Tumoare germinală testiculară nonseminomatoasă cu prognostic rezervat
Non-seminomatous germ cell testicular tumor with poor prognosis
Abstract
We present the case of a 37-year-old man, with no significant medical history, presented to the hospital with dyspnea and chest pain after one episode of haemoptysis, the clinical examination revealing a left-sided testicular mass. His complaints have begun 6 months before the admission. After four courses of BEP (bleomycin, etoposide and cisplatin), orchiectomy was performed and the patient was under surveillance in the oncology department. The degree of AFP elevation in patients with non-seminomatous germ cell tumours (NSGCT) establishes the prognosis and is used to assign an appropriate chemotherapy regimen.Keywords
mature teratomalung metastasesalpha 1-fetoproteinmultidisciplinary treatmentRezumat
Prezentăm cazul unui pacient în vârstă de 37 de ani, fără antecedente patologice semnificative, care s-a prezentat la camera de gardă cu dispnee şi durere toracică apărute după un episod de hemoptizie, la examinarea clinică prezentând o formaţiune palpabilă la nivelul testiculului stâng. Simptomatologia a început în urmă cu şase luni. După patru serii de BEP (bleomycin, etoposide şi cisplatin), se intervine chirurgical, practicându-se orhiectomie pe cale inghinală, ulterior pacientul rămânând în urmărire în Departamentul de oncologie medicală. Creşterea AFP la pacienţii cu tumori germinale testiculare nonseminomatoase (NSGCT) are valoare prognostică şi este folosită pentru stabilirea unui tratament chimioterapic potrivit.Cuvinte Cheie
teratom maturmetastaze pulmonareAFPtratament multidisciplinarCase presentation
We present the case of a 37-year-old man, in clinical state of health until 6 months before the admission, when he began to notice a left-sided testicular swelling, but with no associated pain at the time. The swelling progressed over the next few months, and was associated with dyspnea and chest pain. Also, he complained of weight loss about 10 kg in the past 6 months. He presented to the hospital in August 2011, after one episode of haemoptysis, with severe dyspnea.
The patient had no surgical history, no chronic medical condition that he was aware of, and he wasn’t on any prescription medications. He smoked one pack of cigarettes a day for 12 years, and he used to drink 1-2 alcoholic beverages on weekends. His family history revealed: the father with hypertension, and an uncle with type 2 diabetes.
The patient’s vitals on presentation were: temperature 37.8°C, pulse 125 beats per minute, blood pressure 124/80 mmHg, the respiratory rate was 33 beats per minute, and the oxygen saturation was 81% when the patient was breathing ambient air. The physical examination presented an anxious patient with mild oral cyanosis, in the basal side of the left lung we couldn’t hear normal sounds; he presented a 6/15 cm, hard, intensely painful on palpation, left testicular tumor. The other examinations were normal.
Laboratory tests at first admission
CBC showed a haemoglobin of 11.9, a hematocrit of 31.9%, a white-cell count of 8700 per cubic millimeter and a platelet count of 445,000 per cubic millimeter; serum electrolytes were normal, the urea and the creatinine level – 0.4 mg per deciliter (185.6 µmol per liter). PT (prothrombin time) was 16.5 and APTT (activated partial thromboplastin time) was 53; lactate dehydrogenase (LDH) was 1198 units per liter; alpha 1-fetoprotein level was 13232 ng/ml and beta-human chorionic gonadotropin (B-HCG) level was 776 mUI/ml.
Given the patient’s history and findings of scrotal mass, there was a recommendation for further imaging to evaluate for malignancy.
The imagistic exams performed in September 2011, chest X-ray and CT scan of the chest (Figure 1 and Figure 2), showed multiple nodules with different sizes, and the probability of metastasis became propounded.
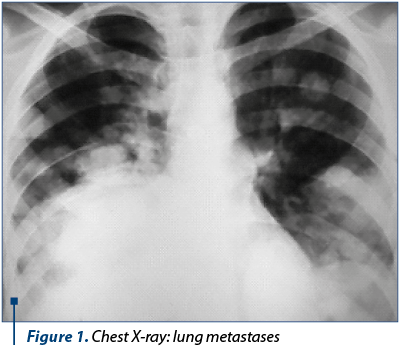
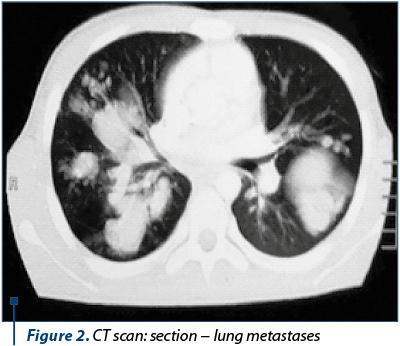
Clinical diagnosis: elevation of AFP, B-HCG and LDH, testicular mass, CT scan revealed multiple nodules in the lungs, being suspected a testicular tumor with metastasis.
Management: the therapeutic decision was planned by BEP (bleomycin, etoposide and cisplatin) as classic five-day protocol. After four cycles of BEP, the level of serum tumor markers reached the normal value, and we decided to perform a left orchiectomy, that had no intra- or postoperative complications. The histopathologic examination showed mature teratoma inflitrative to the tunica albuginea and spermatic cord; the spermatic cord lymph node showed also mature teratoma, with pT3 pN2 stage stadialization.
