Asocierea nivelurilor mari de proteină C reactivă cu tulburările de piele la femeile cu istoric ob-stetrical sărac
Association of high levels of C-reactive protein with skin disorders in women having poor obstetric history
Abstract
Aims. The purpose of this study was to demonstrate the association of high levels of C-reactive protein (CRP) with skin disorders in women having poor obstetric history. Methodology. The patients (n=185) were evaluated for the presence of systemic disorders, as well as skin problems and inflammation. Clinical/demographic features and CRP levels were obtained from the electronic database. Skin disorders were grouped as acne/rosacea/other acneiform disorders, fungal disease, pruritus/xerosis, psoriasis vulgaris, acrochordons/other benign hypertrophic skin conditions, urticaria/dermatitis, and viral diseases. Results. High levels of CRP were found in 37.5% and 16.2% of the patients with (n=80) and without (n=105) skin disorders, respectively (p=0.002). CRP levels above the upper limit were more frequently detected in pruritus/xerosis (p=0.005) and acrochordons (p=0.015). Conclusions. Skin disorders should be further evaluated for the existence of inflammation which might be a risk factor for poor gestational outcomes. Preconceptional counseling is beneficial for women with poor obstetric history and skin disorders.Keywords
preconceptional counselinginflammationC-reactive proteinskin disordersobstetric complicationsRezumat
Scop. Obiectivul acestui studiu a fost demonstrarea asocierii dintre nivelurile mari de proteină C reactivă (PCR) şi tulburările de piele la femeile cu un istoric obstetrical sărac. Metodologie. Pacientele, în număr de 185, au fost evaluate pentru prezenţa afecţiunilor sistemice şi a tulburărilor de piele şi inflamaţiilor. Caracteristicile clinice şi demografice, alături de nivelurile PCR au fost obţinute din baza de date electronică. Tulburările de piele au fost grupate în acnee/rosacea/alte tulburări acneiforme, boli fungice, prurit/xeroză, psoriasis vulgaris, acrocordon/alte tulburări de piele benigne hipertrofice, urticarie/dermatită şi infecţii virale. Rezultate. Niveluri mari ale PCR au fost detectate la 37,5% dintre pacientele cu tulburări de piele (n=80), respectiv la 16,2% dintre pacientele fără tulburări de piele (n=105; p=0,002). Nivelurile PCR peste limita superioară a normalului au fost evidenţiate mai frecvent în caz de prurit/xeroză (p=0,005) şi acrocordon (p=0,015). Concluzii. Tulburările de piele trebuie evaluate ulterior pentru existenţa inflamaţiei, care ar putea fi un factor de risc pentru rezultate gestaţionale slabe. Consilierea preconcepţională este benefică în cazul pacientelor cu un istoric obstetrical sărac şi tulburări de piele.Cuvinte Cheie
consiliere preconcepţionalăinflamaţieproteină C reactivătulburări de pielecomplicaţii obstetricaleIntroduction
One of the primary concerns of perinatologists should be the determination of risk factors for adverse pregnancy outcomes, instead of dealing with the management protocols of the obstetrical complications. Prevention of the disorders is much easier than the treatment of the resulting complications. It has been reported that inflammatory, metabolic and immunological factors may cause placental inflammation and related obstetrical complications(1-5). For this reason, “preconceptional care programs” for women with poor obstetric history should focus on systemic disorders, chronic inflammatory diseases, and metabolic/immunological risk factors which may be responsible for end-organ inflammations(6-10).
C-reactive protein (CRP) is a protein made by the liver and its blood levels increase in response to inflammatory processes(11,12). It is used as a blood test marker for inflammation in the body. Increased levels of CRP may be observed in chronic diseases and autoimmune disorders(11,13). The skin is highly sensitive to autoimmune disorders, allergic problems, metabolic diseases, internal organ diseases, paraneoplastic syndromes, bacterial/viral infections, inflammation, or epigenetic factors(14,15). There are different types of skin-related inflammatory diseases, including psoriasis, dermatomyositis, Sjögren syndrome etc.(16,17) Skin disorders might be the sign of inflammatory disorders which are risk factors for poor gestational outcome.
This study investigated the association of high levels of CRP with skin disorders in women having poor obstetric history. Inflammation and related biological events behind skin disorders may be the risk factors for obstetrical complications and poor gestational outcome.
