Breast and cervical neoplasm continue to represent a major public health issue in Romania, with significant differences between Romania and the European Union, as well as within Romania. In addition to the conceptual errors, we tried to identify the functional determinants of screening failure in Romania. The national program should be reshaped, focusing on the rural population, ensuring the population-based character of screening, using individual invitations through the screening register or database that must be connected to the population record database, with a fixed date of the appointment included in the invitation and a continuum of services for positive cases. The optimal solution should be to set up regional screening centers with financial autonomy, with dedicated financial resources from European funds and funds from national programs of the Ministry of Health and the Health Insurance House, with dedicated staff, well-individualized locations, with equipment intended exclusively for the conduct of screening programs, based on standardized procedures of medical practice guidelines and mechanisms that ensure quality in accordance with European guidelines.
Centre de screening regionale specializate: un proiect de reformă a screeningului în România
Specialized regional screening centers: screening reform project for Romania
First published: 28 octombrie 2021
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/ObsGin.69.3.2021.5554
Abstract
Rezumat
Cancerul mamar şi cel cervical continuă să reprezinte o problemă majoră de sănătate publică în România, cu diferenţe semnificative între România şi Uniunea Europeană, precum şi între diversele regiuni ale ţării. Pe lângă erorile conceptuale, am încercat să identificăm factorii determinanţi funcţionali ai eşecului screeningului în România. Programul naţional ar trebui să fie remodelat, concentrându-se asupra populaţiei rurale, asigurând caracterul populaţional al screeningului, utilizând invitaţii individuale prin registrul de screening sau baza de date care trebuie conectată la baza de date a evidenţei populaţiei, cu o dată fixă a chemării inclusă în invitaţie şi cu un continuu de servicii pentru cazurile pozitive. Soluţia optimă ar trebui să fie reprezentată de înfiinţarea de centre regionale de screening cu autonomie financiară, cu resurse financiare dedicate din fonduri europene şi din programele naţionale ale Ministerului Sănătăţii şi ale Casei de Asigurări de Sănătate, cu personal dedicat, locaţii bine individualizate, cu echipamente destinate exclusiv pentru desfăşurarea programelor de screening, bazate pe proceduri standardizate de orientări şi mecanisme de practică medicală care să asigure calitatea în conformitate cu orientările europene.
Cancer remains one of the leading causes of death in the European Union (EU) and around the world, 40% of the deaths being considered preventable.
On the 2nd of December 2003, in the Official Journal of the European Union, it was published the “Council Recommendation on cancer screening” by which the Council orders the development and implementation of screening programs for cervical, breast and colon cancer(1) in the member states.
Until 2012, Romania completely lacked organized cancer screening programs on a national level. Following a pilot program on a regional level that was carried out in the North-West region, a national program for the early active detection of cervical cancer by Babeș-Papanicolau testing was initiated as a screening financed from the budget of the Ministry of Health. In the latest report on the status of implementation of the European recommendation, Romania is among the few EU member states that do not have yet organized population screening programs for breast and colorectal cancer(2). The organization of a methodical screening program for breast cancer was piloted in the North-West region within a project financed through the financial mechanism Norway – EEA, but the results of this pilot program were not taken over and developed as part of an extended program. The latest report of the European Union dates from July 2020 and focuses on four main areas of cancer issues in Europe: determining factors, screening and early diagnosis, access to cancer treatment, care and research, rare forms of cancer and childhood cancer(2). The figures show a low coverage and wide variability of the implementation of cancer screening among European countries which translates into a coverage rate by mammographic examination between 17% and 84%, a coverage rate by examination for cervical cancer varying between 4% and 71%, and by a coverage rate of screening population for colorectal cancer of 1-53%. Romania declared a coverage rate with mammographic examination of 0.2%, an invitation rate of 0.2%, and a participation rate for breast cancer screening of 82%. As for cervical cancer screening, the examination coverage had a rate of 9.2%, the invitation rate was 65% and the participation rate was 14.2%(2).
Romania has the highest mortality rate from cervical cancer in Europe, a rate estimated for 2019 as being 3.7 times higher than the European average. As for mortality due to breast cancer, between 2007 and 2016 Romania registered an increase of 13%, the rate of breast cancer cases increasing during the last decade by 35% worldwide.
