Transplant integral de piele la o pisică cu avulsie cutanată podală distală - prezentare de caz
Full thickness mesh graft in a cat with degloving wound - case presentation
Abstract
Applying skin grafts is one of the most common methods of closing distal defects in the limbs. At the same time, the results are also aesthetic. This paper shows the operatory steps present in applying a skin graft, approaching the wound before surgery to get granulation tissue and the postoperative care for a cat of about 5 years of age, with unknown history.Keywords
skin mesh graftdegloving woundRezumat
Aplicarea grefelor de piele este una dintre cele mai frecvente metode de închidere a defectelor distale de la nivelul membrelor. În același timp, rezultatele obținute sunt și estetice. Această lucrare arată pașii operatori prezenți în aplicarea unei grefe de piele, abordarea plăgii înainte de intervenție pentru obținerea țesutului de granulație și îngrijirea postoperatorie la o pisică de aproximativ 5 ani, cu istoric necunoscut.Cuvinte Cheie
grefă de pieleplagă prin avulsieIntroduction
An ample loss of skin with underlying tissue and exposure of deep components (e.g., tendons, ligaments, bones) define a degloving injury. These kinds of wounds are most frequently seen on distal limbs, medial tarsus/metatarsus. The main cause of degloving wounds is car accident, especially when the animal is dragged or pushed by a moving car. In all of the cases, bacteria and debris are present in the wound.
Free grafts are described as a piece of skin detached from an area of the body and placed over the wound. There are two types of free grafts when we talk about graft thickness:
- full thickness (epidermis and entire dermis)
- partial/split thickness (epidermis and a variable portion of dermis).
Skin grafts are used when there is a defect that cannot be closed by skin flaps or direct apposition. Two factors influence skin graft survival: revascularization and absorption of the tissue fluid.
Case report
A 4-year-old female, shorthair cat, weighting 3.25 kg, was presented to our clinic. Before that, the owner was at another clinic for consult and he was disappointed because they recommended euthanasia or amputation of the limb. Besides, the first veterinary doctor treated the cat with amoxi+clavulanate and Nekro Veyxym. The owner said that she went missing for about 10 days.
Clinical examination
After a thorough clinical exam, we found that all was normal, except for a degloving injury. The back right leg was affected.
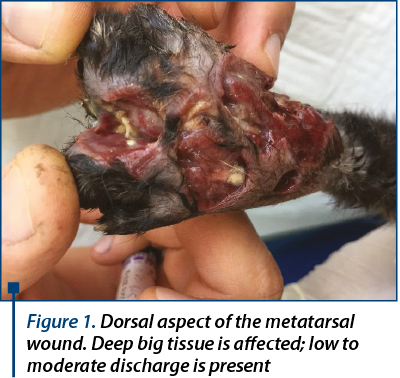
There was a massive inflammation with infection and a lot of debris on the dorsal surface of metatarsal area (figure 1), and ventral above metatarsal pad.
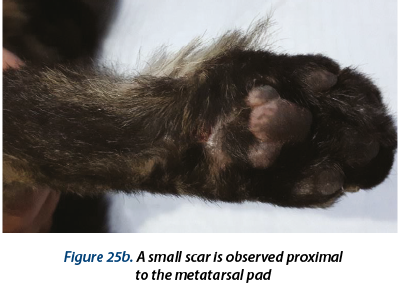
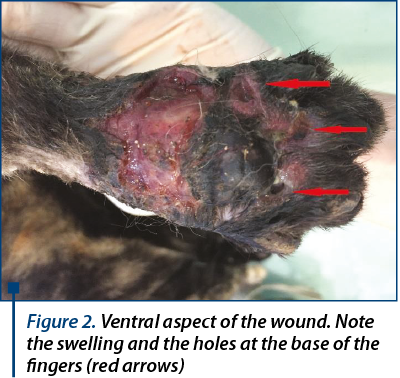
Besides, also in the ventral metatarsal area, another wound located proximal to the metatarsal pad and 3 deep holes were identified at the base of second, third and fourth finger (figure 2). There could be distinguished the chronic aspect.
