Placenta accreta spectrum (PAS), also known as “morbidly adherent placenta”, represents a pathological entity that includes different types of abnormal placental implantation. The worldwide incidence of PAS is increasing rapidly, with the rise in the number of caesarean sections performed. More than 90% of PAS cases occur in women with at least one previous caesarean delivery. This pathological condition is associated with a high rate of maternal morbidity and mortality, presenting a unique challenge for its diagnosis and management. The major symptom is represented by antepartum vaginal bleeding. The diagnosis of PAS is made by imaging methods such as transabdominal or transvaginal ultrasound and magnetic resonance imaging. From the point of view of management, the timely confirmation of the diagnosis is vital to plan the delivery and to avoid a major life-threatening postpartum hemorrhage.
Actualizări privind spectrul de placentă accreta – de la diagnostic la tratament
Updates in placenta accreta spectrum – from diagnosis to treatment
First published: 30 decembrie 2023
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/ObsGin.70.4.2022.7494
Abstract
Rezumat
Spectrul de placentă accreta (PAS), cunoscut şi sub numele de „placenta cu aderenţă morbidă”, reprezintă o entitate patologică ce cuprinde diferite tipuri de implantare placentară anormală. Incidenţa la nivel mondial a PAS este în creştere rapidă, odată cu creşterea numărului de operaţii cezariene efectuate. Mai mult de 90% din cazurile de PAS se întâlnesc la femei cu cel puţin o naştere prin operaţie cezariană în antecedente. Această condiţie patologică este asociată cu o rată mare de morbiditate şi mortalitate maternă, prezentând o provocare unică privind diagnosticarea şi managementul. Simptomatologia majoră este reprezentată de hemoragia antepartum pe cale vaginală. Diagnosticul PAS se realizează prin metode imagistice, precum ecografia transabdominală sau transvaginală şi imagistica prin rezonanţă magnetică. Din punctul de vedere al managementului PAS, confirmarea în timp util a diagnosticului este vitală pentru a planifica naşterea şi pentru a evita o hemoragie post-partum majoră, care poate pune viaţa mamei în pericol.
Placenta accreta represents a pathological entity that includes different types of placental implantation anomalies, such as placenta with abnormal adhesion and invasive placenta(1-4).
The term accreta comes from the Latin language (ad + crescere), translated as growth by adherence, adhesion and attachment(5).
Accretization represents the direct adhesion of the villous tissue to the superficial myometrial layer or near the myometrial fibers, with the partial or total absence of basal decidua, the imperfect development of the fibrinoid or the Nitabuch layer(4,6). The partial or total absence of the decidua determines the absence of the physiological cleavage space.
This abnormal decidualization can be either located strictly in a certain area at the level of the implantation site, or the implantation was carried out in a uterine scar area. Therefore, partially or fully, the placental cotyledons become deeply anchored intramyometrial. The extension of the accreted placentation area as well as the depth of invasion are different from case to case(1,6,7).
Classification
1. Placenta accreta – chorionic villi invade the superficial myometrial layer.
2. Placenta increta – the invasion is deep into the myometrium.
3. Placenta percreta – the placenta penetrates through the myometrium and uterine visceral peritoneum into the neighboring viscera, most often in the bladder.
The term placenta accreta has gradually been replaced by “morbidly adherent placenta”(1).
In classical terminology, any situations in the placenta accreta-increta-percreta spectrum is called placenta accreta spectrum (PAS)(2).
Incidence and risk factors
PAS anomalies are now considered iatrogenic conditions, attributed to the increasing rates of caesarean deliveries in the last decade(1,8). More than 90% of PAS cases occur in women with at least one caesarean section delivery in the past, especially in cases with a history of placenta praevia(1,6) (Table 1). Placenta accreta represents approximately 79% of PAS cases, placenta increta represent 14% of PAS cases, and placenta percreta represents 7% of PAS cases(1).
PAS is associated with an increased risk of maternal morbidity and mortality, and therefore this entity requires multidisciplinary care. It represents the most important cause of hemorrhage and emergency peripartum hysterectomy(9).
Diagnosis
The specific symptomatology in the first and second trimesters is hemorrhage, as a result of the coexistence of PAS with placenta praevia or if hematuria is persistent in cases of placenta percreta with extension into the bladder wall. There are situations when pregnant women who don’t associate placenta praevia end up being diagnosed with PAS in the third stage of labor, when placenta with abnormal adhesion is found(1,9).
Diagnostic ultrasound
Ideally, PAS should be diagnosed antepartum in the second trimester, for an optimal conduit in the third trimester(1,9). The examinations by transabdominal and endovaginal ultrasound (Figures 1 and 2) of the placenta, in combination with color Doppler ultrasound of placental blood flow and power Doppler, represent valuable tools for the diagnosis of PAS(1,2,4,9).
