Consideraţii privind abordarea multidisciplinară a tumorilor uterine de tip STUMP
Considerations about multidisciplinary therapeutic approach to STUMP uterine tumors
Abstract
Introduction. The abbreviation “STUMPs” indicates the smooth uterine muscle of uncertain malignant potential tumors, in the group of uterine smooth muscle tumors (SMTs) that cannot be diagnosed unequivocally as benign or malignant. The diagnosis, medical approach and follow-up of this pathology are still controversial. Materials and method. We reviewed the medical databases using the MeSH words “STUMPs”, “smooth uterine muscle tumors” or “SMTs” to highlight the most important papers on this topic. Results. The literature research revealed 162 articles about STUMPs. The majority are case reports or case series, including less than 10 patients with long-term follow up. The preoperative imaging for STUMP diagnosis or differentiation from leiomyoma-leiomyosarcoma is difficult. The preoperative sonographic tumors appear in 83.3% of cases with well-defined margins, 66.7% hyperechoic, 100% heterogeneous and 66.7% of cases with acoustic shadowing. The patient’s age, fertility desire, imagistic characteristics, size and location of the tumor should be considered before the procedure. The pathologic features expressed a mean number of mitosis about 8, mild atypia in almost 66.7% of cases and necrosis in 33.3% of cases. The recurrence rate after myomectomy was 6.6%. STUMP diagnosis can be managed as a second myomectomy or hysterectomy. Other treatment options for recurrence include adjuvant chemotherapy, radiotherapy and progestins GnRH analogs. Conclusions. STUMP is classified as an intermediate pathological form of uterine tumors, between benign and malignant. Metastases after many years from the initial approach can appear, thus the patients diagnosed with STUMP should be followed on the long term.Keywords
STUMPuterine smooth muscle tumorshistologic diagnosisSTUMP recurrenceRezumat
Introducere. Abrevierea „STUMPs” semnifică tumori uterine netede cu potenţial de malignizare incert, din grupul tumorilor musculare uterine netede (SMT) care nu pot fi diagnosticate cu certitudine ca fiind benigne sau maligne. Diagnosticul, abordarea terapeutică şi urmărirea acestei patologii sunt controversate. Materiale şi metodă. Am revizuit bazele de date medicale folosind cuvintele-cheie „STUMPs”, „tumori musculare uterine netede” sau „SMTs” pentru a evidenţia cele mai importante articole despre acest subiect. Rezultate. Literatura a dezvăluit 162 de articole despre STUMP. Majoritatea sunt prezentări de caz sau serii de cazuri, care includ mai puţin de 10 paciente cu urmărire pe termen lung. Imagistica preoperatorie pentru diagnosticul de STUMP sau diferenţierea de leiomiom-leiomiosarcom este dificilă. Aspectele ultrasonografice preoperatorii relevă în 83,3% din cazuri tumori bine delimitate, 66,7% din cazuri hiperecogene, 100% heterogene şi 66,7% din cazuri cu con de umbră acustică. Vârsta pacientei, dorinţa de procreare, caracteristicile imagistice, dimensiunea şi localizarea tumorii trebuie luate în considerare înainte de procedura chirurgicală abordată. Caracteristicile histopatologice au evidenţiat un număr mediu de aproximativ 8 mitoze, 