The events following the emergence of SARS-CoV-2, in Wuhan, China, in December 2019, established COVID-19 as the pandemic airborne disease of the year 2019. The infection has been declared a global health emergency. In addition to the now already common symptoms, such as fever, fatigue and mild respiratory symptoms like dry cough, the patients with COVID-19 can also develop neurological manifestations, such as headache, anosmia, hyposmia, dysgeusia, meningitis, encephalitis and acute cerebrovascular incidents during the course of this disease. The first case of meningitis associated with COVID-19 was reported in Japan in February 2020. Since then, more cases have been described, each with their own particularities. Although very rare, the neurological manifestations are now part of a large scale of symptoms that SARS-CoV-2 infection may exhibit. Perhaps as rare as the neurological symptoms are the auditory manifestations of the same disease. In this article, we focused on presenting our experience with post-COVID-19 related meningitis and the subsequent vestibular and cochlear loss that followed.
Deficit cohleovestibular central după COVID-19 şi meningită consecutivă
Central cochlear and vestibular deficit post-COVID-19 related meningitis
First published: 27 mai 2022
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/ORL.55.2.2022.6470
Abstract
Rezumat
Evenimentele care au urmat apariţiei SARS-CoV-2, în Wuhan, China, în decembrie 2019, au făcut din COVID-19 pandemia anului 2019. Infecţia a fost declarată o urgenţă sanitară globală. Pe lângă simptomele deja comune, cum ar fi febră, oboseală şi simptome respiratorii uşoare precum tuse uscată, pacienţii cu COVID-19 pot dezvolta şi manifestări neurologice, cum ar fi dureri de cap, anosmie, hiposmie, disgeuzie, meningită, encefalită şi evenimente cerebrovasculare acute în decursul acestei boli. Primul caz de meningită asociat cu COVID-19 a fost raportat în Japonia, în februarie 2020. De atunci au fost descrise mai multe cazuri, fiecare cu propriile particularităţi. Deşi foarte rare, manifestările neurologice fac acum parte dintr-o scară largă de simptome pe care le poate prezenta infecţia cu SARS-CoV-2. Poate la fel de rare precum simptomele neurologice sunt manifestările auditive ale acestei boli. În acest articol ne-am concentrat pe prezentarea experienţei noastre privind un caz de meningită post-COVID-19 soldat cu deficit vestibulocohlear ulterior.
Introduction
The infection with SARS-CoV-2 may lead to a wide range of extrapulmonary symptoms, to sensorial and neural complications, such as sudden loss or dysfunction of smell and/or taste(6), otologic and auditory symptoms(7,8), to nonspecific symptoms and long-term neurological complications(9). It has been proven that the neural invasion of the SARS-CoV-2 is directly associated with the angiotensin-converting enzyme 2 (ACE2) mechanism. This enzyme acts as a functional receptor for the viral breech(10). This enzyme receptor may be found in lungs, in the type 2 alveoli. It is also expressed by many other cells, such as glial cells and neurons, and can cause the neurological involvement during the SARS-CoV-2 infection through direct or indirect mechanisms(11,12). Given the usual symptoms of the SARS-CoV-2 infection, the rare patients who exhibited atypical symptoms such as the ones cited above as first signs of manifestation for COVID-19 did not benefit from an early diagnosis, hence the late onset of treatment in some cases. This might have been part of the cause for which all the cases of atypical COVID-19 with neurological and/or auditory involvement were moderate or severe forms of disease(11).
Despite the research on olfactory and gustatory symptoms of the SARS-CoV-2 infection(5,13) since the start of the pandemic, the impact of the disease on the auditory and vestibular systems has received less attention. In addition, reviews including patients with both neurological and auditory involvement are even rarer(14). The present paper intends to present the case of an elderly female patient who, during the infection with SARS-CoV-2, developed meningitis, accompanying otitis media and bilateral central hearing loss and vestibular deficit.
Case presentation and patient history
We present the case of a 64-year-old female who was brought into the Emergency Department (ED) with a brief history of superior respiratory tract infection (seven days of dry coughing, fever and a sore throat) and elevated blood pressure. The patient was self-isolating at home after getting a positive result of a SARS-CoV-2 rapid antigen test. She had taken the decision with her family doctor (general practitioner) who was checking in with her daily via telephone, helping her monitor her blood pressure levels and corresponding medication and also advising her which symptomatic drugs to resort to in order to keep the respiratory symptoms under control. The patient reported no other pre-diagnosed medical problems upon arriving in the ED and mentioned no known allergies.