Analysis of the implementation issues
of national screening in Romania
Breast and cervical neoplasm continue to be major public health issues in Romania, with significant differences between Romania and EU, as well as within Romania, due to the failure to organize national screening programs for the population. Although the support of the European Union was constant and consistent, in 14 years since Romania joined the EU, pilot centers have been created and, slowly, the experience of these pilot centers has been extended, which statistically proved to be inoperative, the current screening of breast and cervical cancer in Romania remaining mainly opportunistic, incoherent and uncoordinated, imposing additional costs for patients, such as HPV vaccination program, addressed inconsistently and fragmentarily to a much too narrow segment of the population (girls aged 11 to 14 years old) to be able to diminish the prevalence and mortality of cervical cancer.
Romania suffers from insufficient medical practice focused on prevention, on early diagnosis and treatment of preneoplastic lesions, but the poor budget allocated to health does not fully explain this lack of vision on long-term population health.
There are many causes of screening failure in Romania, but they can be summarized in two major errors: the integration of the screening program in the treatment health system, and the granting of the pivotal integrative role to the family doctor within this program.
The medical service providers within the national cervical cancer screening program are represented by the health units with beds that also have, in their own structure, offices specialized in obstetrics and gynecology and medical analysis laboratory in the field of cytology. Considering that the health facilities with beds are not exclusively under the coordination of the Ministry of Health, their budgetary situation is at limit, making it very difficult to provide medical care for emergencies of chronical cases and do not have the necessary computerized systems needed for extensive databases linked to the population health records, national cancer and rare disease registers, and also they do not have the staff dedicated to conducting appointments, it is obvious that the allocation of funds dedicated to screening is lost in a mass of global shortcomings in which the acute needs will always be other than the support of an effective and sustainable screening, as the coverage rates of the eligible population prove it, in the figures for previous years.
The central operational element of the health facilities with beds is the addressability, and that of the screening system is the calling, recalling, scheduling, keeping records, covering the healthy population; the attempt to make these two different systems work simultaneously in the same locations, with the same deficient staff, has led to the malfunctioning so far of testing within the program. The specialized health facilities that are currently implementing the program do not carry out a population screening according to the European Union regulations, but an opportunistic one, testing the patients who come to the health units for other diseases or with symptoms within the gynecological sphere.
The family doctor represents another dysfunctional element within the only existing screening program in Romania. The primary healthcare facilities are not equipped with gynecological operating tables, the family doctors are not accredited and do not want to collect material for the Babeş-Papanicolaou test, and the offices are not connected to a digital network capable of storing the records of the patients. Also, their lack of involvement in taking part in the patients’ scheduling for the COVID-19 vaccination has proved their inefficiency in prophylaxis. The family doctor does not have the qualification to guide the positive patients to screening for targeted explorations, and the results of invasive explorations reach them sporadically through the medical letter brought by the patient. In addition, the journey to the family doctor for referral, to the gynecologist for harvesting, to the gynecologist for result interpretation, and back to the family doctor for referral to the medical unit of diagnosis and treatment implies a daunting time consumption for any employed woman who has to allocate five days of her leave for cervical cancer screening, without including breast cancer screening, for which she will have to address and pay a gynecological consult and a mammography. The involvement of the family doctor must gain a new status; he can detect by anamnesis the presence of abnormal uterine bleeding, he can highlight the presence of risk factors and can inform the patient about the increased risk they have of developing certain neoplasias, thus participating in the medical education of their patients, explaining to them the usefulness of primary and secondary prevention measures; he can inform about the age at which screening programs are recommended, thus contributing to their success. Informing patients about the risks, the modalities of prophylaxis and the abnormal symptoms that they should report immediately is the key role of the family doctor in the early diagnosis of genital and breast cancer.
In addition to these two conceptual errors, we tried to identify the functional determinants of screening failure in Romania:
n The main problem of screening programs in our country is the absence of the computer system and databases capable of storing patients’ records, monitoring the evolution, the five-year survival, and contact the patient for screening or monitoring consultations. Therefore, there should be set up a mandatory rule for the patient to come to regular consultations and announced in advance but, above all, the digital system must be created, funds and staff must be allocated, being essential, as underlined by European guidelines, for quality standards in population screening. As I have shown before, Romania is not only deficient in screening coverage, but also in reporting data at a national and international level.
