Diagnosticul adenomiozei uterine la pacientele de vârstă reproductivă
Diagnosis of uterine adenomyosis in patients of reproductive age
Abstract
Uterine adenomyosis is a gynecologic condition characterized by the presence of islands of ectopic endometrial tissue inside the myometrium. This disease affects 20% of women of reproductive age, causing pelvic pain, abnormal uterine bleeding and infertility. This paper is a review based on information found in literature. The analysis was limited to articles in English language published between January 1st, 2000 and November 13th, 2019 on PubMed. In this review, we discuss the current trends in the management of uterine adenomyosis, especially ultrasound and MRI diagnosis. Adenomyosis remains an underdiagnosed condition. Through a careful description of the sonographic aspects of adenomyosis and using a standardized classification, we can improve the diagnosis rates in women of reproductive age.Keywords
adenomyosistransvaginal ultrasonographymagnetic resonanceRezumat
Adenomioza uterină este o patologie ginecologică ce este caracterizată de prezenţa insulelor ectopice de ţesut endometrial în miometru. Această boală afectează 20% dintre femeile de vârstă reproductivă, cauzând durere pelviană, sângerare anormală uterină şi infertilitate. Această lucrare reprezintă o revizie a literaturii, prin intermediul platformei PubMed. Analiza a fost limitată la articole în limba engleză, publicate între 1 ianuarie 2000 şi 13 noiembrie 2019. În această recenzie a literaturii am discutat tendinţele curente în managementul adenomiozei uterine, punând accentul pe rolul ecografiei şi al rezonanţei magnetice în diagnosticul afecţiunii. Adenomioza rămâne o boală subdiagnosticată. Printr-o descriere atentă a aspectelor ecografice ale adenomiozei şi prin utilizarea unei clasificări, putem îmbunătăţi ratele de diagnostic la femeile de vârstă reproductivă.Cuvinte Cheie
adenomiozăecografie transvaginalărezonanţă magnetică nuclearăIntroduction
Adenomyosis, an underdiagnosed condition, usually reveals an enlarged and globular uterus with areas of ectopic endometrial tissue (endometrial glands and/or stroma), located inside the myometrium, associated with hypertrophy and hyperplasia of the myometrial stroma(1). Two main theories have been proposed to explain the origin and pathogenesis of adenomyosis: the first theory relies on the invagination of the endometrial basalis into the myometrium as a result of activation of the tissue injury and repair mechanism (TIAR), while the second theory states that the adenomyotic lesions result from metaplasia of displaced embryonic pluripotent müllerian residues or differentiation of adult stem cells(2). The importance of the diagnosis of this pathology is due to the fact that adenomyosis is often associated with hormone-dependent pelvic lesions(3). The symptomatology is present in one of three women, and its severity is correlated with increasing ectopic foci and the expansion of the invasion(4,5). The typical symptoms include abnormal uterine bleeding, pelvic pain and infertility(6). Until the introduction of noninvasive ultrasound and magnetic resonance techniques, the diagnosis was relying on histopathologic examination of post-hysterectomy specimens. Since then, several studies have confirmed the high sensitivity and specificity for both two-dimensional transvaginal sonography (TVS) and magnetic resonance imaging (MRI)(7-12). In this review we discuss the management of uterine adenomyosis with a special focus on the role of ultrasound and MRI in diagnosis. Multiparous status has a major role in the development of adenomyosis because under the action of the trophoblast in the myometrium, the junctional zone may be interrupted mechanically(14). Oral contraceptives are not associated with adenomyosis, but it has been found more common in women who use the selective estrogen receptor modulator tamoxifen(13).
Diagnosis
The gold standard for the diagnosis of adenomyosis is the histological examination of a hysterectomy specimen. On histological examination, adenomyosis is classified into two major categories: focal, in the presence of circumscribed nodular aggregates, and diffuse, when the endometrial glands or stroma are distributed diffusely within the myometrium. Focal adenomyosis is different from adenomyoma. Pathologists define this as focal adenomyosis with additional compensatory hypertrophy of the surrounding myometrium. The implementation of modern imaging techniques, such as transvaginal ultrasonography (TVUS) and magnetic resonance imaging, has granted a noninvasive way to diagnose adenomyosis(5-8). The ultrasonography technique is widely available in an office setting, it is relatively economical, it requires no preparation, it has no contraindications and it is relatively accurate, being the preferred imaging technique in gynecology.
The role of ultrasound and MRI
in diagnosis
Adenomyosis can be diagnosed with a high degree of accuracy with the rise of high-resolution imaging techniques. The imaging signs revealed with endovaginal ultrasonography (US) and magnetic resonance imaging correlate with various aspects of this disease at histopathologic analysis.
