations, such as ovary, pelvis, bladder, Fallopian tube, ovary representing the most common location(4,5).
The literature comprises multiple studies highlighting the link between the presence of endometriosis and its malignant transformation in the ovary, although other studies have not identified associations between these two(6,7).
In terms of frequency, the ovarian cancer holds second place after the endometrial carcinoma, accounting for about 3% of cancers diagnosed in women, one of the most common causes of death among women. Ovarian surface epithelial tumors account for about 90% of all ovarian tumors(8).
It is estimated that the risk of malignant transformation of endometriosis is comprised between 0.5% and 1%. Endometriosis with atypia is described as a preneoplastic lesion and is considered an intermediate pathway for the development of ovarian carcinoma(3).
In 1925, Sampson described for the first time the implications of endometriosis in neoplastic lesions by establishing several important criteria: the presence of endometriosis and the tumor in the same ovary, a similar histological pattern, the exclusion of the presence of tumor metastasis from other locations(8).
In 1953, Scott added one more criterion: highlighting the continuity between the benign and the malignant epithelium(9).
Atypia from endometriosis was proposed as a preneoplastic lesion between endometriosis and ovarian cancer, as it refers to two major histological changes: cytological atypia and architectural atypia known as hyperplasia(3-10).
Cytological atypia is highlighted in the epithelium of an endometrial cyst, while the hyperplasia resembles to the hyperplasia present in the endometrium (simple or complex with or without atypia)(11).
Materials and method
The study comprises patients of the Emergency County Hospital of Galaţi diagnosed with ovarian carcinoma during the period January 2015 – December 2018. These patients have undergone surgical interventions, such as hysterectomy or bilateral/unilateral salpingo-oophorectomy. The histopathological results were analyzed retrospectively to identify the presence of endometriosis.
A number of diagnosed cases and ovarian tumors were analyzed in order to identify the presence or absence of endometriosis, as well as the relationship between these two, based on the criteria listed before. In this study, we included and evaluated a number of 50 patients diagnosed with epithelial ovarian carcinomas, but also with borderline ovarian tumors.
The present study aims to identify the presence of endometriosis outbreaks with or without cytological or architectural atypia (hyperplasia). Each case was evaluated from a histopathological point of view. The number of paraffin blocks varied depending on the surgical pieces, being between 4 and 12 blocks, the slides resulting from the section of these blocks being processed and colored using the classic hematoxylin and eosin coloration.
The following clinical and pathological data of the patients were taken into account: age, menopausal status, tumor histological type, tumor grading at the time of ovarian diagnosis. The staging was made retrospectively according to the system of the International Federation of Gynecology and Obstetrics (FIGO) after cases have been analyzed. The cases were divided into two large groups: early stage (FIGO stage I) and late stage (FIGO stage II-IV). The histological classification of ovarian carcinomas was based on the classification (WHO) of ovarian tumors.
For statistical analysis, we used the Fisher’s exact test in order to compare the age of patients with endometriosis and without endometriosis associated with ovarian carcinomas, the unpaired t-test for the percentage calculation of cases with endometriosis and without endometriosis, and Chi-squared test for independence.
Results
The histopathological evaluation performed on the group of 50 patients during the 2015-2018 period has the following results: 45 cases were diagnosed with malignant tumors, 5 cases being borderline tumors. Ovarian carcinoma was identified as being associated with endometriosis in 14% (7/50) of cases, while 86% (43/50) did not associate endometriosis.
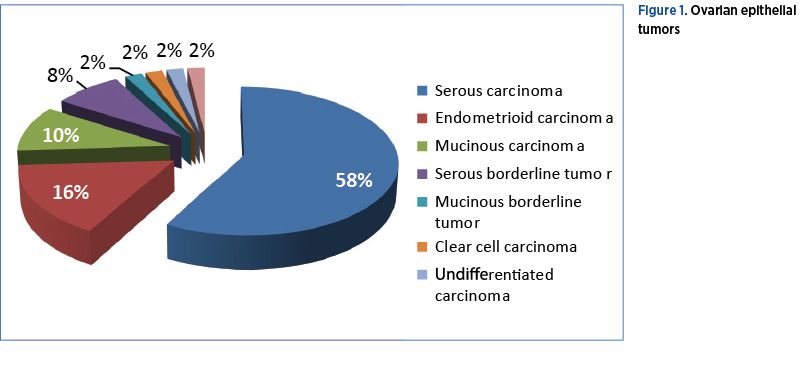
The most common type of tumor was the ovarian serous carcinoma, present in 29 cases out of 50 (58%), followed in descending order by 8 (16%) endometrioid carcinomas, 5 (10%) mucinous carcinoma, 4 (8%) borderline serous tumor, one (2%) borderline mucinous, one (2%) clear cell carcinoma, one (2%) undifferentiated carcinoma, and one (2%) Brenner malignant tumor (Figure 1).