Another lesion was noticed on the same leg, in the medial aspect of the thigh. This wound was deep with a circular shape (figure 3).
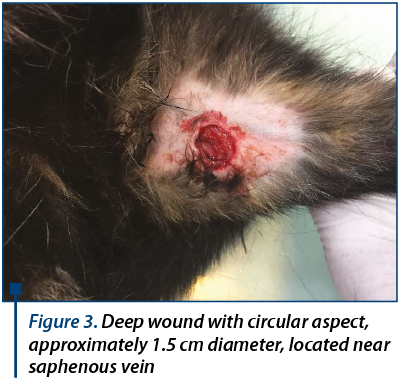
We estimated that the lesion occurred about two weeks ago. We registered pain and high local temperature after palpation.
The cat was stable, normothermic, with normal color of mucous membrane, CRT 3 seconds and normal superficial lymph nodes.
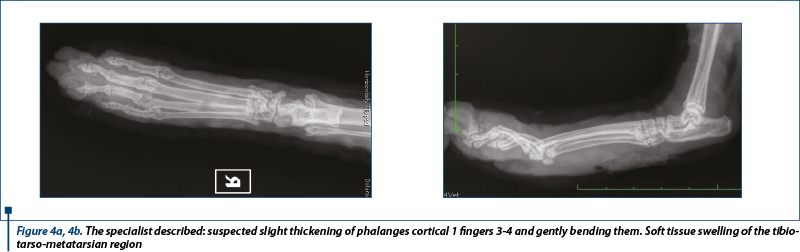
Radiograph of the affected back limb
Two X-ray views were made to eliminate bone changes or foreign bodies (figure 4a, figure 4b).
Approach
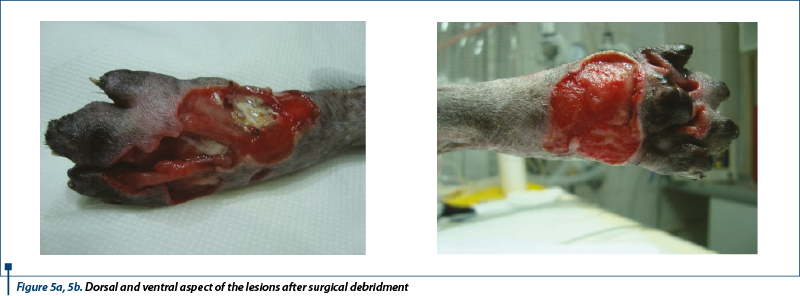
After evaluation, the initial recommendation included a good wound management under anesthesia. Before surgical debridement (figure 5a, 5b), culture was done.
Next, wound lavage was initiated with one bag of 500 ml of warm saline (the most easy way to deliver fluids on the wound is to connect the saline bag with a administration set to the syringe and needle with a 3-way stopcock; a large amount of liquid is needed to be effective).
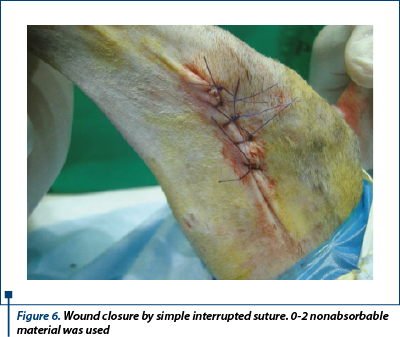
Finally, this first stage ends with a wet-to-dry bandage. A primary wound closure was performed for the lesion placed on the medial aspect of the thigh (figure 6), after intensive cleaning, removal of foreign bodies and dead skin.
Empirically, the cat received cefquinome until the results arrived and for pain management we administered tramadol 3 mg/kg and meloxicam 0.1-0.2 mg/kg. The cat recover well after anesthesia.
Culture result
One day before performing surgery, we received the culture result. Streptococcus canis (++++) was identified and was sensible to many antibiotics. Amoxicilin+clavulanic acid (Synulox® was initiated for general therapy and chloramphenicol ointment (Opticlor - Pasteur) for local therapy.
Next, a full thickness mesh graft was used on the dorsal aspect of the limb due to the length and depth of the wound, and the other wound was left for healing by second intention, both being protected by bandages. In the next 10 days, the limb wounds were treated in the same manner.