Reinhold et al. reported that the sensitivity of endovaginal US was 80-86%, the specificity was 50-96%, and the overall accuracy was 68-86%. Several studies have demonstrated MRI to be highly accurate in the diagnosis of adenomyosis, with a sensitivity and specificity of 86-100%, and an overall accuracy of 85-90.5%(7-9).
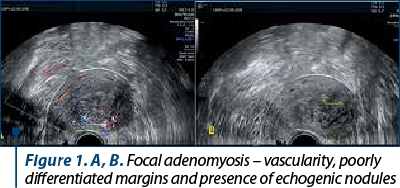
The ultrasound features specific for adenomyosis described in the literature are very various and subtle. In cases with focal adenomyosis, sonographic images may have similarities to those of leiomyomas. The arguments for the diagnosis of adenomyosis presented by Timmerman in 2003 included: uneven ultrasound texture and poorly differentiated margins, presence of elliptical shape at the expense of globular shape, lack of edge shadowing, presence of echogenic nodules, absence of circular vascularity on the edge of the lesion and presence of linear striations from the endometrium to the myometrium, along with the absence of vascularization at the edge of the lesion (Figure 1)(14).
Subsequently, the MUSA (Morphological Uterus Sonographic Assessment), a consensus statement on terms, definitions and measurements to describe the sonographic features of the myometrium, laid the foundation for studies that had different myometrial pathologies. Thereby, in the same year, Van Den Bosch et al. turned their attention to fibroids and adenomyosis. In their study, they presented schematically the ultrasound aspects, typical of adenomyosis: asymmetric growth of the anterior/posterior wall compared to its counterpart, small myometrial hypoecogenic cysts, irregular/interrupted junctional area, linear striations from inside the uterus to the outside, translesional vascularization and hyperecogenic islands at the myometrial level(15).
In 2019, Van den Bosch et al. proposed a classification of adenomyosis according to the ultrasound aspects in order to obtain clear lines for establishing the diagnosis of this disease. Thus, their classification system based on transvaginal ultrasound included the following criteria: the identification of adenomyosis according to MUSA criteria – localization of adenomyotic lesions (anterior, posterior, left lateral, right lateral, fundic), focal/diffuse type, cystic/non-cystic lesions, myometric layer involved (junctional area, myometrium, serosal involvement), the spread of the disease (<25%; 25-50%; >50% of the uterine volume), the size of the lesion(16).
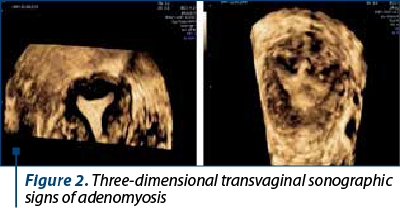
Three-dimensional transvaginal sonographic signs of adenomyosis are based on the evaluation of the junctional area on the acquired volume of the uterus in order to obtain the coronal view. In comparison with the conventional two-dimensional transvaginal sonography (2D-TVS), three-dimensional transvaginal sonography (3D-TVS) seems to be more accurate in detecting adenomyosis (Figure 2).
As mentioned before, magnetic resonance imaging can diagnose adenomyosis with high accuracy. MRI features of adenomyosys include: tiny myometrial cysts, jonctional zone thickening ≥12 mm, abnormal myometrial signal intensity, large regular asymmetric uterus without leiomyomas, myometrial foci or high signal intensity on T1 weighted images(17).
Hysteroscopy is a method of diagnosis and treatment of adenomyosis with the major advantage of allowing the direct visualization of the uterine cavity, as well as taking biopsies for the histopathological examination. It should be noted, however, that hysteroscopy has the disadvantage of having limited access to the superficial level of the endometrium. The suggestive aspects for adenomyosis that can be detected hysteroscopically are: the irregular endometrium that associates tiny openings on the surface, hypervascularization, strawberry pattern, dark blue or chocolate colored bleeding cystic lesions. By hysteroscopy, it is possible to remove superficial focal adenomyosis or to eliminate hemorrhagic cystic lesions with a diameter of less than 1.5 cm with the use of the bipolar electrode. A minimally invasive myometrial resection can also be performed. It should not be overlooked that resectoscopic treatment is not indicated for women who want a future pregnancy. As an adjuvant or alternative therapy, it can be considered a local medical therapy by implanting a levonorgestrel-releasing intrauterine device. The continuous elimination of levonorgestrel in the uterine mucosa is meant to induce regression of adenomyotic lesions and improve the symptomatology(18,19).
Conclusions
With the introduction of newer and better performing indirect imaging techniques, adenomyosis has become a pathology that can be diagnosed in a presurgical phase. The clinical interpretation is considered difficult due to the high incidence of concomitant pathology such as endometriosis and fibroids. With the new methods of ultrasound and magnetic resonance imaging and using a standardized classification system and histological confirmation, we can diagnose adenomyosis.