To establish the diagnosis, the definitions accepted by the European Working Group on Abnormally Invasive Placenta are used(2).
PAS is suspected in the situations presented in Table 2(1,2,9).
Although it is a recent technical acquisition, three-dimensional ultrasound is a very useful tool in the diagnosis of PAS(10).
MRI diagnosis
The role of magnetic resonance imaging (MRI) in the diagnosis of PAS is comparable to that of two-dimensional ultrasound corroborated with color Doppler, performed by specialists with expertise in imaging(2,11). It has a limited role in pregnancies below 24 weeks of amenorrhea(12).
MRI is not recommended as a primary diagnostic tool, but as an adjunct to ultrasound, when the ultrasound results are equivocal, inconclusive. It’s a valuable tool in the evaluation of the anatomy, of the degree of myometrial invasion, the invasion of the ureters, the urinary bladder or the parameters, in the posterior placental locations(1,2,9).
The MRI elements that establish the diagnosis of PAS are presented in Table 3(1,9).
The timely confirmation of the PAS diagnosis is vital in order to plan the birth.

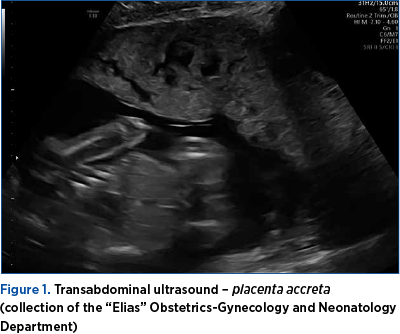
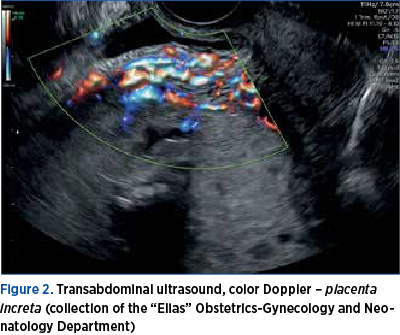


Therapeutic behavior
The therapeutic behavior in case of PAS is planned according to the same principles as those described in case of placenta praevia.
Preterm birth must be anticipated in symptomatic pregnant women, so the administration of antenatal corticosteroids to accelerate fetal lung maturation can be performed starting at 24 weeks of amenorrhea(1).
The basic principle is to plan the birth, to avoid an emergency caesarean section. Current recommendations are for elective delivery at 35-36 weeks of amenorrhea(1,2,4,8). Delivery under 34 weeks of amenorrhea is an option in pregnant women with recurrent episodes of vaginal bleeding, especially in cases of placenta percreta with invasion of the neighborhood viscera(1,8).
The recommended surgical approach is through a median incision, to allow easy conversion from caesarean section to necessary hysterectomy(8). The hysterotomy should be performed in such a way as to avoid the placenta. After the extraction of the fetus, extraction of the placenta is attempted. If abnormal adhesion is found, it is recommended to perform hysterorrhaphy and then perform necessary hysterectomy. This approach minimizes the risk of intraoperative hemorrhage(8). There are cases when it is useful to leave the placenta in its insertion area and perform the hysterectomy in a second time, as it has been reported to decrease the volume of intraoperative bleeding and the need for a massive transfusion(8,13). For women who wish to preserve fertility and only if possible intraoperatively, the placenta can be abandoned in situ, with or without the adjuvant use of methotrexate(14).
Intraoperatively, careful monitoring of hemodynamic balance, blood loss, and evaluation of hemoglobin, blood gases, electrolytes and coagulation factors are necessary. Current protocols recommend a 1:1:1 transfusion of red blood cell mass – fresh frozen plasma – platelets, antifibrinolytic therapy in the first three hours after birth (1 gram of tranexamic acid intravenous)(8,15,16).
Differential diagnosis
Although all the entities discussed belong to PAS, it’s vital to make the differential diagnosis between placenta accreta, increta and percreta. The stage diagnosis of the invasion of neighboring organs is essential before the planned surgical intervention(15).
Prognosis and complications
The prognosis is better for pregnant women with placenta accreta without placenta praevia. Accreted placenta praevia has a higher risk of intraoperative bleeding, requiring hysterectomy for hemostasis, thus increasing maternal morbidity(17).
Pregnant women with placenta percreta have a higher risk of complications compared to those with placenta accreta or placenta increta, due to the higher risk of damage to the neighboring organs, most commonly the bladder and ureters(8). n
Conflict of interests: The authors declare no conflict of interests.
Bibliografie
-
Arulkumaran S, Ledger W, Denny L, Doumouchtsis (eds). Oxford Textbook of Obstetrics and Gynaecology, Oxford University Press, 2020.
-
https://sogr.ro/wp-content/uploads/2019/06/7.Placenta-praevia.pdf.