Removal of bacteria, granulation tissue formation and the beginning of epithelization were supported by next bandages as follows:
- Day 1
Wet-to-dry bandage was used after surgical debridement (this kind of bandages adhere to the wound and remove the little layer of dead tissue when we take off). Soaked in warm saline 1-2 minutes before removing, they were changed after 24 hours one to the other. Cotton gauze was the primary contact layer of the bandage.
- Day 2 and day 3
Moisture retentive dressing (MDR) - calcium alginate (Sorbalgon - Hartmann®) was the primary contact layer. It is good to use it when there exists high exudate like in our patient (figure 7a, 7b).
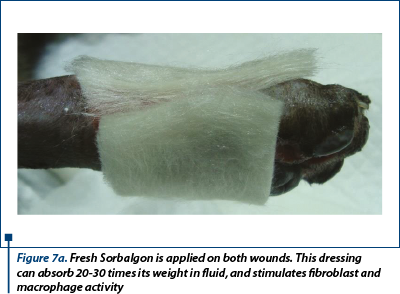
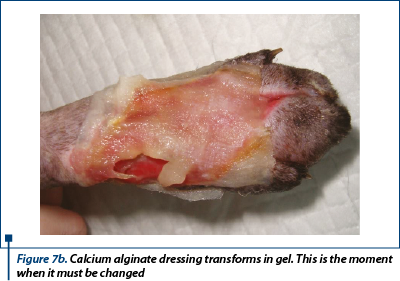
- Day 4, 6 and day 9
Moisture retentive dressing (MDR) - hydrocolloid (Hydrocoll - Hartmann®) was the primary contact layer because the discharge decreased (figure 8).
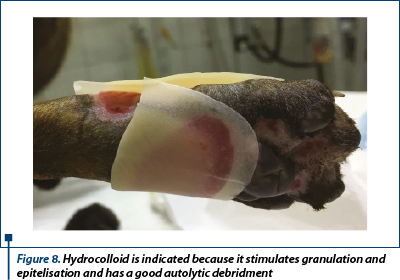
After 9 days of management we inspected the wounds and we anticipated that in day 11 the granulation tissue will be ideal for performing the graft.
- Day 11 - second anesthesia and surgery.
Describing surgical procedure
Preoperative surgical site preparation: the cat was placed in left lateral recumbency, with the wound exposed.
The limb was clipped entirely and povidone iodine and alcohol were used for aseptic surgery. Sterile warm saline 0.9% was used for wound lavage. Meanwhile, a colleague prepared the donor site - lower craniolateral thorax (right side) in the same manner.
Almost 1 mm of epithelium that has started to grow from the wound edges over the granulation tissue was removed using a thumb forceps and a no. 10 scalpel blade (figure 9).
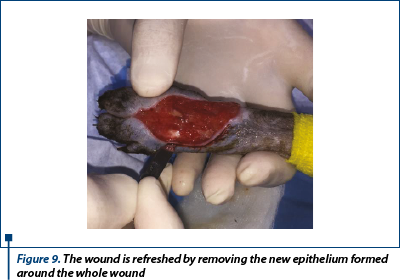
A perpendicular incision was made right at the edge of haired skin with epithelium. The wound was incised all around and after that the epithelium was removed by advancing the scalpel blade under the epithelium around the wound. Then, undermining was performed around the wound edges.
A fragment of sterile surgical drape was used over the wound to get the exact shape. The drape “pattern” was placed to the donor area.
To maintain the wound moist, we placed over a cotton gauze moistened in warm sterile saline 0.9% while the graft was transferred. The direction of hair growth was marked with a black arrow above the donor site, so that the direction of the hair growth on the graft would be the same as the hair growth direction on the skin surrounding the wound.
After that, the margins of the drape “pattern” was traced on the skin.
