Sarcina şi naşterea în timpul pandemiei de COVID-19. Implicaţii pentru gravidă şi naştere
Pregnancy and childbirth during the COVID-19 pandemic. Implications for pregnant women and childbirth
Abstract
SARS-CoV-2, causing an ongoing pandemic, has infected until now more than six million people and is responsible for the death of 370,657 people worldwide (as of June 1, 2020). The emerging infections in the past have had an impact on pregnant women, with greater risks during pregnancy, such as the 2009 pandemic H1N1 influenza virus or SARS and MERS outbreaks, along with the teratogenic effects of the Zika virus during the 2015-2016 Zika virus epidemic. Viral pandemics threaten the general population, including pregnant women. During pregnancy, the maternal immune system must keep a delicate balance between the tolerance of the fetus and the protection against harmful antigens such as viral infections. Pregnancy has been associated with more severe symptoms in respiratory viral infections, especially in the third trimester. Our objective was to present data regarding the impact that the ongoing COVID-19 epidemic has on pregnant women, childbirth and on postpartum care and breastfeeding.Keywords
COVID-19SARS-CoV-2pregnancynewbornbreastfeedingdeliveryRezumat
SARS-CoV-2, responsabil de actuala pandemie, a infectat până acum mai mult de 6 milioane de persoane şi este responsabil de 370.657 de decese în lume (până la 1 iunie 2020). Infecţiile nou apărute în trecut au avut un impact negativ asupra gravidelor, cu riscuri în timpul sarcinii, cum ar fi pandemiile cu virusul influenza H1N1, virusurile SARS sau MERS, dar şi efectele teratogene ale virusului Zika, în special în timpul epidemiei din 2015-2016. Pandemiile au efecte asupra populaţiei generale, incluzînd şi populaţia gravidelor. În timpul sarcinii, sistemul imunitar matern trebuie să susţină balanţa delicată dintre toleranţa fătului şi protecţia împotriva antigenelor din exterior, ca de exemplu infecţiile virale. Sarcina a fost asociată cu simptome mai severe ale infecţiilor respiratorii virale, mai ales în trimestrul al treilea. Obiectivul nostru a fost să prezentăm date legate de impactul actual pe care îl are epidemia de COVID-19 asupra sarcinii, naşterii, alăptării şi îngrijirilor postnatale.Cuvinte Cheie
COVID-19SARS-CoV-2sarcinănou-născutalăptarenaştereIntroduction
In late December 2019, a series of pneumonia cases of unknown cause appeared in Wuhan City, Central China, which were reported to the World Health Organization (WHO) on December 31, 2019. Two months later, due to the rapid increase in the number of cases and the global spreading from Asia to the Middle East, Europe and the United States, WHO declared COVID-19 outbreak a pandemic on March 11th, 2020. The ongoing pandemic that has affected 216 countries and infected over six million people is caused by a novel coronavirus, SARS -CoV-2(1).
Classification and origin of the virus
SARS-CoV-2 is a virus of the order Nidovirales, family Coronaviridae, subfamily Coronavirinae. The subfamily is divided into Alphacoronavirus (aCoV) and Betacoronavirus (bCoV), that encompass viruses known to infect humans, Gammacoronavirus (gCoVs), which includes viruses that affect whales and birds, and Deltacoronavirus (dCoVs), which includes viruses isolated from pigs and birds(2).
Coronaviruses are a large family that include seven zoonotic strains, which crossed the species barriers to cause human disease, four of which having a mild phenotype, being responsible for the common cold, and three being highly pathogenic: SARS-CoV, MERS-CoV, and the new SARS-CoV-2. The novel coronavirus is part of the Betacoronavirus genus, along with SARS-CoV and MERS-CoV, know to have caused severe pneumonia and death in humans in the recent past(2-4).
Coronaviruses are endemic globally and ecologically diverse, with bats and rodents as the natural reservoir host for aCoV and bCoV. The intermediate hosts, usually domestic animals (camels, civets etc.), are believed to play an important role in the transmission and emergence of these coronaviruses from bats to humans as the viruses recombine and mutate while crossing from one species to another. The source of the zoonotic infection was initially considered Huanan Seafood and Wildlife Market in Wuhan. The intermediate host in the COVID-19 pandemic hasn’t been identified, but investigations are ongoing. Studies have been conducted on snakes, pangolins, minx and ferrets, but none of these species seems to be involved in this outbreak(3,5,6).
