Introduction. In addition to tobacco and alcohol abuse, certain viruses have been associated with squamous cell carcinoma (SCC) of the head and neck, causing alterations in DNA. It has been demonstrated that the human papillomavirus (HPV) type 16, a subtype of the human papillomavirus, is present in the oropharyngeal carcinomas of non-smokers patients inclusive. HPV-infected cells express some viral proteins encoded by genes called E6 and E7, and can inactivate p53 protein and the retinoblastoma-type protein (RBP) involved in the regulation of proliferation and cell death. In all studies, the association was very close in tonsillar SCC, where DNA positivity of HPV type frequently exceeded 50%(1).
Materials and method. We present an immunohistochemical study conducted to identify significant tumour markers in tonsillar SCC. The immunohistochemical study was carried out on 52 cases and we studied markers such as p53, RBP, HPV, transforming growth factor alpha (TGFα) and epidermal growth factor receptor (EGFR).
Results. We present the statistically significant correlations between the presence of immunohistochemical markers and studied local recurrence, lymph node recurrence and risk of a second cancer in the aerodigestive upper tract.
Discussion. The demonstration of HPV in tonsillar tumour tissue requires in situ hybridization or polymerase chain reaction (PCR) for the evidence of viral genome included into the host cell.
Conclusions. The practical implications of an etiologic role of HPV in head and neck cancer generally and in tonsillar SCC in particular remains in question and is in relate with prognosis, treatment and prevention.
Impactul HPV-ului şi al altor markeri tumorali în carcinomul cu celule scuamoase de amigdală – studiu retrospectiv
Impact of HPV and other tumoral markers in tonsillar squamous cell carcinoma (SCC) – a retrospective study
First published: 30 noiembrie 2018
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/Orl.41.4.2018.2116
Abstract
Rezumat
Introducere. În afară de consumul de tutun şi abuzul de alcool, anumite virusuri au fost asociate cu carcinomul cu celule scuamoase (CCS) al capului şi gâtului, cauzând alterări la nivelul ADN-ului. Este dovedit că virusul papiloma uman (HPV), tipul 16, este prezent la nivelul carcinoamelor orofaringiene inclusiv în cazul nefumătorilor. Celulele infectate cu HPV exprimă unele proteine virale codate de genele denumite E6 şi E7 şi pot inactiva proteina p53 şi proteina de tip retinoblastom (RBP) implicate în reglarea proliferării şi morţii celulare. În toate studiile, asocierea a fost foarte mare în carcinomul cu celule scuamoase de amigdală, unde pozitivitatea ADN-ului HPV a depăşit frecvent 50%(1).
Materiale şi metodă. Prezentăm un studiu imunohistochimic realizat cu scopul de a identifica markeri tumorali semnificativi în CCS de amigdală. Studiul imunohistochimic a fost realizat pe 52 de cazuri, fiind studiaţi markeri precum p53, RBP, HPV, factorul de transformare tumorală alfa (TGFα) şi receptorul pentru factorul de creştere epidermică (EGFR).
Rezultate. Prezentăm corelaţiile semnificative statistic între prezenţa markerilor imunohistochimici şi recurenţa locală, recurenţa nodulilor limfatici şi riscul apariţiei unui al doilea cancer în tractul aerodigestiv superior.
Discuţie. Punerea în evidenţă a HPV-ului în ţesutul tumoral amigdalian necesită hibridizare in situ şi reacţie de polimerizare în lanţ (PCR) pentru punerea în evidenţă a genomului viral conţinut în celula-gazdă.
Concluzii. Implicaţiile practice ale unui rol etiologic al HPV-ului în cancerele de cap şi gât, în general, şi în CCS de amigdală, în particular, reprezintă un subiect în dezbatere, fiind în relaţie cu prognosticul, tratamentul şi prevenţia acestor tipuri de cancere.
Introduction
The tonsillar squamous cell carcinoma (SCC) is becoming a public health problem because of its rising incidence in the last 20 years, in contrast to the decreasing incidence of carcinomas in other subsites of head and neck associated to the reduced prevalence of smoking. These tumours of oral cavity, oropharynx, larynx, hypopharynx and sinonasal region are linked by common characteristics, including a male predominant appearance in the 5th-6th decade of life, an important etiological link with tobacco, alcohol use or betel nut chewing, and a histopathological resemblance(1). Approximately 90% of the head and neck cancers are squamous cell carcinomas (HNSCC)(2).