The patient’s condition had been progressively worsening for the past three days prior to the ED presentation. She had been complaining of headaches that were progressing in intensity, some muscle weakness which led to difficulty in standing up and additional difficulty in maintaining balance during the walking process itself. She also pointed out that during these past few days her hearing was worsening progressively, to the point where she couldn’t hear her GP over the phone very well, so she decided to call an ambulance and be evaluated in the ED. The initial ENT and neurology assessments carried out in the emergency department pointed out to the following diagnosis: possible viral meningitis and bilateral congestive otitis media. We noted bilaterally congested tympanic membranes, despite no earaches, bilateral hearing loss (which seemed too severe to be solely attributed to conductive etiology – the patient was using a very loud voice when talking and would solicit the doctors to repeat everything they said), gait impairment, neck pain during flexion and hyperextension, and mild muscle weakness in the inferior limbs. The patient was able to stand up with some effort but once in a standing position her gait would widen and proceeding to step forward she would tilt sideways, nearly resulting in falls. We noted ataxic features in her upright standing position. The swaying and tilting when standing up were not systematized and the sharpened or Tandem Romberg test (or any other stepping test) was impossible to perform at the present time. The head impulse test (HIT) was normal(15). The patient was alert, oriented, with a Glasgow coma scale score of 15/15 and negative Babinski and Kernig signs. The blood tests done in the ED indicated slightly elevated white blood cells count (predominantly lymphocytes), elevated inflammation markers (ESR and CRP) and normal levels for CK and CK-MB(16).
The reverse transcription polymerase chain reaction (PCR) test for the SARS-CoV-2 infection was positive in the nasal swab and, thus, the patient was isolated in the COVID-19 ward. A lumbar puncture was performed right after admission into the hospital and we proceeded in booking a head MRI and initiating antibiotic prophylaxis for bacterial meningitis with ceftriaxone. Pain control was achieved with metamizole, acetaminophen and a low dose of steroids, all while maintaining blood pressure levels between 120/90 mmHg and 150/90 mmHg.
Investigations, findings and current outcome
Lumbar puncture results provided us with a cerebrospinal fluid (CSF) analysis depicting signs of viral meningitis and, in addition to antibiotics, our patient was also given antiviral drugs (acyclovir 200 mg administered four times per day). CSF gram staining, Ziehl-Neelsen staining and cultures showed no identifiable bacterial development, and further testing done for herpes simplex type 1, herpes simplex type 2 and varicella zoster virus were negative. D-dimer levels were also elevated above normal values(17). The PCR test was not able to determine the presence of SARS-CoV-2 in the CSF.
The MRI performed after admission showed the inflammation of the meninges adjacent to the junction between the medulla oblongata and the pons. The MRI scan was repeated after discharge and some residual inflammation could be detected located around the bulk of the left vestibulocochlear nerve.
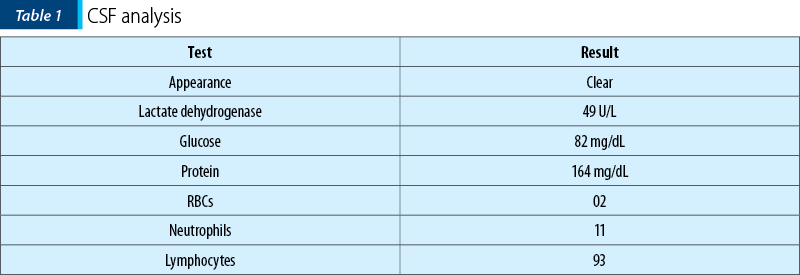
Proper audiology and vestibular assessments could not be carried out while the patient was isolated in the COVID-19 ward. The hearing loss stabilized under treatment, as the patient told us, but she would still complain of extreme difficulty when walking. After seven days of treatment, the congestion of the tympanic membranes had fully disappeared, allowing us to diagnose bilateral tympanosclerosis, and after another three days the patient was discharged into the Neurology Department: the headache and the stiffness of the neck were gone, the patient experienced no fever and muscle weakness, but the hearing loss and walking problems persisted. The patient did not describe any history of vertigo or oscillopsia during the entire period of the disease. Her perception of her own gait and walking difficulty were described as a subtle and sudden feeling of ebriety when she would start pacing, disregarding the direction of movement.