66,7% din cazuri cu atipii uşoare şi necroză în 33,3% din cazuri. Rata de recurenţă după miomectomie a fost raportată ca fiind 6,6%. După diagnosticul de STUMP se poate efectua a doua miomectomie sau histerectomie. Alte opţiuni de tratament pentru recurenţă includ chimioterapia adjuvantă, radioterapia şi analogii GnRH sau progestine. Concluzii. Din punct de vedere histopatologic, STUMP este clasificată ca o formă intermediară, între tumorile uterine benigne şi cele maligne. Se pot observa metastaze după mulţi ani de la abordarea iniţială, iar pacientele diagnosticate cu STUMP trebuie urmărite pe termen lung.Cuvinte Cheie
STUMPtumori uterine netedediagnostic histopatologicrecurenţa STUMPIntroduction
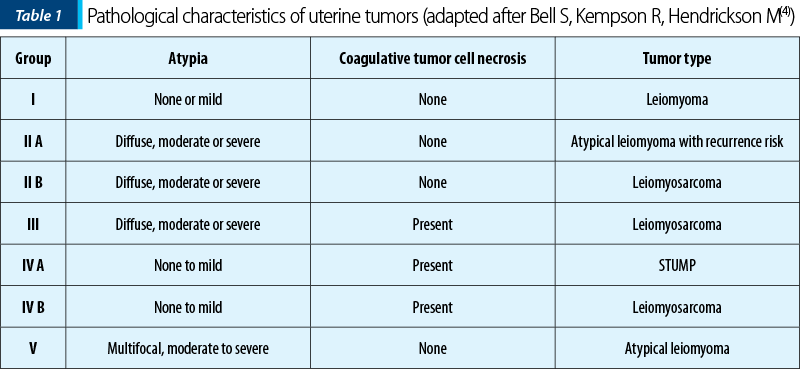
Smooth muscle tumors of uncertain malignant potential (STUMP) are classified as uterine smooth muscle tumors (SMTs), meeting some characteristics of sarcomas, but not the full diagnostic criteria. According to Bell et al., STUMPs are a group of SMTs, characterized by the presence of coagulative tumor cell necrosis, absent to mild cellular atypia, and mitotic index less than 10, so they cannot be diagnosed unequivocally as benign or malignant. Mean mitotic index seems to be 8, coagulative tumor cell necrosis is present in 33.3% of cases, and mild atypia is present in 66.7% of cases(1). STUMP diagnosis implies histological analysis after myomectomy or hysterectomy. If the tumor does not meet the criteria for leiomyosarcoma and has combinations of Stanford’s criteria, STUMP diagnosis is accurate.
Materials and method
We reviewed the medical databases using the MeSH words “STUMPs”, “smooth uterine muscle tumors” and “SMTs” to highlight the most important publications on this topic.
Results and discussion
The literature research revealed 162 articles about STUMPs. The majority are case reports or case series, including less than 10 patients with long-term follow-up. The preoperative imaging for STUMP diagnosis or differentiation from leiomyoma-leiomyosarcoma is difficult. The pathologic features expressed a mean number of mitosis about 8, mild atypia in 66.7% of cases, and necrosis in 33.3% of cases(4). The factor most strongly associated with malignant behavior is coagulative tumor cell necrosis, characterized by an abrupt transition between viable cells and necrotic areas(2).
In addition to Stanford criteria for the histologic diagnosis of SMTs, tumor borders and the presence of tumor invasion into adjacent myometrium may also be associated with malignant outcomes. Irregular, “geographic” shapes like islands on a map and sharp transitions between viable and nonviable tumor tissue are features that predict best the malignant behavior in STUMP (Figure 1).
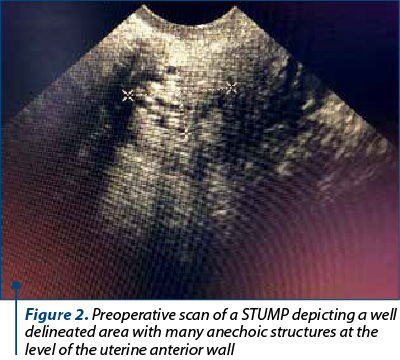
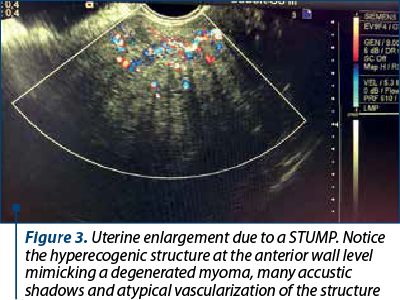
Being a rare finding and with unclear diagnosis criteria, the subjective studies for this entity are scarce. The lack of a consistent definition is a challenge for the outcome prediction and clinical management. Differences in applying histological diagnostic criteria lead to unclear information regarding the frequency of STUMP. Due to the possibility of metastasis or recurrent disease, further research is needed (Table 1).