Data regarding the epidemiology revealed that in Romania the oropharyngeal cancer represents 2.5% of all neoplasms (the seventh place), with a mortality of 5.78/100,000 of habitants (the ninth place)(3). Other data of epidemiology show that, in the USA, 31% of HNSCC were located in the oropharynx in 2004, compared to 18% in 1973. In Sweden, the proportion of HPV-positive oropharyngeal cancer has increased from 23% in 1970 to 93% in 2007. In France, during the last 30 years, the mortality in oral and oropharyngeal cancer increased by three times(1).
Most studies demonstrated that features like tobacco and alcohol consumption are major common risk factors for HNSCC, but over the last 10-15 years, the HPV infection has been increasingly recognized as a major etiological factor for a type of HNSCC, including oropharyngeal SCC (OPSCC)(4). The high-risk oncogenic types HPV 16 and HPV 18 are both recognized as initiators of over 90% of the cervical cancers, 70% of the anogenital cancers, 5% of the non-oropharyngeal SCC and 20-72% of OPSCC(5).
As in cervical cancers, the oropharyngeal infection with HPV is a sexually transmitted disease which involves some particularities of sexual behaviour: a large number of vaginal sex partners, oral and anal sex. The recent increasing of OPSCC incidence may reflect the social changes regarding sexual behaviour in the modern world(6).
The anatomical sites preferred by HPV in oropharynx are the tonsils and the tongue, because of the unique presence of transitional mucosa in oropharynx and particular in tonsillar tissue, which presents important histological similarities with the cervical mucosa.
HPV 16 – which is involved in 90-95% of all the oropharyngeal HPV associated-cancers – can survive in tonsillar crypts epithelium(7). Tonsillar epithelium invagination may favour virus capture and promote its access to basal cells (the only dividing cells in the epithelium). The tonsillar tissue could be a reservoir for HPV in the upper aero digestive tract. The persistence of HPV in tonsillar tissue might be important for the immune response to HPV(1).
We had two premises for our study on tonsillar cancers. The first consists in the strong association between HPV (particularly HPV 16) with tonsillar cancer. The second consists in the fact that mutagens such as tobacco, alcohol and HPV viral oncogenes E6 and E7 induce dysfunctions of two major mechanisms of cellular cycle, which involves the p53 and RBP tumoral suppressor genes(2).
Materials and method
We made an immunohistochemical retrospective study between 2000 and 2010, aiming to identify any correlations between tumoral markers and the evolution and prognosis in tonsillar SCC.
Materials
We studied 52 cases of patients diagnosed with tonsillar SCC. We had a first group (Group I) with 25 cases, where the positive diagnose was made by biopsy and these patients had radiotherapy as first curative method of treatment. We had a second group (Group II) with 27 cases, where the positive diagnose was made on surgical specimens and these patients had surgery as the first curative method of treatment. The two groups were similar regarding age and gender distribution.
Method
We used for the immunohistochemical study some tumoral markers associated with tonsillar SCC: p53, RBT, HPV, EGFR. The dilutions and markers specifications are revealed in Table 1. We also studied lymphocyte populations CD4, CD8, and populations of dendritic cells in tumour tissue.
For the immunohistochemical identification of tumoral antigens we used the three-stadial indirect method Avidine-Biotine-Peroxidase (ABP), after Hsu and colab., modified by Bussolati and Gugliotta, completed in some cases with signal magnification by Tyramine (Sigma 2000 method, after Kerstens and colab).
Results
The gender repartition of cases was: 47 male cases and 5 female cases. The age repartition of cases was: two cases between 31-40 years old, 14 cases between 41-50 years old, 21 cases between 51-60 years old, 10 cases between 61-70 years old, and five cases between 71-80 years old.
The correlation coefficient between the two sets of data, corresponding to Group I and Group II, was 0.9871, so we interpreted together the result obtained form Group I and Group II.
In both groups, we had 48 smoker patients, representing 92.3% of cases, and 46 alcohol consumers, representing 88.46%. The patients who were both smokers and alcohol consumers represented 82.69 % of cases (43 patients).
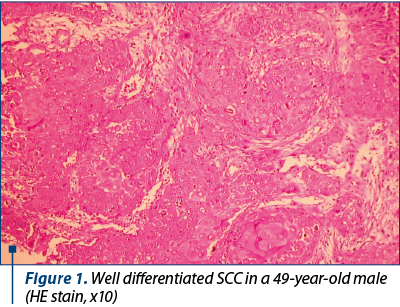
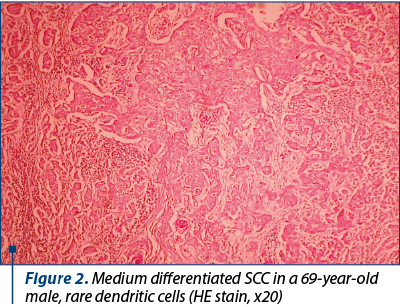
We studied the tumoral markers on 52 cases of squamous cell carcinoma. Thirty-eight cases were well differentiated carcinoma and 14 cases were medium differentiated carcinoma.