SARS-CoV-2 spreads primarily via respiratory droplets through close contact in direct person-to-person transmission and fomite transmission, from surfaces heavily contaminated with the virus. Several reports have described the detection of SARS-CoV-2 RNA from stool samples, even after viral RNA could no longer be detected from upper respiratory tract specimens, and the live virus has been cultured from stool in rare cases(7). Faecal-oral transmission did not appear as a factor in the spread of infection. In symptomatic infections, the incubation period is within 14 days following exposure; most cases are occurring on the fourth or fifth day. The most common symptoms include cough, fever, fatigue, myalgias, diarrhoea, anosmia and dysgeusia. In some patients, the disease worsens approximately 5-10 days after symptom onset, resulting in complications such as acute respiratory distress syndrome (ARDS), cardiomyopathy, thromboembolic complications, including pulmonary embolism and acute stroke. The symptomatic infection can range from mild to critical.
Critical and fatal illness has also been associated with severe lymphopenia or elevated inflammatory markers: C-reactive protein (CRP), ferritin, and elevated D-dimer (>1 mcg/mL). Older age – over 60 – and associated comorbid conditions are at higher risk for a severe form of disease and death. These comorbidities include cardiovascular disease, underlying respiratory conditions, cancer, diabetes and obesity. In comparison, children appear to have a milder clinical course and recover within one to two weeks of disease(8,9).
Prenatal care during COVID-19 pandemic
Immunological and physiological changes that take place during pregnancy can lead to severe respiratory infections due to systemic effects. The cardiovascular and respiratory systems adaptation to pregnancy, including increased heart rate, oxygen consumption, stroke volume, decreased pulmonary capacity and functional residual capacity, may increase the risk of complications for COVID-19(10).
Pregnancy is a unique immunological state, highly adaptative, in which the maternal immune system has to balance the tolerance to the allogeneic fetus and the immunocompetence against any microbial threats. The maternal immune system is dynamic and responsive, continuously adapting to the developing fetus at various gestational stages. In the first trimester, embryo implantation and trophoblast invasion require a local inflammatory environment that promotes cell clearance, angiogenesis, cell growth and tolerance. As the fetus develops and grows during the second trimester, an antiinflammatory state is required. In the third trimester, as the body is preparing for childbirth, it reaches a second proinflammatory state. Viral or bacterial infection can trigger an inflammatory response, leading to pregnancy complications or impaired fetal development(11,12).
UNICEF estimated that around 116 million babies would be born worldwide during the COVID-19 pandemic; the deliveries have been projected for 40 weeks, since the outbreak has been declared pandemic on March 11, 2020(13).
The coronavirus pandemic has profoundly affected life around the globe, as restrictive measures to contain the virus have been applied, such as isolation, social distancing and economic shutdown. Fear of catching the virus, self-isolation, and uncertainty can take a toll on the mental health of the general population, especially of the expectant mothers who worry about their baby’s health, as well as their own(13,14).
Traditional protocols for routine prenatal visits should be modified and adapted for low- versus high-risk pregnant patients with comorbidities. Telemedicine is encouraged for prenatal visits, as a primary way to forward triage and screen potential symptoms of COVID-19. Visits to the doctor’s office should be limited at ultrasound appointments, lab tests and emergencies, and should be timed accordingly, so multiple tests can take place during the same visit.
Communicating constantly with a health care provider can help lessen the levels of stress and anxiety of expectant mothers(15,16). In one study in China, that used an online survey designed to assess the public’s immediate psychological response during the COVID-19 epidemic, approximately one-third of the 1210 respondents reported moderate to severe anxiety(17).
Management of infected pregnant women
Treating COVID-19 pregnant patients is considered more challenging than in case of non-pregnant patients, as certain additional precautions are needed and a multidisciplinary approach, including obstetric-gynaecologists, neonatologists, intensive care and infectious disease specialists and internists. A large study conducted on 16,749 COVID-19 patients hospitalized in the United Kingdom found a similar proportion of pregnant women hospitalized (6%) when comparing to the general population. Also, there was no increased mortality linked to pregnancy(18,19).
The clinical status at the presentation of pregnant women with COVID-19, symptomatic or asymptomatic, can predict the maternal and pregnancy outcomes. A study on 82 patients concluded that pregnant women who presented with COVID-19-related symptoms and subsequently tested positive for SARS-CoV-2 had a higher rate of preterm delivery (27.3%) and need for respiratory support (26.1%) than asymptomatic pregnant women(20,21).