The second time our ENT doctor reviewed the patient the gait was ataxic, and walking was possible if the patient had a caretaker help or if she would hold her hands against the walls or spread them out around her body. The Romberg and Fukuda tests showed a unilateral left-side deviation and rotation. The HIT was mild positive to the left side. No signs of dysdiadochokinesia were detected.
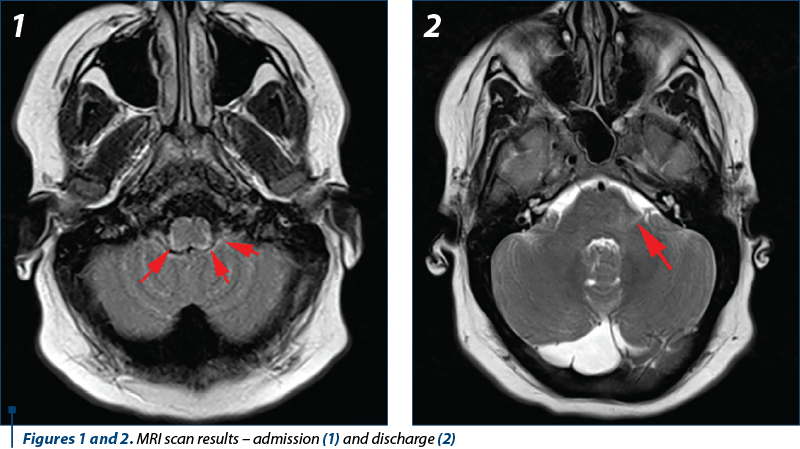
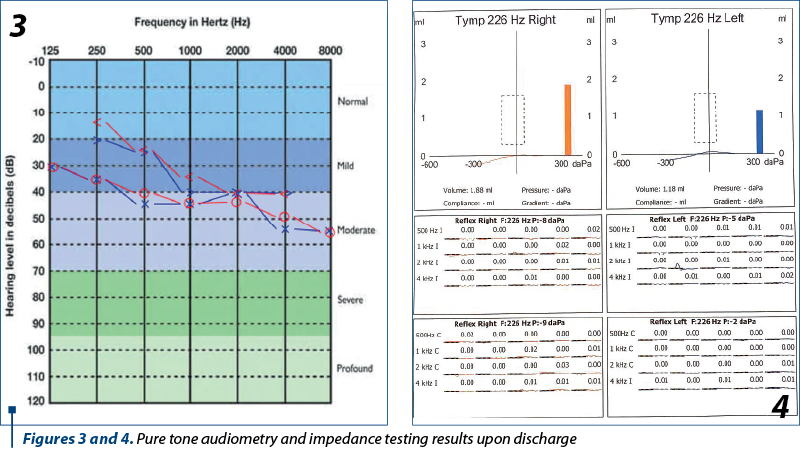
Pure tone audiometry indicated a bilateral mixed moderate hearing loss (with nearly symmetrical bone conduction thresholds). Tympanometry testing revealed bilateral As tympanograms and no present stapedial reflex in ipsilateral or contralateral testing. The Bonnier test was negative.
The infrared camera eye examination showed no spontaneous or positional nystagmus, but head-shaking maneuvers revealed a mildly symptomatic left-beating nystagmus which disregarded gaze and inverted its direction after about a minute. The inverted right-beating nystagmus was asymptomatic. The eye-cover test was normal with no detectable skew deviation of the eyes(18). We considered this to be a central type of nystagmus, indicating a possible lesion of the vestibular nuclei(19).
Our joined ENT and neurology team decided to opt for discharge soon after. After five more days of hospitalization, in which we had the patient move around with the help of a care taker and move her head as much as she could, we noticed an improvement in the patient’s gait. We decided to put the patient on a six-month treatment plan with betahistine, 48 mg per day, nootropic drug support (piracetam) and 30 minutes per day of vestibular rehabilitation exercises(20,21).