n The poor access to medical services for women in rural areas, due to cultural, educational, geographical, economic and information barriers, creates a major social inequity, being very important, as the share of the rural population is increased in Romania. The results of previous personal studies(3) have shown the continuous presence of barriers to access preventive and curative services for the rural population, despite the fact that the legislation guarantees equal access to health services for all citizens. The analysis of cancer mortality by geographical region supports the disparities in socioeconomic development between the various regions of the country, but it should reflect in the provision of health services, as it is unacceptable for a developed European country.
n The lack of genuine national statistical indicators in the conditions of integration of the DRG reporting system and of the contractual relationship with the Health Insurance House. The statistical indicators of a disease have the role of creating an overview necessary to understand the trends of disease evolution both at a population level (incidence, prevalence, survival) and at an individual level, and also to quantify the effectiveness of the diagnosis and therapeutic approaches. It is not possible to carry out an analysis of the costs involved in genital and mammary malignant pathology for individuals, for families or for the health system because Romania doesn’t have any statistics regarding survival rates, nor any evaluation of the quality of life of these patients. The quantitative data on the state of health and the use of health services related to cancer available for Romania are fragmentary, being necessary to combine multiple sources that all together can only provide a partial picture of the impact of this pathology in the population and on the use of healthcare services.
n Cases are reported under a common diagnostic code, lacking etiological individualities; the secondary diagnosis is not fully reported. The correct and efficient way in which the observation sheets are filled should be established through protocols adapted to each health unit/facility, the full diagnosis of a disease should necessarily include the diagnosis of the main condition with its clinical form, evolutionary stage, etiological diagnosis, and full secondary diagnoses. In the case of cancers, the diagnosis must contain the stage of the disease according to the latest FIGO/TNM classification, the histological form according to the WHO classification, the degree of histopathological differentiation, the location of the metastases, if they exist, and the clinical form or the diagnosis of presentation, complemented by the diagnosis of associated diseases.
n The Ministry of Health has achieved a particularly important step in medical practice by developing, publishing and updating clinical guidelines. They are best practice recommendations of good medical care based on published scientific evidence. The guidelines must be adjusted to protocols specific to each health unit/facility. The changes in FIGO/TNM staging in genital and breast malignant pathologys are important and must be implemented. In order for all patients to equally benefit from a standardized way of diagnosing and therapy, the Ministry of Health, through specialized commissions, must create a mechanism for controlling the implementation of these guidelines. Currently, there are huge discrepancies between medical practices in rural-urban areas, between the health units of the same city and even between the doctors belonging to the same health unit/facility. The number of guidelines and protocols must increase, the medical practice must be standardized and homogeneous, so that the continuity of screening programs with coherent programs for the diagnosis and treatment of precursor lesions or early stages of genital and mammary cancers can be ensured. The mortality from genital and mammary cancer will not decrease in Romania until primary, secondary and tertiary prevention will work organically integrated and unfragmented, in a standardized and highly effective manner.
n The lack of the National Cancer Registry, whose centralized functioning has been stopped since 2008, needs to be repaired urgently. Reports of cancer cases worldwide are based on national cancer registries. The national rare disease registry (recently created) is still unfunctional, Lynch syndrome being absent in statistics, although it represents 5% of colorectal cancers and 3% of endometrial ones (about 334 new cases annually), meaning young women who would benefit from specialized genetic investigations and diagnosis, to which we can add descendants and relatives. The Health Insurance House has included in the insurances free testing to determine the mutations of BRCA genes in patients diagnosed with breast cancer; unfortunately, this very important opportunity for the detection of genetic cancer is unknown by both patients and doctors and, as long as positive cases are not included in a national genetic cancer registry, the initiative will remain sporadic, without the expected effect. The specialty of medical genetics, although present among medical specialties, has a reduced and inconsistent representation in health facilities, which must also be improved.
n Although the criteria of the international working groups IETA, IOTA, MUSA, CONUTA and IDEA for reporting the endometrial, ovarian, myometrial ultrasound examination, uterine malformations and endometriosis have been issued since 2000, they do not apply; there is still a nonhomogeneous reporting of the results and an inadequate genital examination, with major consequences in the detection of endometrial, ovarian, myometrial cancer and the extension of the cervical cancer – the endometrial thickness is measured on random sections, the examination is carried out in various phases of the menstrual cycle, the eudiometrical junction, the endometrial morphology and the characteristics of the vacuity line are not included in any ultrasound bulletin, the instillation of the saline solution even in case of suspicion of a focal lesion is a rarity, the uterine dimensions include or not the length of the cervical canal, without mentioning it, the ovarian cystic formations lack descriptive criteria suggestive of malignancy, the linearity of the cervical canal is not investigated, and myometrium or cervical invasion almost never supports the preoperative evaluation. The level of competence of the examiner of the genital sphere is not rigorously established, although ultrasonography is capable, in theory, under certain conditions, of specifying with an accuracy comparable to the one of MRI the degree of myometrium and cervical invasion, extremely important factors for the prognosis of the patient with endometrial cancer.