A systematic review of nine studies that analyzed 89 cases of pregnancy in women aged 20-44 years old, all in the third trimester, infected with SARS-CoV-2, concluded that the clinical manifestations of infection in late pregnancy were similar to those of non-pregnant adults. Low-grade fever (<39°C) and cough were the dominant symptoms presented in all patients; also, there were reported fatigue, dyspnea, sore throat, myalgia and gastrointestinal symptoms (diarrhoea, vomiting). Some clinical manifestations can overlap with symptoms of pregnancy, such as fatigue, shortness of breath, nasal congestion, nausea/vomiting. In all symptomatic cases, chest CT scans revealed abnormal viral changes in lungs, with patchy ground-glass shadows(11).
The most frequently reported laboratory findings so far are normal or low white cell counts, elevated C-reactive protein, elevated lactic acid dehydrogenase, and modified coagulation tests. SARS-CoV-2 infection has been associated with hypercoagulability, resulting in ischemic changes, including ecchymosis of fingers and toes, with reported D-dimer elevation, prolonged prothrombin time and gradual decrease of fibrinogen and platelet count(18). A study was conducted on 20 placentas of SARS-CoV-2 positive mothers to assess a possible correlation between infection and risk of thrombosis in the fetal circulation. Ten of the 20 cases showed evidence of low-grade fetal vascular malperfusion or fetal vascular thrombosis, but further studies are required(22).
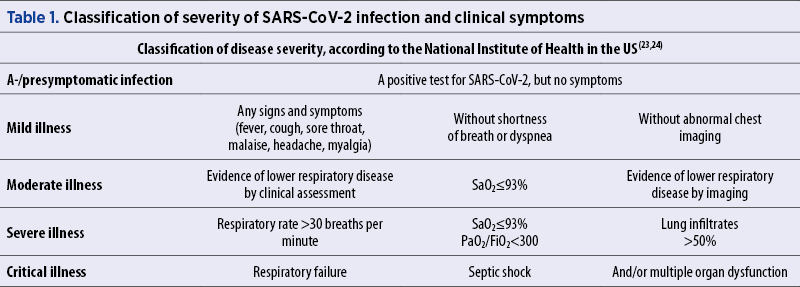
In Table 1, we show the severity of SARS-CoV-2 infection and the signs and symptoms found.
In a retrospective review on 43 pregnant patients from New York City with confirmed COVID-19, the disease course was mild in 37 cases (86%), severe in four cases (9.3%) and critical in two cases (4.7%)(23,24). In a scoping review of 33 studies that addressed SARS-CoV-2 infection during pregnancy, the spectrum of illness severity in 385 pregnant women was 368 (95.6%) considered mild, 14 (3.6%) considered severe, and 3 (0.8%) critical cases. Out of 385 women, 17 (4.4%) required admission to an intensive care unit (ICU) and six (1.6%) were mechanically ventilated, including one on extracorporeal membrane oxygenation (ECMO)(25).
A retrospective case series was published, documenting the evolution of nine pregnant women diagnosed with severe COVID-19 in their second or third trimester. The severe cardiopulmonary disease included the need for ventilator support and cardiopulmonary collapse. Seven out of the nine pregnant women had comorbidities, such as obesity, subclinical thyroid, gestational diabetes or advanced maternal age. At the time of reporting, there were seven maternal deaths, one patient was critically ill and ventilator-dependent and another recovered after prolonged hospitalization but had intrauterine fetal death. The patients were treated according to the WHO guidelines using a three-drug regimen recommended for COVID-19 pneumonia that included 75 mg oseltamivir p.o. b.i.d. for 5 days, 400 mg hydroxychloroquine sulfate daily/1000 mg chloroquine sulfate tablet as a single dose, and 400-100 mg lopinavir/ritonavir b.i.d. for 5 days. A fourth drug, ribavirin, was added when there were signs of severe disease such as loss of consciousness, tachypnea, hypotension (BP<90/60 mm Hg), multilobar lung infiltration with consolidation by chest imaging (CT or radiograph), or hypoxemia (SaO2<90%). The recommended dose was p.o. ribavirin, 1,200 mg/day twice a day, administered for 5 days. In all nine patients, thromboprophylaxis was achieved with either enoxaparin (40 mg s.c. daily) or heparin (5000 units s.c., b.i.d.)(26).