Patients/materials and method
This retrospective study involved 185 women having poor obstetric history/background. Poor obstetric history was defined as having miscarriage, fetal growth restriction, preterm birth, preeclampsia, stillbirth and neonatal morbidity/mortality in previous gestations. All patients were examined and evaluated for the presence of systemic disorders, as well as skin problems and inflammation within a framework of preconceptional care program.
Necessary laboratory tests, including CRP, were performed. Demographic and clinical data were obtained from the electronic database of the Division of Perinatology (2016-2019). Skin disorders were grouped according to their dermatological findings into seven categories: acne/rosacea/other acneiform disorders, fungal disease, pruritus/xerosis, psoriasis vulgaris, acrochordons and other benign hypertrophic skin conditions, urticaria/dermatitis, and viral diseases.
The statistical analysis was performed using the Statistical Package for the Social Sciences (SPSS, version 23). The groups were compared using the Yates’ Chi-square and Fisher’ exact tests. P-values <0.05 were considered statistically significant. The study was approved by the Local Ethic Committee (GO 19/1064). This study was performed in accordance with the Declaration of Helsinki. The written informed consent was obtained from the patients.
Results
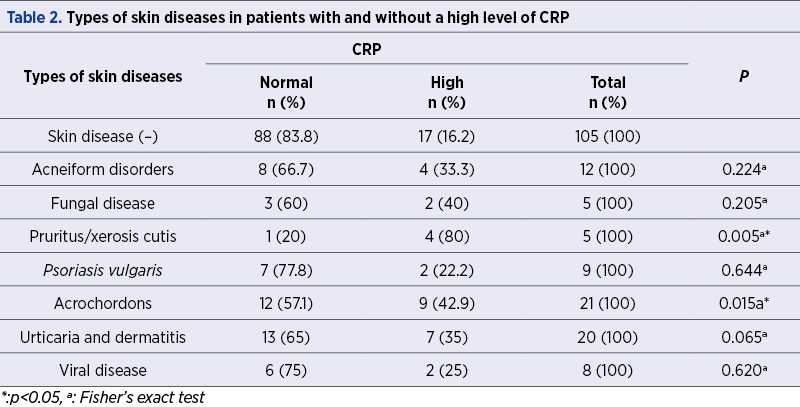
CRP is measured in milligrams of CRP per liter of blood (mg/L). Normal CRP levels are below 3 mg/L. We have demonstrated high levels of CRP (>3 mg/L) in 37.5% and 16.2% of the patients with (n=80) and without (n=105) skin disorders, respectively (p=0.002) – Table 1.
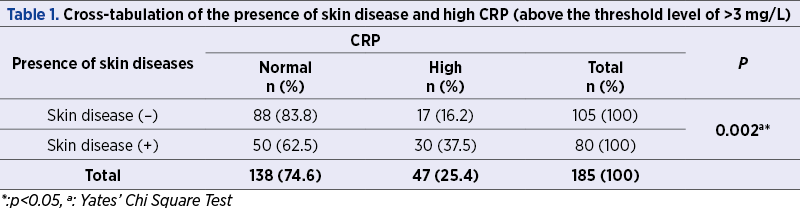
When different skin disease categories were considered, CRP levels above the upper limit were more frequently detected in pruritus/xerosis, acrochordons and other benign hypertrophic skin conditions, fungal disease, and urticaria/dermatitis subgroups (Table 2). However, a statistically significant increase was only found in patients with pruritus/xerosis and acrochordons (p=0.005 and p=0.015).
Discussion
Perinatal medicine aims at reducing obstetrical complications. Etiology-based medical management should be encouraged to achieve better pregnancy outcomes. Metabolic, infectious/inflammatory and immunological factors may injure the cellular structures of intervillous space, leading to placental inflammation(1,6,10). Therefore, physicians should attempt to identify the risk factors leading to placental inflammation and associated obstetrical complications. Thus, inflammation and immune system related biological events behind skin disorders may be the risk factors for placental inflammation and obstetrical complications(1,1,6,14).
CRP is an acute-phase inflammatory protein and exhibits elevated expression during inflammatory conditions(11). There are many factors that may change the baseline CRP levels, including age, gender, smoking, weight, lipid levels and blood pressure(18). CRP is mainly classified as an acute marker of inflammation, but recent studies have indicated the critical roles that CRP plays in inflammatory processes(11). It is primarily synthesized by interleukin-6-dependent hepatic biosynthesis(19). CRP can activate the fluid phase complement pathway (activation of C1q molecule), as well as cell-mediated pathways by activating complement. CRP binds to Fc receptors with the resulting interaction leading to the release of proinflammatory cytokines(11,20,21). CRP is not merely a marker of inflammation, but also plays a role in the inflammatory process(11). High CRP levels have been reported in skin diseases such as urticaria, acne, rosacea etc.(22) This study demonstrated that CRP exhibits elevated expression in cases with certain skin disorders. Such skin disorders associated with immunological problems and inflammation may be the reason for high CRP levels.