n Although aspiration biopsy was described by Cornier in 1984, many studies showing its diagnostic accuracy, although it is the first line of endometrial biopsy in the USA and in most European countries, although it is a reliable method with wide acceptability of patients, cheap and usable in outpatient services, the current management of abnormal uterine bleeding, the monitoring of endometrial hyperplasia under treatment and the investigation of suspicious ultrasound aspects detected incidentally at ultrasound is done by biopsy uterine curettage that involves at least day hospitalization, additional costs, anesthesia, a significantly increased risks, discomfort, with a consequent decrease in addressability, which decreases even more, considering that the examinations must be repeated, in stages, due to the duration of 3-4 weeks necessary to obtain the histopathological result of the biopsy. The proposal to use this technique is based on the universal experience accumulated in many years, the method being a beginning of specific exploration and not a final stage.
n The low level of health education is the main reason of the low addressability and, consequently, of the late detection of genital and breast cancer. Patients should know the characteristics of a normal menstrual cycle, the potential severity of any type of bleeding in pre- and postmenopausal, the risk factors involved in the development of cervical, breast or endometrial cancer and they should present to the doctor both for screening, periodical exams, and for the detected symptomatology. In this respect, the conduct of screening and early investigations should be encouraged to be carried out in an outpatient system, without burdening hospitals with unjustified costs and wasted time at the expense of diagnosing the disease in early stages.
Human and material resources necessary for the implementation of screening
and early diagnosis methods within
specialized centers
The aim of the proposed project is to improve the performance of the existing cervical cancer screening program, to initiate the screening program for breast cancer, and to create the screening program for breast and endometrium genetic cancer in order to achieve the following objectives:
n
decrease of mortality from genital and breast cancer in Romania by early detection of the disease through population screening;
n
improvement of medical practice by focusing on screening and early detection;
n
improvement of the women’s addressability and compliancy towards screening programs;
n
improvement of the medical information system.
The premises for a successful implementation of a screening program are the acceptance of the society, central and local coordination and, last but not least, the application of a scientifically proven practice. Given the complexity of the implementation process, it is not surprising that the time required for a screening program to function is at least 10 years.
To ensure the sustainability of a screening program, a permanent updating of screening methods is necessary, as well as the evaluation of their applicability for the unfolding of the program, an example being the primary HPV testing. In principle, a good screening test should be simple and easy to use. The full preventive potential of a screening test can only be obtained within the framework of an optimally functional screening program, which equates to a complex organization.
In order to facilitate the future implementation of screening programs in European countries, an expert group under the aegis of the European Science Advisory Network for Health met in February 2011 to summarize the experience of the programs already under way at the European level (Table 1).