Pregnancy complications and childbirth
The universal screening for SARS-CoV-2 in women admitted to delivery is considered essential due to the rapid, uncontrollable spread of the virus(27). Caesarean delivery and preterm birth rates are increased among pregnant women with COVID-19. A systematic review including 252 patients, four of whom had twin pregnancies, reported that out of the 256 newborns, nearly 70% (175) were delivered by caesarean section, while only 30% had a vaginal birth. Preterm birth (<37 weeks of gestation) occurred in 15%(39) of the newborns. It is important to note that more than 70% of the pregnant women who were included in the review were symptomatic(24,25).
There have been only a few published studies on the neonatal infection with SARS-CoV-2, with conflicting results. Some studies found no traces of the virus in samples of the placenta, amniotic fluid, cord blood and neonatal throat swabs. In contrast, others have reported several cases of peripartum maternal infection in the third trimester, suggesting that congenital infection is possible but uncommon. A review on 179 cases of newborns tested for SARS-CoV-2 at birth from pregnant women infected in the third trimester of pregnancy reported only six positive results taken more than 16 hours after birth, thus the timing of transmission cannot be determined. Zeng et al. and Dong et al.(28,29) have described three cases of newborns with positive anti-SARS-CoV-2 IgM and IgG serologies at birth, but no positive RT-PCR in nasopharyngeal samples, nor in the blood. The presence of IgM antibodies suggests the in utero exposure of the fetus to SARS-CoV-2, given the fact that they cannot cross the placenta as IgG(30).
The majority of women who test positive for SARS-CoV-2 in the nasopharynx have negative samples of vaginal and amniotic fluid; there has been a report of a patient with a positive vaginal swab. Most placentas studied so far had no evidence of infection, but the virus has been identified in a few cases. A report described two mothers with positive results from the samples from their placentas and their neonates(26).
A well-documented case of probable congenital SARS-CoV-2 infection is of a newborn delivered at 35 weeks and 5 days by planned caesarean with airborne, droplet, and contact precautions because of worsening maternal COVID-19 in a patient with familial neutropenia, gestational diabetes and history of persistent bacterial infections. The neonate was not in contact with vaginal secretions, membranes were intact before birth, and there was no skin-to-skin contact with the mother before the collection of the first neonatal nasopharyngeal swab(31). However, all three of the neonate’s nasopharyngeal swabs were positive for SARS-CoV-2 by PCR testing, neonatal plasma tested positive on the fourth day and stool sample was positive on the seventh day. The placental swabs also tested positive, and the analyzed placental samples showed signs of inflammation and extensive early infarction, suggestive of primary viral infection. The mother’s nasopharyngeal swab, breast milk and vaginal swab were also positive for SARS-CoV-2. The extensive dissemination of the virus throughout body tissues and secretions may be secondary to immunosuppression associated with the maternal familial neutropenia. The neonate was noted to be neutropenic, had mild hypothermia, feeding difficulties consistent with prematurity and intermittent hypoglycemic episodes, without respiratory difficulties. It is possible that the neonate may have inherited the maternal condition, altering his immunologic state and contributing to the acquisition of SARS-CoV-2 infection(24,31).
Neonatal care and breastfeeding
The infants of mothers with COVID-19 are considered COVID-19 suspects, and should be tested, isolated from other healthy infants, and cared for according to infection control precautions. Limiting mother-baby contact has been proposed to reduce the risk of transmission. The temporary separation of mothers with known or suspected COVID-19 from their newborns can have negative effects on their bonding, may disrupt breastfeeding and increase stress levels in the mother, as well as the infant(24).
WHO supports the practice of skin-to-skin contact and rooming-in throughout the day and night, especially right after birth during the establishment of breastfeeding, whether or not the mother or child has suspected, probable or confirmed COVID-19. Hand and respiratory hygiene is recommended if the mother presents respiratory symptoms, and the use of a medical mask is required during breastfeeding. If direct breastfeeding cannot be achieved due to the severity of the illness or other complications, mothers should consider expressing the milk, restarting breastfeeding after a gap, wet nursing (another woman breastfeeding the child), or using human donor milk(32).
CDC advises that separation between mother and child should be determined taking into consideration the following factors: the clinical condition of mother and infant, whether the mother’s infection is suspected or confirmed, the infant’s SARS-CoV-2 testing result (the separation is not necessary if the infant has a positive test), the mother’s desire to breastfeed, the facility’s ability to accommodate mother-infant separation or colocation. In a clinical setting, CDC recommends temporary separation by having a separate room for the newborn, maintaining a physical distance of ≥6 feet between the mother and neonate or placing the neonate in a temperature-controlled area in the mother’s room(33,34).