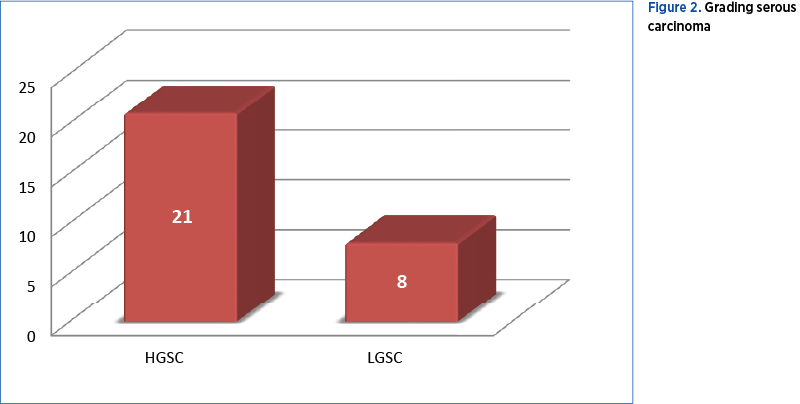
Of the 29 cases diagnosed with ovarian serous carcinoma, 21 (42%) were high-grade serous carcinoma (HGSC) and 8 (16%) were low-grade serous carcinoma (LGSC) – Figure 2.
According to FIGO, the 50 cases were classified as follows: 20 (40%) FIGO stage I, 14 (28%) stage II, 14 (28%) stage III, two stage IV (4%), 40% of the cases being in an early stage, and the remaining 60% being in a late stage.
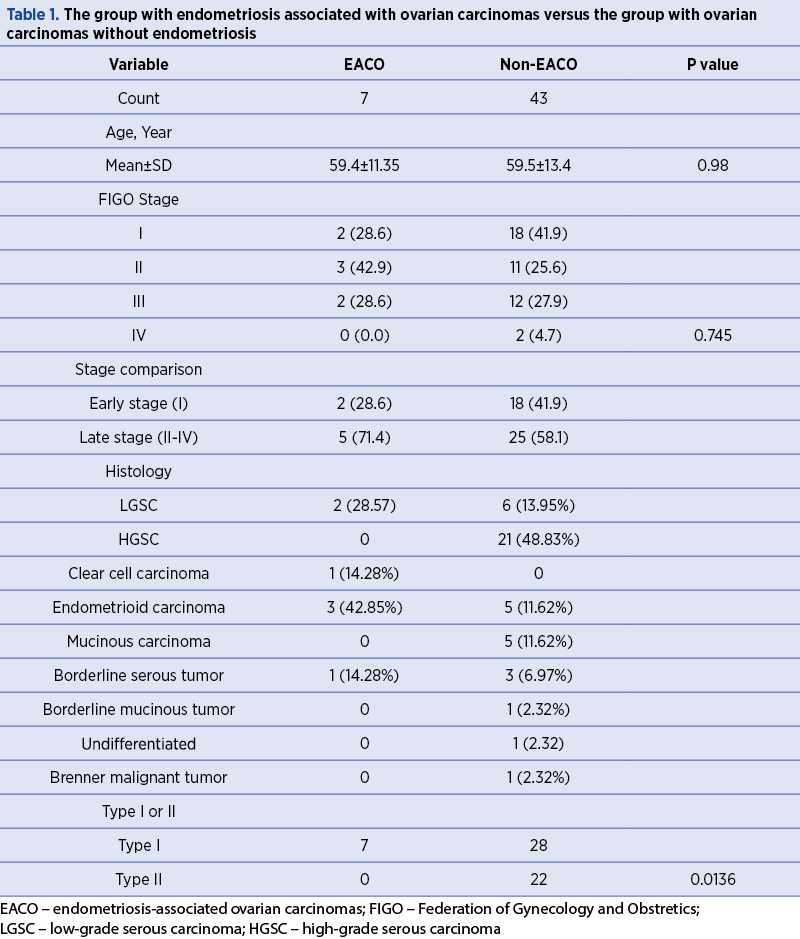
From the seven patients with endometriosis, 42.85% (3/7) were identified with endometrioid carcinoma, 28.57% (2/7) had low-grade serous carcinoma, 14.28% (1/7) had clear cell carcinoma, and 14.28% (1/7) had borderline serous tumors. None of the cases of borderline mucinous tumors, mucinous carcinoma, and undifferentiated carcinoma presented associated endometriosis outbreaks (Table 1).
From the point of view of the localization of the ovarian carcinoma at the level of the appendages, there is a higher frequency of the left appendage compared to the right (38% compared to 18%), also the bilateral localization being present in a relatively increased percentage (44%). The presence of endometriosis in the appendages was 10.5% (2/19) for the left one, 33.3% (3/9) for the right one, and 9.09% (2/22) for both appendages.