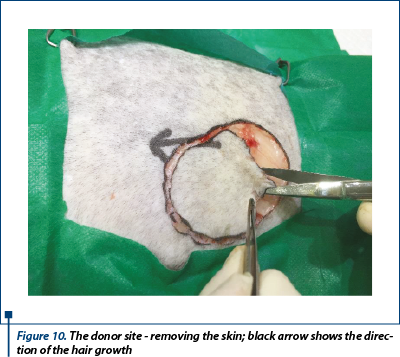
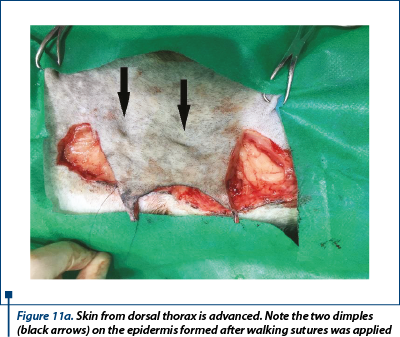
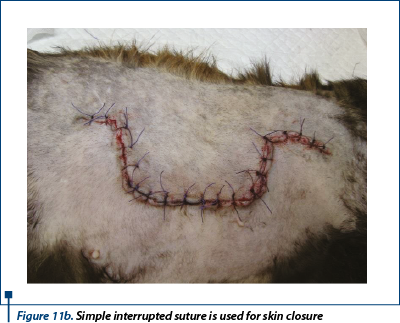
The skin of the donor bed was incised with no. 10 scalpel blade and removed using thumb forceps and Metzenbaum scissors (figure 10). The defect left after removing the graft was primary closed by undermining and advancing the skin edges with walking sutures using 3-0 monofilament absorbable suture material and finally the skin was sutured in a simple interrupted suture using 2-0 monofilament nonabsorbable suture (figure 11a, 11b).
Preparing the graft
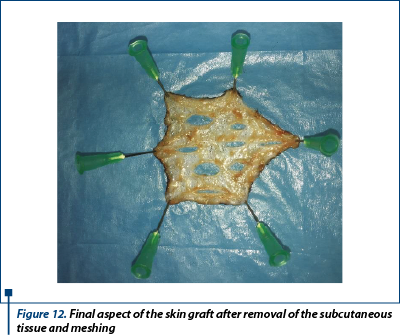
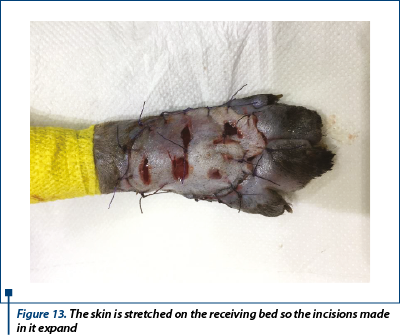
The dermal side of the graft was placed on a polystyrene board with a thickness of 10 cm covered with a sterile drape and after that we fixed and stretched with 21G needles. The subcutaneous tissue was removed from the graft. Next, parallel incisions was made in the graft, 0.5-0.7 cm long and apart (figure 12). At the end, the graft was placed on the granulation bed and sutured with 4-0 monofilament nonabsorbable suture in a simple interrupted suture manner. Additional tacking suture was placed to ensure the expansion of the mesh incision and allow the fluid drainage (figure 13).
Choosing the right bandage after grafting and aftercare
It is important to use a nonadherent primary dressing. My initial choice was Grassolind® (Hartmann). This dressing is sufficiently porous to allow easy passage of exudate from the wound surface and preventing maceration of surrounding tissue (figure 14). The ventral metatarsal wound maintain hydrocolloid dressing (Hydrocoll - Hartmann®) as primary layer. A thin layer of chloramphenicol oinment (Opticlor - Pasteur®) was used all around both wounds and over the graft.

