Cancerul colorectal în sarcină
Colorectal cancer during pregnancy
Abstract
The incidence of colorectal cancer during pregnancy is reduced, being estimated at approximately one in 1000 preganancies. Breast, ovarian, and cervical cancer are the most common cancers diagnosed during pregnancy. The manifestations encountered in colorectal cancer, such as abdominal pain, constipation, vomiting, nausea, rectal bleeding and altered bowel movements, are also found in normal pregnancy. In this paper, we present a case of colorectal cancer with hepatic metastasis diagnosed in a 36-year-old preganant woman (IIG, 1P), at 32 weeks of pregnancy.Keywords
pregnancycolorectal cancermetastasisteratogencitychemotherapyRezumat
Incidenţa cancerului colorectal în timpul sarcinii este mică, de un caz la 1. 000 de sarcini. Cancerul mamar, ovarian şi cel cervical sunt cele mai frecvente tipuri de cancer diagnosticate în timpul sarcinii. Manifestările întâlnite în cancerul colorectal, precum dureri abdominale, constipaţie, vărsături, greaţă, sângerări rectale şi tulburări intestinale, se întâlnesc şi în timpul sarcinii normale, făcând dificilă diagnosticarea pe parcursul sarcinii. În această lucrare vă prezentăm cazul unei paciente de 36 de ani (IIG, 1P), în săptămâna a 32-a de sarcină, diagnosticată cu cancer colorectal, prezentând şi metastaze hepatice.Cuvinte Cheie
cancer colorectalmetastazeteratogenitatechimioterapieIntroduction
The incidence of colorectal cancer during pregnancy is reduced, being estimated at approximately one in every 1000 preganancies(1-5). Breast, ovarian and cervical cancer are the most common cancers diagnosed during pregnancy(2). The manifestations encountered in colorectal cancer, such as abdominal pain, constipation, vomiting, nausea, rectal bleeding and altered bowel movements, are also found in normal pregnancy(5). Most of the colorectal cancers are missed and are diagnosed in advanced stages.
Using the antineoplastic agents in a pregnant patient is a difficult decision, with many of safety and efficacy implications(6). The treatment plan depends on the desire of the pregnant woman, the stage of the disease, the possible teratogenic effects of the antineoplastic agents and abortion(7-9). We present in this paper a case of colorectal cancer with hepatic metastasis diagnosed in a patient at 32 weeks of pregnancy.
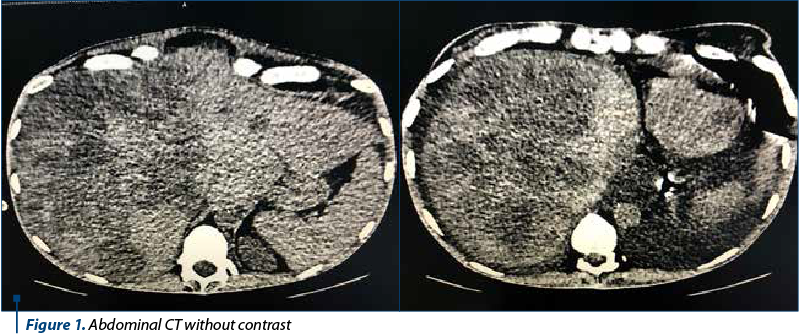
Case report
A 36-year-old preganant woman (IIG, 1P) complained of abdominal pain that persisted throughout her pregnancy. She also had constipation and anemia.
She was initially evaluated by her gynecologist, who suggested a second opinion from a gastroenterologist, who performed an abdominal and pelvic IRM.
The abdominal and pelvic IRM evaluation revealed global hepatomegaly (262 mm cranio caudal right lobe, 97 mm antero-posterior left lobe), and more lobular contour space replacement formation. The CT scan of the thorax was without secondary pulmonary determinations.
There was no family history of cancer. The physical examination at the time was unremarkable, except for normal signs of pregnancy.
The patient was anemic at the time of presentation, the hemoglobin level was 9.10, and the renal and hepatic functions were within normal limits. The colorectal markers CEA and CA19-9 were 959 ng/ml and 1018U/ml, respectively.
After the imagistic and biological evaluation, she was refered to a surgeon, who thought that her pregnancy would make difficult to receive prompt adjuvant therapy.