The first assessment of the patient after discharge from the hospital was scheduled a month later. The patient could walk on her own, using a cane for support as she had no one accompanying her for the appointment or while using public transportation or going shopping. The Romberg and Fukuda tests still indicated leaning and rotating toward the left side, despite a normal bilateral HIT result. Infrared camera eye examination could still trace the same type of nystagmus and hearing levels were stationary despite the bettering in gait and posture. The patient decided to follow through with the treatment plan up to at least six months and opted for the bilateral use of hearing aids as hearing rehabilitation.
Discussion
There is some debate in literature whether the viral replication of the SARS-CoV-2 may directly cause inflammation, even within the central nervous system (CNS) and adjoining structures (cranial nerves). Some types of viral infections may cause meningitis and encephalitis(22). There have been COVID-19-positive patients who exhibited meningeal irritation signs (nuchal rigidity, muscle weakness, present Kernig and Brudzinski signs), along with positive extensor plantar response(3). After excluding bacterial infections of the CNS, SARS-CoV-2 encephalitis may be suspected and diagnosed. However, SARS-CoV-2 staining may be negative in the CSF specimen, which might be due to the extremely low titer of the virus in the CSF, or due to insufficient standardization for such a novel viral test(23). We had reports from Japan where a case report described the first patient who was sent to the emergency department with convulsions and who was subsequently diagnosed with aseptic encephalitis with SARS-CoV-2. The RNA was detected in CSF(4). In addition, two patients with acute meningoencephalitis during the course of the SARS-CoV-2 infection were reported in Switzerland, and a case of rhombencephalitis was reported as a rare complication of acute COVID-19 infection in the United Kingdom(24,25).
Ischemic strokes as CNS involvement in the course of COVID-19 have been described in the recent literature. In a study published by Li et al., 10 out of 219 patients hospitalized for SARS-CoV-2 infection developed an ischemic stroke(26). The average time span from the first symptoms of the SARS-CoV-2 infection to cerebrovascular pathology was about 10 days. Given the fact that SARS-CoV-2 also interacts with endothelial cells through the ACE receptors, this mechanism seems plausible as means of affecting the smaller blood vessels inside the CNS. Elevated blood pressure and cardiovascular involvement prior to COVID-19 means the possibility of a cardiac embolism may also be discussed. There is evidence that COVID-19 can associate myocarditis(27), which could lead to such events. Multiple ECGs were performed during admission and, although the result was normal and blood pressure was stable in our patient, paroxysmal arrhythmias cannot be fully excluded. Elevated values of D-dimers seen in our patient may also be discussed as they can rise during a SARS-CoV-2 infection, and abnormal levels potentially indicate a poor prognosis of the disease(28). Our patient (as well as her GP) didn’t report any previous cardiovascular complications or previously elevated D-dimer levels. Their values were already elevated at the onset of the neurological symptoms during the course of the SARS-CoV-2 infection. Increased D-dimer levels may indicate microthrombosis which may easily cause a plethora of minor lesions at the level of the CNS(29).
Conclusions and challenges
Despite the fact that, in the particular case of our patient we concluded that we faced a central lesion which caused the vestibular deficit, early vestibular rehabilitation seemed to improve the patient’s gait and walking performance. Provided that there is no strong recommendation against vestibular rehabilitation for central vestibular system lesions in able-bodied patients, we set up a three-month rehabilitation plan for the present case(30). The evaluation of the patient one month after discharge showed remarkable improvements in gait and balance, so we recommended that the daily duration of vestibular rehabilitation should increase from 30 minutes to 60 minutes, and that it should be done in at least two separate sessions, in order to try and maximize the benefit of the rehabilitation exercises and, ideally, rid the patient of any walking support(31).
The hearing loss was difficult to evaluate due to a multitude of factors. The initial congestion of the tympanic membranes which suggested otitis media, the audiometry testing which in our hospital is unavailable to the COVID-19 ward, the later diagnosed tympanosclerosis, all increased the difficulty in properly monitoring the hearing loss. With the help of the current tests we tend to consider that, at least to some extent, the hearing loss that our patient experienced may have a central origin in addition to the modified aspect of the tympanic membranes and possible cochlear damage. Our patient opted for bilateral hearing aids, but it remains to be seen if these will provide a long-term benefit for her, due to the mixed nature of her lesions(32).
Conflicts of interests: The authors declare no conflict of interests.
Bibliografie
-
Lu H, Stratton CW, Tang YW. Outbreak of pneumonia of unknown etiology in Wuhan, China: the mystery and the miracle. J Med Virol. 2020;92:401–2. doi: 10.1002/jmv.25678.