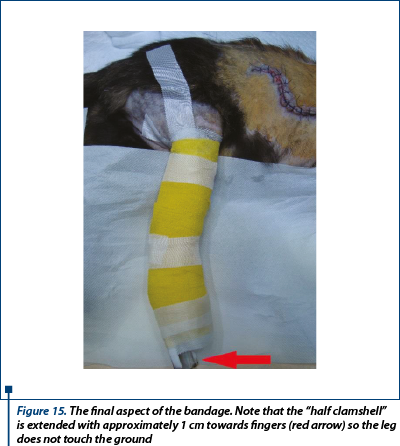
Over the first dressings was applied 5 cm x 5 cm of compresses (Medicomp - Hartmann®) and a roll gauze was the second layer. After a few laps of gauze stirrups was placed to secure the bandage in place. Extemporaneous half “clamshell” splint (figure 15) was made from plastic material which was curved in such a way that the limb was fixed in semi flexion. The splint was a little bit longer than the extremity of the pelvic limb (“toe-dancing” position), thus provided a maximum relief pressure. In the proximal area, under the splint, I put cotton to prevent pressure injuries on the caudal aspect of the thigh. Applied from proximal to distal and with moderate tension, elastic warp was the final protective layer of the bandage and it was secured at the proximal end with tape.
Changing bandages
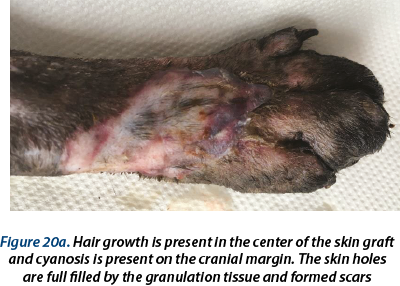
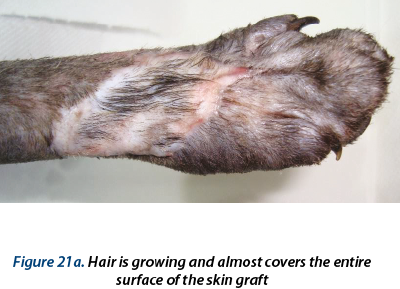
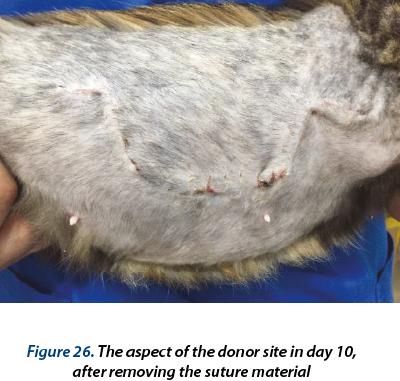
The bandage was changed in days 1, 3, 5, 7 and 10 postoperatively. In day 10 the suture material was removed from the graft and from the donor site. From day 17 to day 29 hydrogel (Hydrosorb - Hartmann®) was used as primary bandage layer and the bandage was changed from 4 to 4 days. In day 28 no discharge was present in the bandage; the wound was completely healed and 0.2-0.4 mm of hair was present in the center of the graft.
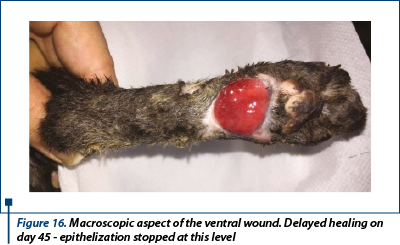
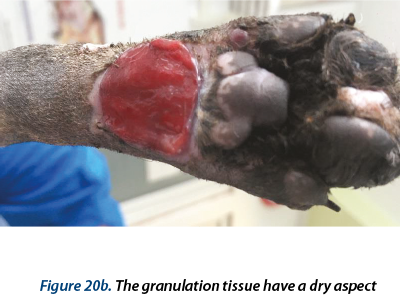
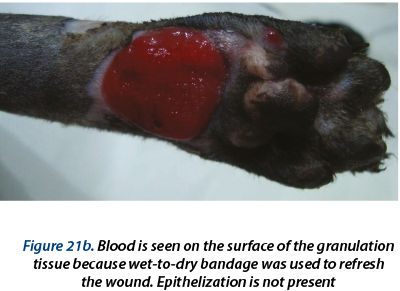
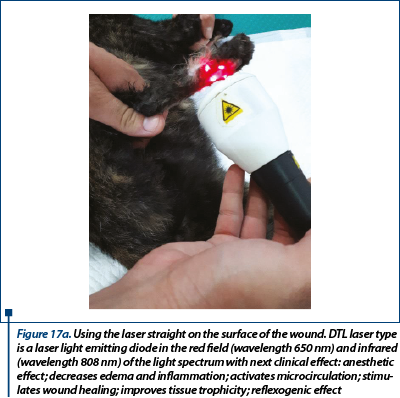
A delayed healing occurred at the wound in the ventral region (figure 16). From day 28 to day 59 epithelization has advanced very slow and granulation tissue has captured an appearance of ulcer (in this time, the wound was aseptic prepared and hydrocolloid and hydrogel were used as primary layer bandage and without the splint). In day 59 the wound was refreshed on the surface with a scalpel blade and laser therapy (figure 17a) and medical Manuka honey (figure 17b) was used daily for 14 days. After that, a complete healing was reached.