The potential risk of viral transmission of SARS-CoV-2 from ingestion of breast milk is unclear. Some studies found no traces of the virus in samples of breast milk from mothers with COVID-19(25,36), while other investigators reported identifying samples of breast milk positive for the virus(33,35).
The consensus is that breastfeeding should be encouraged because of its many maternal and infant benefits. In the setting of maternal SARS-CoV-2 infection, the newborn may receive passive protection from the virus through breast milk, a rich source of antibodies and other anti-infective factors.
Conclusions
Although the current evidence shows that the new coronavirus infection in pregnant women seems to be less aggressive than the previous severe coronavirus infections, all suspected pregnant women need to be systematically screened, monitored and followed-up.
The extent and clinical significance of vertical transmission remain unclear, and the possible mechanisms of transmission need to be clarified.
Further investigations and follow-up studies of pregnant mothers infected with SARS-CoV-2 are critical to understand the potential risks, to improve the care and to develop specific treatments of the current or future outbreak. Because specific antiviral treatments and vaccines are still under development, testing, quarantine and social distancing are encouraged to prevent the virus spread.
Bibliografie
-
1. Coronavirus disease (COVID-19) outbreak situation. World Health Organization, 2020.
-
2. Harapan H, Itoh N, Yufika A, et al. Coronavirus disease 2019 (COVID-19): A literature review. J Infect Public Health. 2020;13(5):667-673.
-
3. Paules CI, Marston HD, Fauci AS. Coronavirus Infections – More Than Just the Common Cold. JAMA. 2020 Feb 25;323(8):707-708.
-
4. Kasraeian M, Zare M, Vafaei H, Asadi N, Faraji A, Bazrafshan K, Roozmeh S. COVID-19 pneumonia and pregnancy; a systematic review and meta-analysis. J Matern Fetal Neonatal Med. 2020 May 19;1-8.
-
5. Hu B, Ge X, Wang LF, Shi Z. Bat origin of human coronaviruses. Virology Journal. 2015 Dec;12:221.
-
6. Liu YC, Kuo RL,Shih SR, COVID-19: The first documented coronavirus pandemic in history. Biomedical Journal. 2020 May 5;S2319-4170(20)30044-5.
-
7. Hirsch MS, Bloom A (Eds). Coronavirus disease 2019 (COVID-19): Epidemiology, virology, and prevention. Up to date. 2020.
-
8. Report of the WHO-China Joint Mission on Coronavirus Disease 2019 (COVID-19). World Health Organization.
-
9. Deville JG, Song E, Ouellette CP. Coronavirus disease 2019 (COVID-19): Considerations in children. Up to date. 2020.
-
10. Mor G, Aldo P, Alvero A. The unique immunological and microbial aspects of pregnancy. Nat Rev Immunol. 2017;17:469–482.
-
11. Muhidin S, Behboodi Moghadam Z, Vizheh M. Analysis of Maternal Coronavirus Infections and Neonates Born to Mothers with 2019-nCoV; a Systematic Review. Arch Acad Emerg Med. 2020;8(1):e49.
-
12. Liu H, Wang LL, Zhao SJ, Kwak-Kim J, Mor G, Liao AH. Why are pregnant women susceptible to COVID-19? An immunological viewpoint. J Reprod Immunol. 2020 Jun;139:103122.
-
13. Navigating pregnancy during the coronavirus disease (COVID-19) pandemic. UNICEF.
-
14. Narang K, Ibirogba ER, Elrefaei A, Trad ATA, Theiler R, Nomura R, Picone O, Kilby M, et al. SARS-CoV-2 in Pregnancy: A Comprehensive Summary of Current Guidelines. J Clin Med. 2020;9(5):1521.
-
15. Perrin PB, Pierce BS, Elliott TR. COVID-19 and telemedicine: A revolution in healthcare delivery is at hand. Health Sci Rep. 2020;3(2):e166.
-
16. Cioti AM, Stănescu AMA, Grăjdeanu IV, Şerban B, Popescu E, Bratu OG, Diaconu CC. Telemedicine in Europe – Current Status and Future Perspectives. Modern Medicine. 2019;26(4):165-168.
-
17. Wang C, Pan R, Wan X, Tan Y, Xu L, Ho CS, Ho RC. Immediate Psychological Responses and Associated Factors during the Initial Stage of the 2019 Coronavirus Disease (COVID-19) Epidemic among the General Population in China. Int J Environ Res Public Health. 2020;17(5):1729.