Conflict of interests: The authors declare no conflict of interests.
Bibliografie
-
Donnez J, Donnez O, Dolmans MM. Introduction: uterine adenomyosis, another enigmatic disease of our time. Fertil Steril. 2018; 109(3):369-70.
-
García-Solares J, Donnez J, Donnez O, Dolmans M. Pathogenesis of uterine adenomyosis: invagination or metaplasia? Fertil Steril. 2018; 109(3):371-79.
-
Levy G, Dehaene A, Laurent N, Lernout M, Collinet P, Lucot JP, Lions C, Poncelet E. An update on adenomyosis. Diagn Interv Imaging. 2013; 94(1):3-25.
-
Levgur M, Abadi MA, Tucker A. Adenomyosis: symptoms, histology, and pregnancy terminations. Obstet Gynecol. 2000; 95(5):688-91.
-
Sammour A, Pirwany I, Usubutun A, Arseneau J, Tulandi T. Correlations between extent and spread of adenomyosis and clinical symptoms. Gynecol Obstet Invest. 2002; 54(4):213-6.
-
Gordts S, Grimbizis G, Campo R. Symptoms and classification of uterine adenomyosis, including the place of hysteroscopy in diagnosis. Fertil Steril. 2018; 109(3):380-388.e1.
-
Habiba M, Benagiano G (Eds). Uterine adenomyosis. Springer. 2016.
-
Reinhold C, Tafazoli F, Mehio A, et al. Uterine adenomyosis: endovaginal US and MR imaging features with histopathologic correlation. Radiographics. 1999; 19:S147–60.
-
Dueholm M, Lundorf E, Hansen ES, Sørensen JS, Ledertoug S, Olesen F. Magnetic resonance imaging and transvaginal ultrasonography for the diagnosis of adenomyosis. Fertil Steril. 2001; 76(3):588–94.
-
Bazot M, Cortez A, Darai E, et al. Ultrasonography compared with magnetic resonance imaging for the diagnosis of adenomyosis: correlation with histopathology. Hum Reprod. 2001; 16(11):2427–33.
-
Champaneria R, Abedin P, Daniels J, Balogun M, Khan KS. Ultrasound scan and magnetic resonance imaging for the diagnosis of adenomyosis: systematic review comparing test accuracy. Acta Obst Gynecol Scand. 2010; 89(11):1374–84.
-
Shwayder J, Sakhel K. Imaging for uterine myomas and adenomyosis. J Minim Invasive Gynecol. 2014;21(3):362-76.
-
Cohen I, Beyth Y, Tepper R, Figer A, Shapira J, Cordoba M, Yigael D, Altaras MM. Adenomyosis in postmenopausal breast cancer patients treated with Tamoxifen: A new entity? Gynecol Oncol. 1995; 58(1):86-91.
-
Devlieger R, D’Hooghe T, Timmerman D. Uterine adenomyosis in the infertility clinic. Hum Reprod Update. 2003; 9(2):139-47.
-
Van den Bosch T, Dueholm M, Leone FP, Valentin L, Rasmussen CK, Votino A, Van Schoubroeck D, Landolfo C, Installé AJ, Guerriero S, Exacoustos C, Gordts S, Benacerraf B, D’Hooghe T, De Moor B, Brölmann H, Goldstein S, Epstein E, Bourne T, Timmerman D. Terms, definitions and measurements to describe sonographic features of myometrium and uterine masses: a consensus opinion from the Morphological Uterus Sonographic Assessment (MUSA) group. Ultrasound Obstet Gynecol. 2015; 46(3):284-98.
-
Van den Bosch T, de Bruijn AM, de Leeuw RA, Dueholm M, Exacoustos C, Valentin L, Bourne T, Timmerman D, Huirne JAF. Sonographic classification and reporting system for diagnosing adenomyosis. Ultrasound Obstet Gynecol. 2019; 53(5):576-82.
-
Agostinho L, Cruz R, Osório F, Alves J, Setúbal A, Guerra A. MRI for adenomyosis: a pictorial review. Insights Imaging. 2017; 8(6):549–56.
-
Vannuccini S, Luisi S, Tosti C, Sorbi F, Petraglia F. Role of medical therapy in the management of uterine adenomyosis. Fertil Steril. 2018; 109(3):398-405.
-
Di Spiezio Sardo A, Calagna G, Santangelo F, Zizolfi B, TanosV, Perino A, Leon De Wilde R. The role of hysteroscopy in the diagnosis and treatment of adenomyosis. Biomed Res Int. 2017; 2518396.