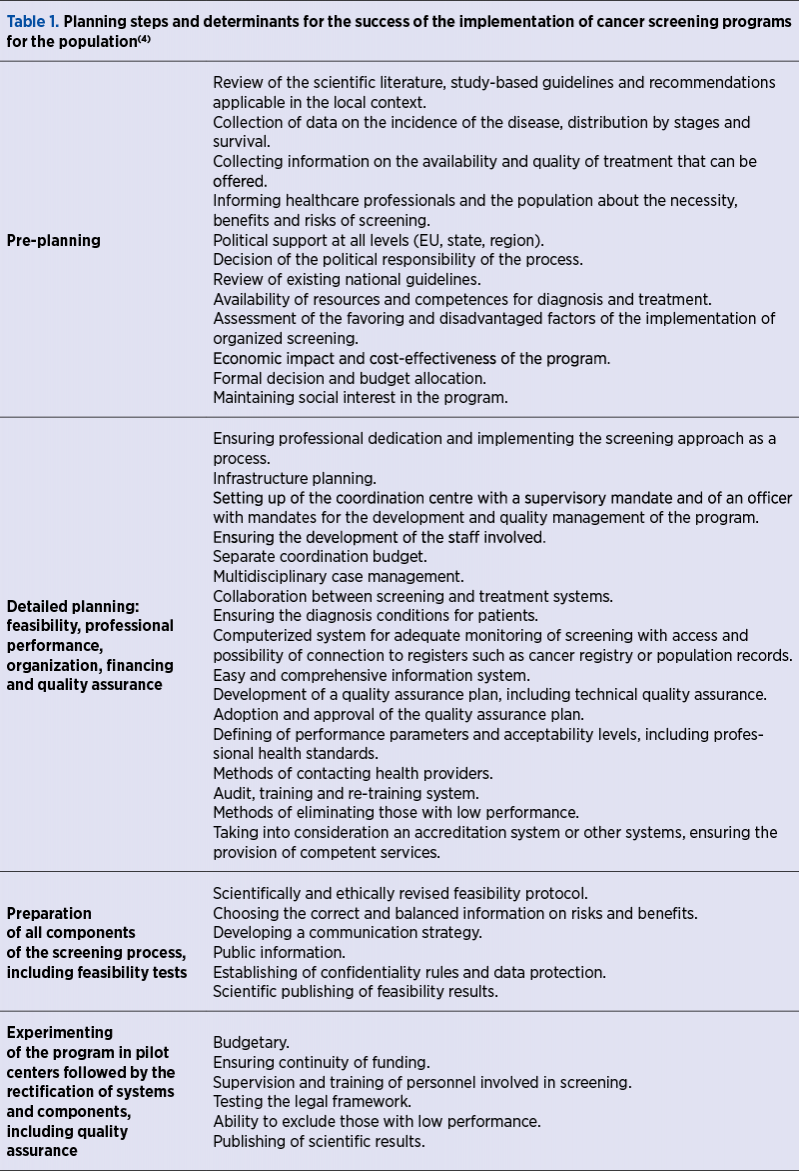
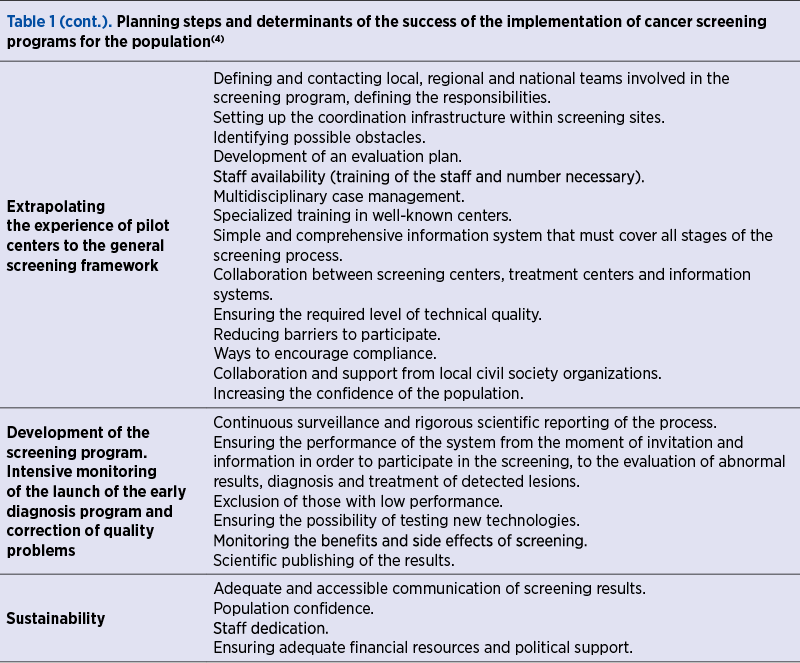
The early diagnosis and monitoring programs for cervical, breast and endometrial precursor lesions must have a distinct functionality, parallel to the screening programs which they cross when taking over and reintroducing positive patients to screening, further explored.
One vital and pragmatic question arises: what exactly should be done to improve cervical cancer control in the future in Romania? Clearly, there is a priority for cervical cancer screening in the country. The national screening program has been in place for many years, despite its failure to ensure systematic population screening and adequate participation. Certain interventions are being planned, such as the project to integrate primary screening for HPV into the national program. This project is planned to be implemented in two phases: a training and planning phase, which is being implemented, and the screening itself, which is planned to be implemented in the North-West, Centre, South and North-East regions, with at least 170,000 beneficiaries selected per region.