The patient was only 32-week pregnant at the time of diagnosis. A caesarean section was performed, resulting in the birth of a premature baby, weighing 1750 g, who received an Apgar score of 9. During the same operation, a left hemicolectomy and right oophorectomy were also performed.
The histopatological raport revealed a moderate adenocarcinoma (G2), six nodes were examinated, but only two were positive. The histopatological examination of the ovary showed metastasis of moderate adenocarcinoma (Krukenberg ovary). Her disease was patologically classified as stage pT4, Pn1Mx, and clinically as stage IV.
The IHC test showed moderate adenocarcinoma of the colon, CK7(-), CK(+), ER(-), PR, Vimentin (+) and Ki67 (+) 90%. She performed BRAF examination, that identified the mutation of BRAF V600. Mutations in K-RAS wild-type and N-RAS wild-type were also present.
Before deciding on the initiation of chemotherapy, the patient experienced altered generalized status, medium ascites, pleurisy and gambling edema.
She received a cycle of adjuvant chemotherapy consisting of cetuximab 600 mg. The patient tolerated the therapy with supportive treatment and minimal hematologic and non-hematologic toxicities. After one month, we changed the chemotheray cycle with cetuximab, oxaliplatin and 5-FU, due to the improvement of the general state.
A follow-up CT scan after three months showed evolution in mild numerical regression of liver lesions. The hepatomegaly was maintained. Fine fluid blade in dimensional regression. Without bone metastases.
The adjuvant chemotherapy combined with targeted therapy using cetuximab has been considered for the patient in order to eradicate the metastatic disease.
Discussion
Despite the low incidence rate of 0.07% to 0.1%, cancer accounts as a leading cause of death in women at childbearing ages(1-5). Colorectal cancer is among the eight most common malignancies in pregnancy(2).
Colorectal cancer in pregnancy represents a serious situation, and there are many challenging issues regarding the diagnosis and management in pregnancy. Because the signs and simptoms are similar in pregnancy and colorectal cancer, the colorectal cancer can be concealed(10). In our patient, the abdominal pain was misdiagnosed as a sign and simptom of a normal pregnancy. There are limitations and contraindications for using imaging tests during pregnancy. Ultrasound, a safe images test, has limited accuracy in detecting colorectal cancer, and colonoscopy – which is the gold standard test for diagnosing colorectal cancer – can’t be used in pregnancy because of its complications(11-13). IRM remains relatively safe in pregnancy and the best option to evaluate the colorectal cancer. Serum CEA is an important test used in the evaluation of patients with colorectal cancer(14). CEA levels during pregnancy may be elevated and can be used for monitoring the response of the treatment(15,16).
Due to the fact that colorectal cancer is encoutered in the eldery and rarerly in young patients, it is assumed that there are a number of predisposing factors, such as Lynch syndrome, Peutz-Jeghers syndrome and inflammatory bowel disease(17).
Another challenging issue is the treatment, which should be started as early for the mother, and is based on the gestational age and tumor stage. If diagnosis occurs during the first trimester of pregnancy, it may be expected until the fetus becomes viable, but with significant tumor progression(18,19). If the diagnosis occurs after the 20th week of pregnancy, colon resection may be delayed.
Chemotheray is safer in the second and third trimesters, when organogenesis is complete(20). We can’t use 5-fluorouracil and oxaliplatin in the first trimester because of their teratogenic effects. There are no human data for the effect of cetuximab in pregnancy(21).
The ovarian metastatic disease from colorectal cancer is another challenge. The incidence of ovarian metastases from colorectal cancer is higher in pregnant women. The survival of the pregnant woman with ovarian metastases is poor. Nesbitt et al. recommend obtaining a biopsy of the ovaries during surgery(22).
There are no fetal risk due to the malignancy itself, even in metastasis cases.
The pregnant woman with colorectal cancer has a poor prognosis. A review of 15 cases revealed that all patients with colorectal cancer presented in stage IV, and these patients died in 12 months after delivery(23).
Conclusions
Colorectal cancer in pregnancy represents a diagnostic and therapeutic challenge. Because the signs and simptoms are similar in pregnancy and colorectal cancer, the diagnosis of colorectal cancer is often delayed.
The therapeutic decison for a pregnant patient with colorectal cancer should involve a multidisciplinary team, and there must be taken into account the life of the unborn child and the survival of the mother.