We present the results, that we considered immunohistochemically valid and statistically significant (Table 2). We verified the statistic hypothesis for p=0.02 by Student test application for afferent statistic series.
We realised a correlation between the presence of the tumoral marker of a certain type (positive and slowly positive results) and the post-therapeutic evolution – local recurrence, nodal relapse, the occurrence of second cancers in upper aerodigestive upper ways and distance metastases. We have had patients who had more than one recurrence in the same time.
Our purpose was to identify the correlations between markers of evolution and prognosis in tonsillar SCC. Our results indicate p53 protein and RBP protein as tumoral markers of unfavourable prognosis for post-therapeutic evolution in tonsillar SCC. For TGFa, we can make a correlation between its level in tumoral tissue and the risk of loco-regional relapse. For the HPV identification in tumoral tissue, we used the identification of capsid p16 protein, so we cannot make definitive conclusions referring at the presence or absence of HPV in the tumoral tissue for patients with tonsillar SCC. But we realised a correlation between the presence of HPV and the type of post-therapeutic evolution (Figures 1-5).
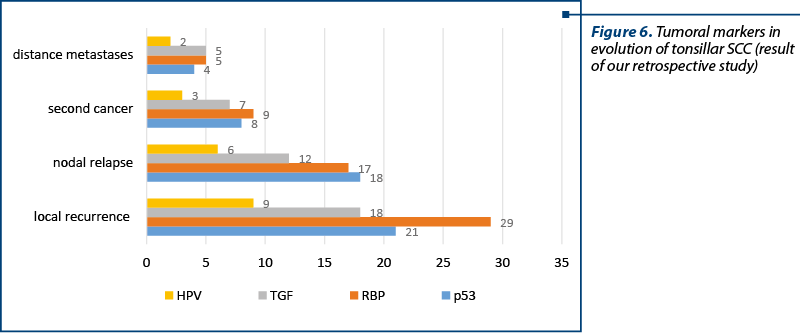
The presence of p53 protein (36 positive and slowly positive cases) was associated with local recurrence in 21 cases (58.33%), 18 nodal relapse cases (50%), the occurrence of a second cancer in upper aerodigestive ways in eight cases (22.22%) and the occurrence of distance metastases take in four cases (11.11%).
The presence of RBP protein (48 positive and slowly positive cases) was associated with local recurrence in 29 cases (60.42%), 17 nodal relapse cases (35.41%), the occurrence of a second cancer in upper aerodigestive ways in nine cases (18.75%) and the occurrence of distance metastases take in five cases (10.41%).
The presence of TGF protein (41 positive and slowly positive cases) was associated with local recurrence in 18 cases (43.90%), 12 nodal relapse cases (29.26%), the occurrence of a second cancer in upper in seven cases (17.07%) and the occurrence of distance metastases take in five cases (12.19%).
The presence of HPV capsid protein (14 positive cases) was associated with local recurrence in nine cases (57.14%), six nodal relapse cases (42.85%), the occurrence of a second cancer in upper aerodigestive ways in three cases (21.43%) and the occurrence of distance metastases take in two cases (14.29%) (Figure 6).
From our data, we can certify as prognostic factors in tonsillar SCC: T stage, N stage, performing or not an elective type of clinical negative neck N0, type of neck dissection, the total dose of radiotherapy. We can propose the positivity in tumour tissue of tonsillar SCC p53 protein, RBP protein and TGF alpha as tumoral markers of unfavourable prognosis.
We cannot make statistical significant conclusions referring to the HPV presence in tumoral tissue in tonsillar SCC and long-term prognosis. Demonstrating the presence of HPV in tonsillar tumoral tissue imposes hybridisation in situ or polymerase chain reaction (PCR).