-
18. The Royal College of Obstetricians and Gynaecologists (RCOG). Coronavirus (COVID-19) Infection in Pregnancy. Information for healthcare professionals. Version 9: May 13, 2020.
-
19. Docherty AB, Harrison EM, Green CA, Hardwick HE, Pius R, Norman L, Holden KA, Read JM, et al. Features of 20,133 UK patients in hospital with COVID-19 using the ISARIC WHO Clinical Characterisation Protocol: prospective observational cohort study. BMJ. 2020;369:m1985. doi:10.1136/bmj.m1985.
-
20. London V, McLaren R Jr, Atallah F, Cepeda C, McCalla S, Fisher N, Stein JL, et al. The Relationship between Status at Presentation and Outcomes among Pregnant Women with COVID-19. Am J Perinatol. 2020;37(10):991-994.
-
21. Li T, Lu H, Zhang W. Clinical observation and management of COVID-19 patients. Emerg Microbes Infect. 2020;9(1):687-690.
-
22. Baergen RN, Heller DS. Placental Pathology in COVID-19 Positive Mothers: Preliminary Findings. Pediatr Dev Pathol. 2020;23(3):177-180.
-
23. Wu Z, McGoogan JM. Characteristics of and Important Lessons From the Coronavirus Disease 2019 (COVID-19) Outbreak in China: Summary of a Report of 72,314 Cases From the Chinese Center for Disease Control and Prevention. JAMA. 2020 Apr 7;323(13):1239-1242.
-
24. Overview of COVID-19: Epidemiology, Clinical Presentation, and Transmission. Available at: https://www.covid19treatmentguidelines.nih.gov/overview/
-
25. Breslin N, Baptiste C, Gyamfi-Bannerman C, et al. COVID-19 infection among asymptomatic and symptomatic pregnant women: Two weeks of confirmed presentations to an affiliated pair of New York City hospitals. Am J Obstet Gynecol MFM. 2020;2(2):100118.
-
26. Berghella V. Coronavirus disease 2019 (COVID-19): Pregnancy issues. In: Charles J Lockwood, Vanessa A Barss (Eds). 2020; UptoDate. Available at: https://www.uptodate.com/contents/coronavirus-disease-2019-covid-19-pregnancy-issues?topicRef=127926&source=see_link. Accesed: June 1, 2020.
-
27. Sutton D, Fuchs K, D’Alton M,Goffman D. Universal Screening for SARS-CoV-2 in Women Admitted for Delivery. N Engl J Med. 2020;382:2163-2164.
-
28. Zeng L, Xia S, Yuan W, et al. Neonatal Early-Onset Infection With SARS-CoV-2 in 33 Neonates Born to Mothers With COVID-19 in Wuhan, China. JAMA Pediatr. 2020;174(7):722–725.
-
29. Dong L, Tian J, He S, et al. Possible Vertical Transmission of SARS-CoV-2 From an Infected Mother to Her Newborn. JAMA. 2020;323(18):1846–1848.
-
30. Egloff C, Vauloup-Fellous C, Picone O, Mandelbrot L, Roques P. Evidence and possible mechanisms of rare maternal-fetal transmission of SARS-CoV-2. J Clin Virol. 2020;128:104447.
-
31. Liu W, Wang J, Li W, Zhou Z, Liu S, Rong Z. Clinical characteristics of 19 neonates born to mothers with COVID-19. Front Med. 2020;14(2):193-198.
-
32. Groß R, Conzelmann C, Müller JA, et al. Detection of SARS-CoV-2 in human breastmilk. Lancet. 2020;395:1757-1758.
-
33. Evaluation and Management Considerations for Neonates At Risk for COVID-19,2020. Centers for Disease Control and Prevention (CDC).
-
34. Hantoushzadeh S, Alireza A. Shamshirsaz, Nouri B, Nekooghadam SM, et al. Maternal death due to COVID-19. Am J Obstet Gynecol. 2020;223(1):P109.E1-109.E16.
-
35. Elshafeey F, Magdi R, Hindi N, Elshebiny M, Farrag N, Mahdy S, et al. A systematic scoping review of COVID-19 during pregnancy and childbirth. Int J Gynaecol Obstet. 2020;150(1):47-52.
-
36. World Health Organization. Nutrition. Breastfeeding advice during the COVID-19 outbreak.