Conflict of interests: The authors declare no conflict of interests.
Bibliografie
- Girard RM, Lamarche J, Baillot R. Carcinoma of the colon associated with pregnancy: report of a case. Dis Colon Rectum. 1981; 24(6):473–5.
- Chêne G, Tardieu AS, Favard A, Lebel A, Voitellier M. Colorectal cancer discovered during pregnancy. J Gynecol Obstet et Biol Reprod. 2006; 35(5, pt 1):504–12.
- Salani R, Billingsley CC, Crafton SM. Cancer and pregnancy: an overview for obstetricians and gynecologists. Am J Obstet Gynecol. 2014; 211:7–14
- Pavlidis N. Cancer and pregnancy: What should we know about the management with systemic treatment of pregnant women with cancer? Eur J Cancer. 2011; 47(suppl 3):S348–S352.
- Koren G, Carey N, Gagnon R, et al. Cancer chemotherapy and pregnancy. J Obstet Gynaecol Can. 2013; 35:263–80.
- Cardonick E, Iacobucci A. Use of chemotherapy during human pregnancy. Lancet Oncol. 2004; 5:283–91.
- Selig BP, Furr JR, Huey RW, et al. Cancer chemotherapeutic agents as human teratogens. Birth Defects Res A Clin Mol Teratol. 2012; 94:626–50.
- Amant F, Han SN, Gziri MM, et al. Chemotherapy during pregnancy. Curr Opin Oncol. 2012; 24:580–6.
- Friedman JM, Weber Schöndorfer C. Antineoplastic drugs. In: Schaefer C, Peters PWJ, Miller RK, ed. Drugs During Pregnancy and Lactation: treatment options and risk assessment. 3rd ed. Amsterdam: Academic Press; 2015; 374–99.
- Pigeau H, Dupré PF, Benouna J et al. Management of rectal cancer in pregnant women. Bull Cancer. 2005; 92(11):953–8.
- Mechery J, Ikhena SE. Cancer of the descending colon during pregnancy. J Obstet Gynaecol. 2007; 27(3):311–2.
- Harma M, Harma M, Uzunkoy A. Colorectal cancer presenting with uncommon soft tissue invasion during pregnancy. Acta Obstet Gynecol Scand. 2005; 84(5):491–3.
- Chourak M, Tahiri MH, Majbar A et al. Colorectal cancer discovered during pregnancy. Gastroenterol Clini Biol. 2009; 33(12):1118–23.
- Colecchia G, Nardi M. Colorectal cancer in pregnancy. A case report. G Chir. 1999; 20(4):159–61.
- Bernstein MA, Madoff RD, Caushaj PF. Colon and rectal cancer in pregnancy. Dis Colon Rectum. 1993; 36(2):172–8.
- Cappell MS. Gastrointestinal endoscopy in high-risk patients. Dig Dis. 1996; 14(4):228–44.
- Brent RL. The effect of embryonic and fetal exposure to X-ray, microwaves, and ultrasound: counseling the pregnant and nonpregnant patient about these risks. Semin Oncol. 1989; 16(5):347–68.
- Nies C, Leppek R, Sitter H et al. Prospective evaluation of different diagnostic techniques for the detection of liver metastases at time of primary resection of colorectal carcinoma. Eur J Surg. 1996; 162(10):811–6.
- Seidman DS, Heyman Z, Ben-Ari GY, Mashiach S, Barkai G. Use of magnetic resonance imaging in pregnancy to diagnosis intussusception induced by colonic cancer. Obstet Gynecol. 1992; 79(5):822–3.
- Slattery ML, Samowitz WS, Holdenc JA. Estrogen and progesterone receptors in colon tumors. Am J Clin Pathol. 2000; 113(3):364–8.
- Rogers JE, Dasari A, Eng C. The Treatment of colorectal cancer during pregnancy: cytotoxic chemotherapy and targeted therapy challenges. Oncologist. 2016; 21(5):563-70.
- Nesbitt JC, Moise KJ, Sawyers JL. Colorectal Carcinoma in Pregnancy. Arch Surg. 1985; 120(5):636–40.
- Jeppesen JB, Østerlind K. Successful twin pregnancy outcome after in utero exposure to FOLFOX for metastatic colon cancer: A case report and review of the literature. Cl Colorectal Cancer. 2011; 10(4):348-52.