Discussion
Slaughter et al. proposed the “cancerization field”, a theory based on carcinogen-induced cytological modifications in aerodigestive upper ways mucosa. They explained the greater risk for multiple primary cancers(8). This theory is sustained by the higher incidence of a second cancer in patients with oropharyngeal carcinoma – the risk is about 4% for every year, increasing with 25% after 10 years. Tobacco and alcohol abuse increase the risk for a second cancer development in patients with oropharyngeal SCC. The risk to develop the second cancer in patients who quit smoking after the first lesion treatment is 1/6 of the risk of the pacients who continue to smoke(8). Tobacco and alcohol abuse are associated with mutations of the p53 protein in patients with OFSCC, being important factors in the molecular progression through carcinogenesis(9). Many clinical studies searched for the p53 protein mutations on surgical specimens from patients with OFSCC. The patients with surgical positive edges for p53 protein mutations have a higher risk of local relapse(10).
P53 protein mutations are involved in the loco-regional failure at OFSCC tonsillar with curative radiotherapy(11). The oncogenic nature of the high-risk types of HPV (HPV 16 and HPV 18) is due to the transforming properties of HPV oncoproteins E6 and E7, which target the p53 and RBP proteins tumour suppressor pathways, making the infected cells susceptible to mutations and cancer formation(1,2). In all studies, the association was very straight in tonsillar SCC, where the positivity of DNA of HPV type was frequent in over 50% of cases(4). HPV 16 is the most common HPV type associated with tonsillar SCC. The HPV 16 DNA was identified only in primary tumour cells and in their metastases in similar manner with cervical cancer(3). The presence of HPV 16 genome was revealed by polymerase chain reaction (PCR) or the method of hybridisation in situ, which certified the presence of viral genome included in host cell genome(12).
In our study is important to note that more than 83% of the patients were both smokers and alcohol consumers, so it is difficult to delimitate the genetic alterations induced by these mutagens, on one hand, and HPV16 infection, on the other hand. It is necessary to make a study on HPV tonsillar infection in non-smokers and non-alcoholic consumers.
Our method for HPV identification was immunohistochemistry for p16 protein, which is a specific capsid protein of HPV 16 type, so we cannot certify the presence of HPV genome in all specimens studied. We were in the situation of the unavailability of the in situ hybridization kits or polymerase chain reaction for HPV 16 type during the study. We have to mention the high cost for identifying viral markers.
We can ask ourselves which are the practical implications of the etiological role of HPV in HNSCCC in general and OFSCC in particular. The prognosis of HNSCC hasn’t increased significantly in the last 20 years, despite the understanding of tumoral invasion biology, the progress of surgical techniques and the development of combined therapies(13). Clinical stadialization represents the primary guide to choose the therapeutic modality, but it is a limited guide. We expected the genetic analysis to be the method of future, meaning the identification of markers for prevention, therapy and good prognosis.
The biology of HPV-positive OPSCC showed that in OPSCC associated with HPV infection, the degradation of p53, RBP downregulation and p16 upregulation were present. In OPSCC associated with tobacco and alcohol use, we have mutations of p53, RBP upregulation and p16 downregulation(1). Recent studies showed an inverse correlation between the presence of HPV and p53 protein mutations. We have now some arguments for a better prognosis in HPV-positive OPSCC(14). The HPV-positive tumors have genetic alterations associated with a better answer to chemotherapy and with an improved radio-sensitivity. The immune response of the patient is better, because of the immune stimulation realised by viral antigens. We don’t have cancerisation field in HPV-positive tumors in non-smoker patients. The younger age with less comorbidities may contribute to a better prognosis(15).
The perspectives in OPSCC are preventive and we are talking about vaccination. Prophylactic vaccination is not efficient in already diagnosed infections and in malignant lesions, so it is necessary to study the efficiency of therapeutic HPV vaccination in the treatment of HPV-associated cancer(1). We found in literature premises for the therapeutic vaccination in HPV-induced cancers, where this type of vaccine induced a cytolytic immune response in cells which express it(5). It is necessary to study the carriage of HPV in apparently normal tonsillar tissue, to have screening programmes and to select the patients at risk for OFSCC(16).
Conclusions
The prognosis in tonsillar SCC is mediocre, due to local recurrence, nodal relapse, the occurrence of second cancers in upper aerodigestive upper ways(17) and to distance metastases.
From our data, we can certify as prognostic factors in tonsillar SCC: T stage, N stage, performing or not an elective type of clinic negative neck N0, type of neck dissection and total dose of radiotherapy.
We propose the positivity in tumor tissue of tonsillar SCC p53 protein, RBP protein, and TGF alpha as tumoral markers of unfavourable prognosis.
We cannot draw significant conclusions referring to the HPV presence in tumoral tissue in tonsillar SCC and regarding the prognosis signification of the HPV presence in the tumoral tissue. Demonstrating the presence of HPV in tonsillar tumoral tissue imposes hybridisation in situ or PCR. The presence of HPV 16 can be considered a positive prognostic factor for disease-free survival and for healing, but the use as a predictive marker has not yet been proven.