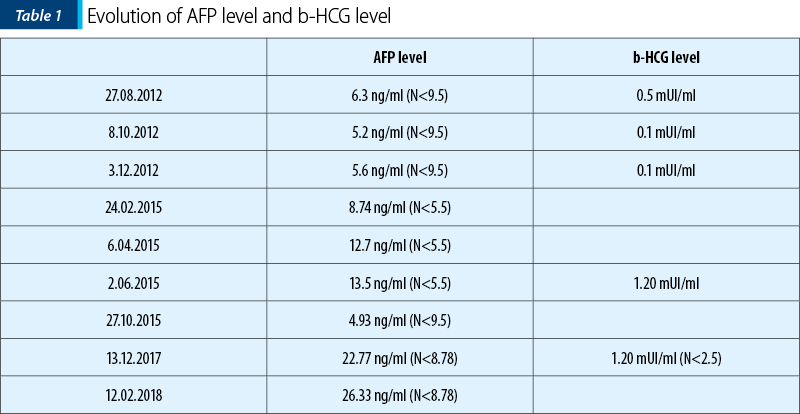
In subsequent repeated checks, pulmonary images of M1PUL persisted, but without a clinical correspondent. In June 2012, PET-CT scan showed several metabolically active masses in some of the M1PUL, the serum level of AFP was 26 ng/ml (N<9.5), and B-HCG level was 0.5 mUI/ml (N<0.5). Therefore, the decission was to start chemotherapy. After four cycles of cisplatin + etoposide and four cycles of paclitaxel + ifosfamide, well tolerated, the PET-CT (February 2013) showed multiple M1PUL in both lungs, dimensional stationary, metabolic inactive, lombo-aortic lymph node, also metabolic inactive. In March 2015, the check-up PET-CT showed once again activity in his lungs metastases, and also active metabolic lymph nodes (latero-tracheal, hilar lymph nodes), with no other sites involvment. Besides, the AFP serum level was slightly elevated, thus the therapeutic decission was to resume chemotherapy with cisplatin + etoposide. After three cycles of this chemotherapy regimen, the AFP serum level was still high, then it was changed to paclitaxel + carboplatin (7 cycles). In February 2018, the AFP level was again elevated (Table 1), and we resumed the chemotherapy to paclitaxel + carboplatin.
At this moment, our patient is at his fourth cycle of chemotherapy (since we restarted). In the future, we plan to reevaluate the patient using PET-CT and serum tumor markers.
Discussion
Testicular cancer is commonly designated into seminomatous and non-seminomatous tumours(1), with the former accounting for 40% and the latter accounting for 60%. This distinction guides the clinician in both prognosis and treatment strategies. The methods used for establishing the metastatic stage are α-fetoprotein level (elevated in 50-60% of NSGCT(2)) and CT scan. Positron-emission thomography scan can also be used to highlight the hypermetabolic lymph node metastases.
Under the Comprehensive TNM staging system developed by the American Joint Committee on Cancer and the International Union Against Cancer(3), our patient would be classified as follows:
- pT3 –Tumour invades the spermatic cord, with or without vascular/lymphatic invasion.
- N2 – Metastasis with a lymph node mass >2 cm, but not >5 cm in its greatest dimension, or more than 5 nodes positive, none >5 cm, or evidence of extranodal extension of tumor.
- M1a – Non-regional nodal or pulmonary metastasis.
- S3 – LDH >10 × N or hCG (mIU/mL) >50000 or AFP (ng/mL) >10000.
This classifies the patient into the poor prognostic group (stage III C), giving him a 5-year survival rate of around 45%(3).
The patient is a unique case, showing a special significance and connection between AFP serum level, PET-CT hypermetabolic lymph nodes and the respose to chemotherapy. In case of future evolution of his metastatic disease (M1PUL), we have to take into account – according to the guidelines – gemcitabine + oxaliplatin, gemcitabine + paclitaxel(4). We are also looking further for new molecular targeted therapies with a good response in the treatment of germ cell tumors of patients with recurrence after salvage chemotherapy.
Conflict of interests: The author declares no conflict of interests.
Bibliografie
- Eble JN, Sauter G, Epstein JI et al. (eds). Pathology and Genetics of Tumors of the Urinary System and Male Genital Organs. Lyon, France: IARC Pres. 2004.
- de Takats PG, Jones SR, Penn R, et al. Alpha-foetoprotein heterogeneity: what is its value in managing patients with germ cell tumours?. Clin Oncol (R Coll Radiol). 1996; 2013: 323–6.
- International Germ Cell Consensus Classification: a prognostic factor-based staging system for metastatic germ cell cancers International Germ Cell Cancer Collaborative Group. J Clin Oncol. 1997; Feb;15(2):594-603.
- De Vita et al. (eds.). Cancers of the Genitourinary System. In: DeVita, Hellman and Rosenberg’s Cancer Principles & Practice of Oncology, 10th Edition. LWW Health Library. 2015; 1000-1001.