Xerosis cutis – known as dry skin – usually accompanies pruritus(23). Pruritus is a sensory reaction in response to cutaneous and systemic diseases(24). Pruritus can also be seen in urticaria/dermatitis. According to Senturk et al., CRP levels were higher in patients with pruritus in a statistically significant manner (p=0.026)(25). Also, higher serum levels of CRP were found to relate to intensity of pruritus in patients with chronic urticaria(26). In our study, high CRP levels were more frequent in patients with pruritus and xerosis cutis, with a rate of 80%. There was also a statistically significant relationship between the presence of xerosis/pruritus and high level of CRP (p=0.005).
Acrochordons (skin tags) are benign skin growths on the surface of the skin(27). Higher levels of CRP were found in patients with acrochordons(28,29). In all, 42.9% of patients with skin tags had elevated CRP levels (p=0.015). Acrochordons are highly associated with metabolic syndrome and related disorders, which is now accepted as an inflammatory condition(30). This is likely an explanation of the high CRP levels in patients with acrochordons.
The limitations of this study are represented by the limited number of patients in skin disease subgroups and by the absence of follow-up results. Our cohort was diagnosed with a wide spectrum of skin disorders and we believe that disorder-specific studies are necessary in this field. Other inflammation markers should also be considered in future studies in order to have better clinical comments.
The patients with skin diseases should be further evaluated in terms of the existence of inflammatory disorders which might be a risk factor for poor gestational outcome. Preconceptional counseling is beneficial for women with poor obstetric history and skin problems.
Bibliografie
-
Beksac MS, Tanacan A, Ozten G, Cakar AN. Low-dose low-molecular-weight heparin prophylaxis against obstetrical complications in pregnancies with metabolic and immunological disorder-associated placental inflammation. J Matern Fetal Neonatal Med. 2020 Apr 30: 1-8. Doi: 10.1080/14767058.2020.1760834.
-
Fadiloglu E, Unal C, Tanacan A, Cagan M, Beksac MS. Effect of hypocomplementemia on perinatal outcomes of pregnancies with autoimmune disorders. Hum Antibodies. 2020;28(2):179-184.
-
Unal C, Fadiloglu E, Tanacan A, Zaim OC, Beksac MS. Retrospective evaluation of pregnancies with ankylosing spondylitis in a tertiary center in Turkey. Int J Rheum Dis. 2020;23(1):101-5.
-
Tanacan A, Fadiloglu E, Ozten G, Gunes AC, Orgul G, Beksac MS. Myasthenia gravis and pregnancy: retrospective evaluation of 27 pregnancies in a tertiary center and comparison with previous studies. Ir J Med Sci. 2019;188(4):1261-7.
-
Gurbuz RH, Atilla P, Orgul G, Tanacan A, Dolgun A, Cakar AN, et al. Impaired placentation and early pregnancy loss in patients with MTHFR polymorphisms and type-1 diabetes mellitus. Fetal Pediatr Pathol. 2019;38(5):376-86.
-
Tanacan A, Beksac MS, Orgul G, Duru S, Sener B, Karaagaoglu E. Impact of extractable nuclear antigen, anti-double stranded DNA, antiphospholipid antibody, and anticardiolipin antibody positivity on obstetrical complications and pregnancy outcomes. Hum Antibodies. 2019;27(2):135-41.
-
Beksaç K, Örgül G, Çağan M, Karaağaoğlu E, Arslan S, Beksaç MS. Retrospective evaluation of pregnant women with celiac disease. J Turk Ger Gynecol Assoc. 2017;18(1):56-9.
-
Beksac K, Orgul G, Can GS, Oktem A, Kav T, Beksac MS. Management of inflammatory bowel disease and pregnancy using prophylactic low dose low molecular weight heparin and corticosteroids. Journal of Clinical and Diagnostic Research. 2017;11(11):1-3.
-
Beksac K, Tanacan A, Cagan M, Dönmez HG, Fadiloglu E, Unal C, et al. Relationship of cholelithiasis and urolithiasis with methylenetetrahydrofolate reductase polymorphisms. J Invest Surg. 2020 Mar 31;1-4. Doi: 10.1080/08941939.2020.1742402.