Two of the seven cases in which the areas of endometriosis were visualized presented also endometriosis with atypia (one case of complex hyperplasia, and the other with cytological atypia in the wall of an endometrial cyst).
Discussion
The prevalence of endometriosis in tumors of the epithelial surface of the ovary was 14% (7/50), falling within the limits of 4.2-29.1% revealed by literature(5,12).
In our study, the average age at the moment of diagnosis was 59.4±11.35 in patients with endometriosis and 59.5±13.4 in patients without associated endometriosis, which is not a significant difference from the statistical point of view (p=0.98). One of the studies reported in the literature identified an average age of 49.93±9.36 in the group of patients with associated endometriosis and 50.18±12.8 in the group of patients without associated endometriosis, which is not a significant difference from the statistical point of view(12).
The association between endometriosis and ovarian cancer is reported in the expert studies under two categories. The transition between endometrial lesion and ovarian carcinoma was present in a percentage comprised between 36% and 42%. In all other cases, the ovarian tumor literally coexisted with endometriosis without the identification of a transition(13,14).
Ogawa et al. reported in their studies 78% of endometriosis cases with atypia associated with ovarian carcinoma. In this study, the transition between endometriosis and ovarian carcinoma was present only in two cases, representing 28.51%(15).
According to the classification proposed by Kurman and Shih, all identified ovarian tumors were classified into two large groups: type I and type II. Type I contains low-grade serous carcinoma, endometrioid carcinomas, clear cell carcinomas, mucinous carcinomas, and transitional type carcinomas, while in type II the ovarian tumors are comprising high-grade serous carcinoma, mixed malignancies and undifferentiated carcinomas(16).
According to the classification of Kurman and Shih, all seven cases of endometriosis in this study were classified as type I ovarian tumors, resulting a p=0.0136, which is statistically significant (Table 1).
Most studies on this topic show that the prevalence of endometriosis is higher in the case of endometrioid carcinomas and especially those with clear cells compared to the mucinous and serous carcinomas(2,5,12,17).
The association of endometriosis is found in up to 31% of endometrioid carcinomas and between 50% and 70% of clear cell carcinomas of the ovaries(10,16,18).
Another study showed that 18.3% of endometriosis cases associated with ovarian carcinomas were type I(2).
This study comprises also an evaluation of the presence or absence of endometriosis in patients with ovarian tumors and the transition between endometriosis outbreaks and ovarian tumors. According to a study by Van Gorp et al., the cases of associated ovarian tumors and areas of endometriosis were classified into three categories: A, B, and C(5).
Category A consists of cases with areas of endometriosis present in the same ovary, with areas of histological transition between endometriosis and ovarian tumor. Category B contains cases with outbreaks of endometriosis without transition between endometriosis and tumor, while category C contains cases with endometriosis present in any other location, for example in the pelvis, the contralateral ovary in extragonadal endometriosis(5).
In our study, from the seven cases of carcinomas presenting endometriosis outbreaks associated, in two cases there were highlighted areas of transition between the benign and the malignant process identified by the criteria listed by Sampson and Scott, and included in category A. From the five cases, the remaining four were classified in category B, and one case in category C.
The presence of areas of endometriosis with atypia was made based on criteria which included the increase in volume or hyperchromatic of nuclei, or contrary, hypochromic nuclei, eosinophilic cytoplasm, branching and agglomeration or stratification, all according to the criteria suggested by LaGrenada and Silverberg(10).
The study is limited due to the low number of cases for some of the histological types.
Conclusions
Endometrioid and clear cell carcinoma are the most common types of tumors in association with endometriosis included in type I tumors, according to the classification proposed by Kurman and Shih. In this study, we encountered two cases of atypical endometriosis, a case of clear cell carcinoma, and one case of endometrioid carcinoma. In both cases, we observed the transition from a preneoplastic lesion to the neoplastic lesion, which makes us think that endometriotic atypia can be considered a precursor to type I carcinomas.
Conflict of interests: The authors declare no conflict of interests.
Bibliografie
1. Banz C, Ungethuem U, Kuban R-J, Diedrich K, Lengyel E, Hornung D. The molecular signature of endometriosis-associated endometrioid ovarian cancer differs significantly from endometriosis-independent endometrioid ovarian cancer. Fertil Steril. 2010 Sep; 94(4):1212–7.
2. Wang S, Qiu L, Lang JH, Shen K, Yang JX, Huang HF, et al. Clinical analysis of ovarian epithelial carcinoma with coexisting pelvic endometriosis. Am J Obstet Gynecol. 2013 May; 208(5):413.e1–5.