The patients with STUMP are mostly in their mid-forties and underwent surgery usually for preoperative leiomyoma diagnosis(1). Most cases appear in premenopausal women. The clinical manifestations of STUMP are also a mix of benign uterine leiomyomas simptoms and uterine sarcomas ones, including rapidly growing pelvic mass, abnormal vaginal bleeding, symptoms of anemia, low abdomen pressure and pelvic pain. In a study analyzing cases of STUMP over a 10-year period, menometrorrhagia was described by all participants.
There is no imaging tehnique that can be reliable in order to differentiate STUMP tumors from other uterine neoplasms. These kind of tumors are usually diagnosed following a pathological examination after a myomectomy or hysterectomy(4,5).
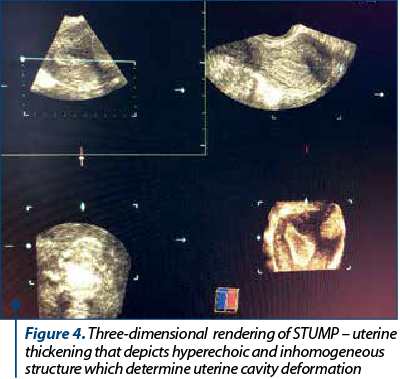
Transvaginal ultrasonography is the first choice in terms of imaging techniques for such symptoms, although it has certain limitations in presenting the global image of large tumors and tissue characterization. There seems to be an overlap in the ultrasonographic aspects of a degenerating leiomyoma and malignant tumors. Thus, a single tumor, with a non-myometrial origin, with absence of acoustic shadowing, thickened endometrium and ascites, is associated with STUMP in an ultrasonographic examination (p=0.001, p<0.001, p=0.03, p<0.0001, and p=0.03, respectively). The preoperative sonographic tumors appear in 83.3% of cases with well-defined margins, 66.7% hyperechoic, 100% heterogeneous and in 66.7% of cases with acoustic shadowing. Color Doppler examinations are difficult because Doppler results fluctuate according to menopausal status, the presence of cystic alterations, and with the localization and size of the mass(5). Three-dimensional power Doppler angiography is a technique that allows the estimation of uterine tumoral volume, and a more objective assessment of uterine vascularization with calculation of vascularization index (it measures the number of color voxels in the volume, representing the vessels in the tissue), flow index (it represents the color value in the color voxels, which indicates the average intensity of blood flow) and vascularization-flow index (it is the mean color values in all the voxels in the volume, which represents both vascularization and blood flow)(4). There is no significant relevance between sonographic findings, cellular atypia, presence of necrosis or mitotic index.
Conventional, perfusion and diffusion MRI are used to further investigate the characteristics of uterine masses, thus playing an important role in preoperative differentiation; however, they do not provide a definitive diagnosis(3,4,5). Regarding conventional MRI, a single, large tumor, with non-myometrial origin, poorly defined margins, thickened endometrium, peritoneal implants, intermediate or high signal intensity in T1 or T2 sequences, heterogenous T1 signal, cystic alteration of the tumor and heterogeneity of the tumor’s enhancement were significantly associated with STUMP. As for DWI (diffusion weighted imaging), a high-intensity signal at b=1000 s/mm2 was associated with STUMP (p<0.001)(5).
As mentioned before, STUMP growth is slow, with the possibility of delayed recurrences (for a mean of 51 months after the initial diagnosis). Mean survival was 61.5 months, and the five-year survival was 92-100%. Recurrence rate after myomectomy and hysterectomy is similar (between 8.7% and 11%)(6,7). Recurrent STUMPs proved to be low-grade uterine smooth muscle tumors. When STUMP is diagnosed in myomectomy specimens, hysterectomy represents the gold standard for those women who have completed their childbearing(2). Patient’s age and fertility should be considered before deciding on the procedure. There have been reported successful pregnancies following fertility sparing surgery, although these patients should be enrolled in systematical follow-up programs, including clinical and imaging studies(8).