-
Mao L, Jin H, Wang M, Hu Y, Chen S, He Q, et al. Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in Wuhan, China. JAMA Neurol. 2020;77:1–9. doi: 10.1001/jamaneurol.2020.1127.
-
Ahmed MU, Hanif M, Ali MJ, Haider MA, Kherani D, Memon GM, et al. Neurological manifestations of COVID-19 (SARS-CoV-2): a review. Front Neurol. 2020;11:518. doi: 10.3389/fneur.2020.00518.
-
Moriguchi T, Harii N, Goto J, Harada D, Sugawara H, Takamino J, et al. A first case of meningitis/encephalitis associated with SARS-Coronavirus-2. Int J Infect Dis. 2020; 94:55–8. doi: 10.1016/j.ijid.2020.03.062.
-
Jafari Z, Kolb BE, Mohajerani MH. Hearing Loss, Tinnitus, and Dizziness in COVID-19: A Systematic Review and Meta-Analysis. Can J Neurol Sci. 2022;49(2):184-195. doi:10.1017/cjn.2021.63.
-
Agyeman, AA, Chin, KL, Landersdorfer, CB, Liew, D, Ofori-Asenso, R. Smell and taste dysfunction in patients with COVID-19: a systematic review and meta-analysis. Mayo Clin Proceed. 2020;95:1621–31.
-
Almufarrij I, Uus K, Munro KJ. Does coronavirus affect the audio-vestibular system? A rapid systematic review. Int J Audiol. 2020;59:487–91.
-
Maharaj S, Bello Alvarez M, Mungul S, Hari K. Otologic dysfunction in patients with COVID-19: A systematic review. Laryngoscope Investig Otolaryngol. 2020;5(6):1192-1196. Published 2020 Nov 17. doi:10.1002/lio2.498.
-
Abboud H, Abboud FZ, Kharbouch H, Arkha Y, El Abbadi N, El Ouahabi A. COVID-19 and SARS-CoV-2 infection: pathophysiology and clinical effects on the nervous system. World Neurosurg. 2020;140:49–53.
-
Özçelik Korkmaz M, Eğilmez OK, Özçelik MA, Güven M. Otolaryngological manifestations of hospitalised patients with confirmed COVID-19 infection. Eur Arch Otorhinolaryngol. 2021;278(5):1675-1685. doi:10.1007/s00405-020-06396-8.
-
Mao L, Jin H, Wang M, et al. Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in Wuhan, China. JAMA Neurol. 2020;77:683–90.
-
Wang D, Hu B, Hu C, et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA. 2020;323:1061–9.
-
Rocke J, Hopkins C, Philpott C, Kumar N. Is loss of sense of smell a diagnostic marker in COVID-19: a systematic review and meta-analysis. Clin Otolaryngol. 2020;45:914–22.
-
Fancello V, Hatzopoulos S, Corazzi V, et al. SARS-CoV-2 (COVID-19) and audio-vestibular disorders. Int J Immunopathol Pharmacol. 2021;35:20587384211027373. doi:10.1177/20587384211027373.
-
Kattah JC. Use of HINTS in the acute vestibular syndrome. An Overview. Stroke Vasc Neurol. 2018;3(4):190-196. doi:10.1136/svn-2018-000160.
-
Zinellu A, Sotgia S, Fois AG, Mangoni AA. Serum CK-MB, COVID-19 severity and mortality: An updated systematic review and meta-analysis with meta-regression. Adv Med Sci. 2021;66(2):304-314. doi:10.1016/j.advms.2021.07.001.
-
He X, Yao F, Chen J, et al. The poor prognosis and influencing factors of high D-dimer levels for COVID-19 patients. Sci Rep. 2021;11(1):1830. doi:10.1038/s41598-021-81300-w.
-
Strupp M, Kremmyda O, Adamczyk C, et al. Central ocular motor disorders, including gaze palsy and nystagmus. J Neurol. 2014;261 Suppl 2(Suppl 2):S542-S558. doi:10.1007/s00415-014-7385-9.
-
Pierrot-Deseilligny C, Milea D. Vertical nystagmus: clinical facts and hypotheses. Brain. 2005;128(Pt 6):1237-1246. doi:10.1093/brain/awh532.