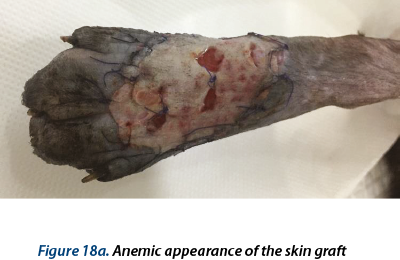
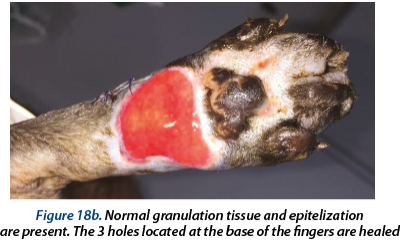
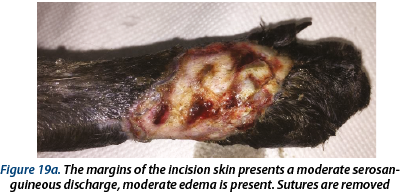
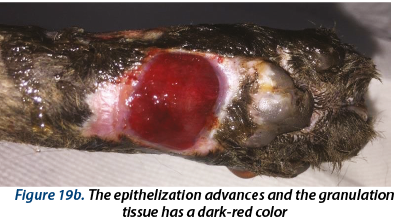
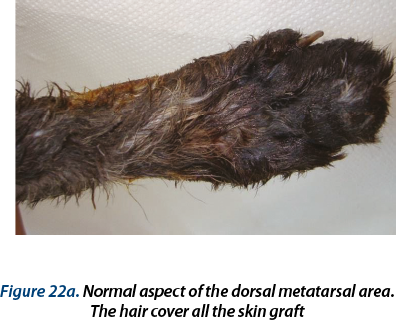
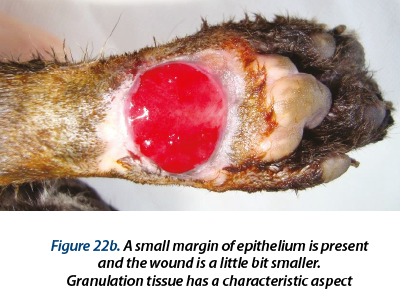
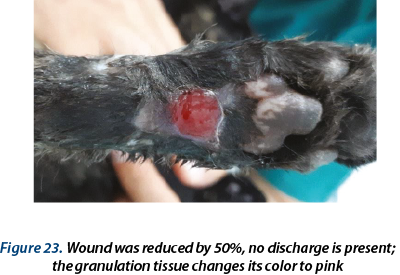
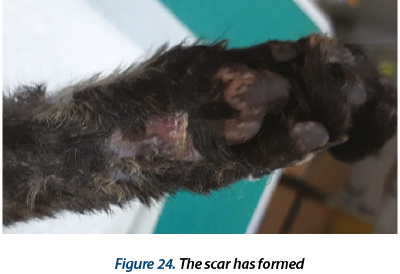
Illustrating wounds evolution after surgery
Bibliografie
1. Steve F. Swaim, DVM, MS; Janet Welch, DVM, DACVS; Robert L. Gillette, DVM, MSE (2015) - Management of Small Animals Distal Limb Injuries, by Tenton NewMedia, USA.
2. Michael M. Pavletic, DVM - Atlas of Small Animal Wound Management and Reconstructive Surgery, Third Edition (2010), W.B. Saunders, USA.
3. Karen M. Tobias, DVM, MS, DACVS – Veterinary Surgery Small Animal (2012), Saunders , USA.