Recurrent tumors, either STUMP or leiomyosarcoma, can occur in uterus, pelvis, retroperitoneum, lung, liver and bone. There seems to be no difference in recurrence rates for women who underwent myomectomy and those with hysterectomy(7). In a study where immunohistochemical stains were done for p16, p53, ki-67 and estrogen and progesterone receptors were performed, the recurrent cases of STUMP showed positive staining for p16 and p53, therefore testing for these markers proved to be helpful in the prediction of STUMP behavior(9,10).
Follow-ups should be repeated every 6 months for the first 5 years, then annual surveillance for the next 5 years, and they should include history and clinical examination, along with imaging data (chest radiography, pelvic ultrasound, MRI).
Conclusions
STUMP is a rare heterogenous tumor. History and clinical examination do not provide important data for diagnosis. Ultrasonography and MRI are useful for diagnosis and patient follow-up, but the final diagnosis is established only after the pathological examination. Since this type of tumor is considered at a border between benign and malignant outcome, hysterectomy should be the gold standard management. It is difficult to estimate the biological behavior pattern and prognosis of STUMP. Recurrences can often occur, therefore patients should undergo frequent follow-ups in order to prevent a malignancy.
Conflict of interests: The authors declare no conflict of interests.
Bibliografie
-
Bacanakgil BH, Deveci M, Karabuk E, Soyman Z. Uterine smooth muscle tumor of uncertain malignant potential: clinicopathologic-sonographic characteristics, follow-up and recurrence. World J Oncol. 2017; 8(3):76–80.
-
Dall’Asta A, Gizzo S, Musarò A, Quaranta M, Noventa M, Migliavacca C, et al. Uterine smooth muscle tumors of uncertain malignant potential (STUMP): pathology, follow-up and recurrence. Int J Clin Exp Pathol. 2014; 7(11):8136–42.
-
Wu T, Yen T. Clinical presentation and diagnosis of uterine sarcoma, including imaging. Best Pract Res Clin Obstet Gynaecol. 2011; 25(6):681-9.
-
Bell S, Kempson R, Hendrickson M. Problematic uterine smooth muscle neoplasms. A clinicopathologic study of 213 cases. Am J Surg Pathol. 1994; 18(6):535-58.
-
Bonneau C, Thomassin-Naggara I, Dechoux S, Cortez A, Darai É, Rouzier R. Value of ultrasonography and magnetic resonance imaging for the characterization of uterine mesenchymal tumors. Acta Obstet Gynecol Scand. 2014; 93(3):261–8.
-
Vilos GA, Marks J, Ettler HC, Vilos AG, Prefontaine M, Abu-Rafea B. Uterine smooth muscle tumors of uncertain malignant potential: diagnostic challenges and therapeutic dilemmas. Report of 2 cases and review of the literature.J Minim Invasive Gynecol. 2012; 19(3):288–95.
-
Guntupalli SR, Ramirez PT, Anderson ML, Milam MR, Bodurka DC, Malpica A. Uterine smooth muscle tumor of uncertain malignant potential: A retrospective analysis. Gynecol Oncol. 2009; 113(3):324–6.
-
Shapiro A, Ferenczy A, Turcotte R, Bruchim I, Gotlieb WH. Uterine smooth-muscle tumor of uncertain malignant potential metastasizing to the humerus as a high-grade leiomyosarcoma. Gynecol Oncol. 2004; 94(3):818–20.
-
Ip PPC, Tse KY, Tam KF. Uterine smooth muscle tumors other than the ordinary leiomyomas and leiomyosarcomas: A review of selected variants with emphasis on recent advances and unusual morphology that may cause concern for malignancy. Adv Anat Pathol. 2010; 17(2):91–112.
-
O’Neill CJ, McBride HA, Connolly LE, McCluggage WG. Uterine leiomyosarcomas are characterized by high p16, p53 and MIB1 expression in comparison with usual leiomyomas, leiomyoma variants and smooth muscle tumours of uncertain malignant potential. Histopathology. 2007; 50(7):851–8.