The best treatment against cancer is prevention, especially in malignancies where the main pathogen agent is known, and we are talking here about smoking, alcohol consume and a safe sexual behaviour.
Ethical approval: All authors hereby declare that all experiments have been examined and approved by the appropriate ethics committee and have therefore been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki.
Acknowledgment: We would like to acknowledge the contribution of professor Carmen Ardelean from “Victor Babeş” Virology Institute, Bucharest, Romania. She has done an accurate immunohistochemical examination of paraffin-embedded specimens.
Conflict of interests: The authors declare no conflict of interests.
Bibliografie
- Elrefaey S, Massaro M, Chiocca S, Chiesa F, Ansarin M. Hpv In Oropharyngeal Cancer: The Basics To Know In Clinical Practice. Acta Otorhinolaryngol Italica. 2014; 34: 299-309.
- Mork J, Lie Ak, Glattre E et al. Human papillomavirus infection as a risk factor for squamous-cell carcinoma of the head and neck. New England Journal of Medicine. 2001; 344(15), 1125-31.
- Boscolo-Rizzo P, Del Mistro A Büssü F, et al. New insights into human papillomavirus-associated head and neck squamous cell carcinoma. Acta Otorhinolaryngol Italica. 2013 Apr; 33(2):77-87.
- Hemminki K, Dong C, Frisch M. Tonsillar and other upper aerodigestive tract cancers among cervical cancer patients and their husbands. European Journal of Cancer Prevention. 2000, 9, 433-7.
- Mannarini L, Kratochvil V, Calabrese L, et al. Human Papilloma Virus (HPV) in head and neck region: review of literature. Acta Otorhinolaryngol Italica. 2009 Jun; 29(3): 119–126.
- Grasso F, Negri DR, Mochi S, et al. Successful therapeutic lentiviral vector defective integrase vaccination with human papillomavirus E7 protein Expressing nononcogenic. Int J Cancer. 2013(132): 335-44
- Slaughter DP, Southwick HW, Smejkal W. „Field cancerization” in oral stratified squamous ephitelium: clinical implications of multicenter origin. Cancer. 1953, 6: 963-968.
- Somers K, Merrick MA, Lopez ME, Incognito LS, Schechter GL, Casey G. Frequent p53 mutations in head and neck cancer. Cancer Res. 1992, 52: 5997-6000.
- Jonhson RE, Sigman JD, Funk GF et al. Quantification of surgival margin shrinkage in the oral cavity. Head and Neck. 1997, 19: 281-286.
- Koch WM, Brennan JA, Zahurak M, et al. p53 mutation and locoregional treatmnet failure in head and neck squamous cell carcinoma. Journal of National Cancer Institute. 1996, 88: 1580-1586.
- Mellin H, Friesland S, Auer G, Dalianis T, Munck-Wikland E. Human papillomavirus and DNA ploidy in tonsillar cancer – correlation to prognosis. Anticancer Researches. 2003, 23: 2821-2828.
- Wolf GT, Carey TE, Schmaltz SP, et al. Altered antigen expression predicts outcome in squamous cell carcinoma of the head and neck. Journal of National Cancer Institute. 1990, 74: 1566-1572.
- Mork J, Lie AK, Glattre E, et al. Human papillomavirus infection as a risk factor for squamous-cell carcinoma of the head and neck. New England Journal of Medicine. 2001; 344 (15): 1125-1131.
- Li W, Thompson C, O’Brien C, et al. Human papillomavirus positivity predicts favourable outcome for squamous carcinoma of the tonsil. International Journal of Cancer. 2003, 106: 553-558.
- Niedobitek G, Pitteroff S, Herbst H, et al. Detection of human papillomavirus type 16 in carcinoma of the palatine tonsil. Journal of Clinical Pathology. 1990, 43: 918-921.
- León X, Quer M, Diez S, et al. Second neoplasm in patients with head and neck cancer. Head Neck. 1999; 21 (3): 204-210.
Articole din ediţiile anterioare
Strategii de dezescaladare ale tratamentului oncologic pentru cancerul de orofaringe HPV-pozitiv
Cancerele orofaringiene HPV-pozitive prezintă o incidenţă crescută în ultimele decenii, în special în rândul adulţilor tineri nefumători.
HPV – schiţă a stilului său de viaţă
Papilomavirusurile umane (HPV) sunt virusuri mici neînvelite cu ADN icosaedric, adaptate fiinţelor umane chiar de la început, de la apariţia prim...