-
Turgal M, Gumruk F, Karaagaoglu E, Beksac MS. Methylenetetrahydrofolate reductase polymorphisms and pregnancy outcome. Geburtshilfe Frauenheilkd. 2018;78(9):871-8.
-
Sproston NR, Ashworth JJ. Role of C-Reactive Protein at Sites of Inflammation and Infection. Front Immunol. 2018;9:754.
-
Gabay C, Kushner I. Acute-phase proteins and other systemic responses to inflammation. N Engl J Med. 1999;340(6):448-54.
-
Du Clos TW, Mold C. C-reactive protein: an activator of innate immunity and a modulator of adaptive immunity. Immunol Res. 2004;30(3):261-77.
-
Beksac B, Donmez HG. Association of bacterial vaginosis and skin disorders in patients with autoimmune antibody positivity. Gynecol Obstet Reprod Med. 2020. Doi:10.21613/GORM.2020.1103
-
Hu Y, Zhu Y, Lian N, Chen M, Bartke A, Yuan R. Metabolic syndrome and skin diseases. Front Endocrinol (Lausanne). 2019;10:788.
-
Tseng CC, Chang SJ, Tsai WC, Ou TT, Wu CC, Sung WY, et al. Sex differential association of dermatomyositis with Sjögren syndrome. CMAJ. 2017;189(5):E187-E193.
-
Ayala-Fontánez N, Soler DC, McCormick TS. Current knowledge on psoriasis and autoimmune diseases. Psoriasis (Auckl). 2016; 6: 7-32.
-
Hage FG, Szalai AJ. C-reactive protein gene polymorphisms, C-reactive protein blood levels, and cardiovascular disease risk. J Am Coll Cardiol. 2007;50(12):1115-22.
-
Baumeister D, Akhtar R, Ciufolini S, Pariante CM, Mondelli V. Childhood trauma and adulthood inflammation: a meta-analysis of peripheral C-reactive protein, interleukin-6 and tumour necrosis factor-α. Mol Psychiatry. 2016;21(5):642-9.
-
Pradhan AD, Manson JE, Rifai N, Buring JE, Ridker PM. C-reactive protein, interleukin 6, and risk of developing type 2 diabetes mellitus. JAMA. 2001;286(3):327-34.
-
Du Clos TW. Function of C-reactive protein. Ann Med. 2000;32(4):274-8.
-
Namazi MR, Parhizkar AR, Jowkar F. Serum levels of hypersensitive-C-reactive protein in moderate and severe acne. Indian Dermatol Online J. 2015;6(4):253-7.
-
Norman RA, editor. Diagnosis of Aging Skin Diseases. London: Springer; 2008.
-
Kremer AE, Feramisco J, Reeh PW, Beuers U, Oude Elferink RP. Receptors, cells and circuits involved in pruritus of systemic disorders. Biochim Biophys Acta. 2014;1842(7):869-92.
-
Senturk N, Ozkaya O, Aytekin S, Bek K, Açikgöz Y, Aydin F, et al. Characteristics of pruritus in children on peritoneal dialysis. Nephron Clin Pract. 2008;109(3):c168-72.
-
Czarnecka-Operacz M, Szulczyńska-Gabor J, Leśniewska K, Teresiak-Mikołajczak E, Bartkiewicz P, Jenerowicz D, et al. Acute-phase response and its biomarkers in acute and chronic urticaria. Postepy Dermatol Alergol. 2018;35(4):400-7.
-
Luba MC, Bangs SA, Mohler AM, Stulberg DL. Common benign skin tumors. Am Fam Physician. 2003;67(4):729-38.
-
El Safoury OS, Ezzat M, Abdelhamid MF, Shoukry N, Badawy E. The evaluation of the impact of age, skin tags, metabolic syndrome, body mass index, and smoking on homocysteine, endothelin-1, high-sensitive c-reactive protein, and on the heart. Indian J Dermatol. 2013;58(4):326.
-
Hegazy SK, El-Ashmawy NE. Leptin and c-reactive protein are implicated in the pathogenesis of skin tags. Journal of Diabetes Research and Clinical Metabolism. 2013;2:13.
-
Reddy P, Lent-Schochet D, Ramakrishnan N, McLaughlin M, Jialal I. Metabolic syndrome is an inflammatory disorder: A conspiracy between adipose tissue and phagocytes. Clin Chim Acta. 2019;496:35-44.