-
Royal College of Obstetricians and Gynecologists. Placenta praevia, Placenta praevia accreta and vasa praevia: diagnosis and management, Green Top Guidelines no. 27a and 27b, London: RCOG Press 2018, September 2018.
-
https://www.uptodate.com/contents/placenta-previa-management#H3240118738.
-
Baldwin HJ, Patterson JA, Nippita TA, et al. Maternal and neonatal outcomes following abnormally invasive placenta: a population-based record linkage study. Acta Obstet Gynecol Scand. 2017;96(11):1373-1381. doi:10.1111/aogs.13201.
-
Jauniaux E, Hussein AM, Elbarmelgy RM, et al. Failure of placental detachment in accreta placentation is associated with excessive fibrinoid deposition at the utero-placental interface. Am J Obstet Gynecol. 2022;226:243.e1-10.
-
Park HS, Cho HS. Management of massive hemorrhage in pregnant women with placenta previa. Anesth Pain Med (Seoul). 2020 Oct 30;15(4):409-416. doi: 10.17085/apm.20076.
-
Shepherd AM, Mahdy H. Placenta Accreta. In: StatPearls. Treasure Island (FL): StatPearls Publishing; September 26, 2022. https://www.ncbi.nlm.nih.gov/books/NBK563288/.
-
Cunningham FG, Leveno KJ, Bloom SL, et al (eds). Williams Obstetrics. 24th Ed. McGrawHill, 2013.
-
Mulligan KM, Bartels HC, Armstrong F, et al. Comparing three-dimensional models of placenta accreta spectrum with surgical findings. Int J Gynaecol Obstet. 2022;157(1):188-197. doi:10.1002/ijgo.13743.
-
D’Antonio F, Iacovella C, Palacios-Jaraquemada J, Bruno CH, Manzoli L, Bhide A. Prenatal identification of invasive placentation using magnetic resonance imaging: systematic review and meta-analysis. Ultrasound Obstet Gynecol. 2014;44(1):8-16. doi:10.1002/uog.13327.
-
Horowitz JM, Berggruen S, McCarthy RJ, et al. When Timing Is Everything: Are Placental MRI Examinations Performed Before 24 Weeks’ Gestational Age Reliable?. AJR Am J Roentgenol. 2015;205(3):685-692. doi:10.2214/AJR.14.14134.
-
Lee PS, Kempner S, Miller M, et al. Multidisciplinary approach to manage antenatally suspected placenta percreta: updated algorithm and patient outcomes. Gynecol Oncol Res Pract. 2017;4:11. doi:10.1186/s40661-017-0049-6.
-
Sentilhes L, Ambroselli C, Kayem G, et al. Maternal outcome after conservative treatment of placenta accreta. Obstet Gynecol. 2010;115(3):526-534. doi:10.1097/AOG.0b013e3181d066d4.
-
American College of Obstetricians and Gynecologists; Society for Maternal-Fetal Medicine. Obstetric Care Consensus No. 7: Placenta Accreta Spectrum. Obstet Gynecol. 2018 Dec;132(6):e259-e275.
-
WOMAN Trial Collaborators. Effect of early tranexamic acid administration on mortality, hysterectomy, and other morbidities in women with post-partum haemorrhage (WOMAN): an international, randomised, double-blind, placebo-controlled trial [published correction appears in Lancet. 2017 May 27;389(10084):2104]. Lancet. 2017;389(10084):2105-2116. doi:10.1016/S0140-6736(17)30638-4.
-
Mulla BM, Weatherford R, Redhunt AM, et al. Hemorrhagic morbidity in placenta accreta spectrum with and without placenta previa. Arch Gynecol Obstet. 2019;300(6):1601-1606. doi:10.1007/s00404-019-05338-y.
Articole din ediţiile anterioare
Refacerea facilitată după operaţia cezariană (ERAS), recomandări preoperatorii şi intraoperatorii
Refacerea facilitată după operaţia cezariană este un concept nou, standardizat, care include un complex de măsuri pre-, intra- şi postoperatorii, c...
Infecţiile asociate plăgilor operatorii în pandemia de COVID-19: un studiu comparativ
Pandemia de COVID-19 a impus noi abordări, cu singurul scop de a prioritiza resursele în oferirea, în continuare, de servicii medicale calitative.
Placenta accreta – o preocupare tot mai mare în epidemia de operaţii cezariene
În ultimii ani, este de remarcat creşterea îngrijorătoare a numărului de operaţii cezariene efectuate la nivel global. Această intervenţie urmează ...
Sarcina cicatricială după operaţia cezariană – o continuă dilemă terapeutică. Serie de cazuri şi review al literaturii
Sarcina cicatricială după operaţie cezariană (CSP) este o tulburare iatrogenă care pune viaţa în pericol, cu o incidenţă tot mai mare, din cauza cr...