3. Munksgaard PS, Blaakaer J. The association between endometriosis and ovarian cancer: a review of histological, genetic and molecular alterations. Gynecol Oncol. 2012 Jan; 124(1):164–9.
4. Worley MJ, Welch WR, Berkowitz RS, Ng S-W. Endometriosis-associated ovarian cancer: a review of pathogenesis. Int J Mol Sci. 2013 Mar 6; 14(3):5367–79.
5. Van Gorp T, Amant F, Neven P, Vergote I, Moerman P. Endometriosis and the development of malignant tumours of the pelvis. A review of literature. Best Pract Res Clin Obstet Gynaecol. 2004 Apr; 18(2):349–71.
6. Kokcu A. Relationship between endometriosis and cancer from current perspective. Arch Gynecol Obstet. 2011 Dec; 284(6):1473–9.
7. Somigliana E, Vigano’ P, Parazzini F, Stoppelli S, Giambattista E, Vercellini P. Association between endometriosis and cancer: a comprehensive review and a critical analysis of clinical and epidemiological evidence. Gynecol Oncol. 2006 May; 101(2):331–41.
8. Sainz de la Cuesta R, Eichhorn JH, Rice LW, Fuller AF, Nikrui N, Goff BA. Histologic transformation of benign endometriosis to early epithelial ovarian cancer. Gynecol Oncol. 1996 Feb; 60(2):238–44.
9. Chene G, Caloone J, Moret S, Le Bail-Carval K, Chabert P, Beaufils E, et al. L’endométriose est-elle une lésion pré-cancéreuse? Perspectives et implications cliniques. Gynécologie Obstétrique Fertil. 2016 Feb 1; 44(2):106–12.
10. LaGrenade A, Silverberg SG. Ovarian tumors associated with atypical endometriosis. Hum Pathol. 1988 Sep; 19(9):1080–4.
11. Clement PB. The pathology of endometriosis: a survey of the many faces of a common disease emphasizing diagnostic pitfalls and unusual and newly appreciated aspects. Adv Anat Pathol. 2007 Jul; 14(4):241–60.
12. Oral E, Ilvan S, Tustas E, Korbeyli B, Bese T, Demirkiran F, et al. Prevalence of endometriosis in malignant epithelial ovary tumours. Eur J Obstet Gynecol Reprod Biol. 2003 Jul 1; 109(1):97–101.
13. Garry R. Is insulin resistance an essential component of PCOS? The endometriosis syndromes: a clinical classification in the presence of aetiological confusion and therapeutic anarchy. Hum Reprod. 2004 Apr; 19(4):760–8.
14. Kvaskoff M, Mu F, Terry KL, Harris HR, Poole EM, Farland L, et al. Endometriosis: a high-risk population for major chronic diseases? Hum Reprod Update. 2015 Aug; 21(4):500–16.
15. Ogawa S, Kaku T, Amada S, Kobayashi H, Hirakawa T, Ariyoshi K, et al. Ovarian Endometriosis Associated with Ovarian Carcinoma: A Clinicopathological and Immunohistochemical Study. Gynecol Oncol. 2000 May 1; 77(2):298–304.
16. Shih I-M, Kurman RJ. Ovarian tumorigenesis: a proposed model based on morphological and molecular genetic analysis. Am J Pathol. 2004 May; 164(5):1511–8.
17. Kondi-Pafiti A, Papakonstantinou E, Iavazzo C, Grigoriadis C, Salakos N, Gregoriou O. Clinicopathological characteristics of ovarian carcinomas associated with endometriosis. Arch Gynecol Obstet. 2012 Feb 1; 285(2):479–83.
18. Terada T. Endometrioid adenocarcinoma of the ovary arising in atypical endometriosis. Int J Clin Exp Pathol. 2012 Oct 20; 5(9):924–7.
Articole din ediţiile anterioare
Impactul endometriozei asupra sarcinii
Endometrioza şi adenomioza sunt patologii care afectează frecvent femeile de vârstă reproductivă. Obţinerea şi menţinerea unei sarcini se fac de mu...
Teorii privind originea şi carcinogeneza carcinomului ovarian epitelial de grad înalt şi implicaţiile lor clinice
Cancerul ovarian este o afecţiune eterogenă, cu prezentare clinică, patogeneză şi histologie diverse, rămânând în continuare una din principalele c...
Dificultăţile de diagnostic al leziunilor endometriozice gastrointestinale cu localizare ileală
Endometriosis is a gynecological disease with a chronic evolution, being common among women of reproductive age, affecting up to 15% of patients.
Profilul de risc clinic asociat cancerului ovarian
Acest studiu a fost efectuat pentru a evalua caracteristicile profilului de risc clinic al pacientelor cu tumori ovariene care au fost tratate chir...