-
Georgescu M. Vestibular Rehabilitation – Recommended Treatment for Permanent Unilateral Vestibular Loss. Int J Neurorehabilitation Eng. 2017;4:4. doi: 10.4172/2376-0281.1000282.
-
Han BI. Vestibular Rehabilitation in Central Dizziness. In: Simplified Vestibular Rehabilitation Therapy. 2021;17-20.
-
Meyfroidt G, Kurtz P, Sonneville R. Critical care management of infectious meningitis and encephalitis. Intensive Care Med. 2020;46:192–201. doi: 10.1007/s00134-019-05901-w.
-
Ye M, Ren Y, Lv T. Encephalitis as a clinical manifestation of COVID-19. Brain Behav Immun. 2020;88:945–6. doi: 10.1016/j.bbi.2020.04.017.
-
Bernard-Valnet R, Pizzarotti B, Anichini A, Demars Y, Russo E, Schmidhauser M, et al. Two patients with acute meningoencephalitis concomitant with SARS-CoV-2 infection. Eur J Neurol. 2020;27(9):e43-e44. doi:10.1111/ene.14298.
-
Wong PF, Craik S, Newman P, Makan A, Srinivasan K, Crawford E, et al. Lessons of the month 1: a case of rhombencephalitis as a rare complication of acute COVID-19 infection. Clin Med. 2020;20:293–4. doi: 10.7861/clinmed.2020-0182.
-
Li Y, Li M, Wang M, Zhou Y, Chang J, Xian Y, Wang D, Mao L, Jin H, Hu B. Acute cerebrovascular disease following COVID-19: a single center, retrospective, observational study. Stroke Vasc Neurol. 2020;5(3):279–284.
-
Siripanthong B, Nazarian S, Muser D, Deo R, Santangeli P, Khanji MY, Cooper LT Jr, Chahal CAA. Recognizing COVID-19-related myocarditis: The possible pathophysiology and proposed guideline for diagnosis and management. Heart Rhythm. 2020;17(9):1463–1471.
-
Yu B, Li X, Chen J, Ouyang M, Zhang H, Zhao X, Tang L, Luo Q, Xu M, Yang L, Huang G, Liu X, Tang J. Evaluation of variation in D-dimer levels among COVID-19 and bacterial pneumonia: a retrospective analysis. J Thromb Thrombolysis. 2020;50(3):548–557.
-
Chen W, Pan JY. Anatomical and Pathological Observation and Analysis of SARS and COVID-19: Microthrombosis Is the Main Cause of Death. Biol Proced Online. 2021 Jan 20;23(1):4. doi: 10.1186/s12575-021-00142-y.
-
Han BI, Song HS, Kim JS. Vestibular rehabilitation therapy: review of indications, mechanisms, and key exercises. J Clin Neurol. 2011;7(4):184-196. doi:10.3988/jcn.2011.7.4.184.
-
Szturm T, Ireland DJ, Lessing-Turner M. Comparison of different exercise programs in the rehabilitation of patients with chronic peripheral vestibular dysfunction. J Vestib Res. 1994;4(6):461-479.
-
McCandless GA, Parkin JL. Hearing aid performance relative to site of lesion. Otolaryngol Head Neck Surg. 1979 Nov-Dec;87(6):871-5. doi: 10.1177/019459987908700622.
Articole din ediţiile anterioare
Sindrom vestibular acut unilateral indus de infecţia cu SARS-CoV-2
Infecţia cu SARS-CoV-2 prezintă o simptomatologie vastă, în perioada pandemiei fiind raportate o serie de manifestări în sfera ORL, o parte din el...
Tromboza sinusului venos lateral
Tromboza sinusului venos lateral (TSL) poate să apară ca o complicaţie intracraniană a otitei medii. În era preantibiotică, mortalitatea e...
Consideraţii asupra tratamentului pacientului cu epistaxis în timpul pandemiei de COVID-19
Autorii îşi propun să treacă în revistă unele consideraţii rezultate din activitatea cotidiană cu privire la una dintre cele mai frecvente urge...
Revizuirea simptomelor COVID-19 – tulburări olfactive. Ceea ce ştim în prezent şi ipoteze pentru cercetări viitoare
Disfuncţia olfactivă este una dintre cele mai frecvente simptome asociate cu infecţia cu SARS-CoV-2, în cadrul populaţiei generale.