In my opinion, the national program should be reshaped, focusing on the rural population and, in particular, on the western, southern and south-western regions, which still have major mortality differences compared to the national level. Another essential prerequisite for the national program is to ensure the population-based character of screening, which is in line with European guidelines and Romanian legislation that guarantees universal access to preventive services for all citizens. It should also be taken into account that access to cancer screening programs must be equally available to persons that have health insurance, as well as to those who do not have it, the precarious socioeconomic environment being in itself an increased risk factor. Although it is reported as a population-based program, the current program has an unacceptably low participation rate, which is why its main steps should be reconsidered, identifying more feasible remedial mechanisms. Another important aspect is the active nature of the program, with individual invitations through the screening register or database (which does not exist in Romania) that must be connected with the population record database, with a fixed date of the appointment included in the invitation and a continuum of services for positive cases. The literature data show that active inviting with a fixed date for the screening test is the safest way to increase participation in the program by 20%, up to 33% if the test can be collected by specialized and dedicated staff on the same day as the invitation is made(5,6). A joint effort of the society, aware of the importance of prevention programs, should consist in granting a paid day off by state or private employers for participation in screening, provided that the medical system ensures a common and thus efficient and justifiable centralized screening, which supports the proposal of this draft, presented before. Given the financial efforts of the state aimed at prevention programs, the increase in the percentage amount of health insurance of people who refuse to participate in screening is justifiable by the additional major costs of the health system necessary to treat the advanced stages of genital and breast cancers.
A correct and easy analysis of the screening database can be easily linked to cancer registries, the rare disease registry and mortality databases. The national cancer registry has been functioning since 2008 at a regional level and in an incomplete manner, without being an annual centralisation of activity for the national level. It should be underlined that the unavailability of a national register deprives medical practice of understanding the trends of disease evolution both at the population level (incidence, prevalence, survival) and at an individual level – the effectiveness of diagnostic and therapeutic approaches. It is therefore absolutely necessary and urgent to ensure the functioning of the cancer registry at a national level, as well as that of the already existing National Register of Rare Diseases. The confidentiality of personal data is guaranteed by Law no. 363 of 28 December 2018 on the protection of individuals with regard to the processing of personal data by the competent authorities for the purposes of prevention, discovery, investigation, prosecution and combating crimes or the execution of sentences, educational and security measures, as well as on the free movement of such data.
Informing the population about the screening must be extensive, including clear data on the nature of the testing, on the preventable disease and on the safety of the method. The increase of the level of health education of the general population has as objectives the detection of preneoplastic lesions by participating in screening and diagnosing in early stages of genital and breast cancers by increasing the addressability at the time of occurrence of the specific symptomatology. The role of the family doctor in informing patients about the screening possibilities is as important as the role of scientific reporting of screening results in order to increase the population confidence in it, to increase the performance of the program, for the dedicated medical staff, for the implementation of health policies and, last but not least, for Romania’s image in the EU. The communication campaigns informing on screening programs are currently nonexistent, therefore they must be initiated and supported consistently, as they play the simultaneous role of creating demand for services. Dedicated human, financial resources and procedures, as well as clear mechanisms to ensure quality should also be analyzed and implemented.
Given the results of the current cervical cancer screening program, the lack of the national screening program for breast cancer, the lack of any screening program or early diagnosis of endometrial cancer, the lack of national registers of cancer, genetic cancer and rare diseases, but especially the serious situation of Romania regarding the prevalence and mortality especially through cervical and breast cancer, I propose the radical change of the national approach to screening, through the complete financial-operational separation of medical services intended for screening from medical services for therapeutic purposes. Conducting screening in emergency hospitals, chronic or outpatient hospitals can maintain a minimum screening rate on short term, including elderly patients with chronic conditions or disabilities who address medical services in emergency or in the context of chronic illness, but the impossibility of monitoring the screening results of these patients represents a major disadvantage for the sustainability of the program and is strongly not recommended by specialists. Therefore, I believe that the optimal solution should be to set up regional screening centers with financial autonomy, with dedicated financial resources from European funds and funds from national programs of the Ministry of Health and the Health Insurance House, with dedicated staff, well-individualized locations, with equipment intended exclusively for the conduct of screening programs, based on standardized procedures of medical practice guidelines and mechanisms that ensure quality in accordance with European guidelines. Setting up these autonomous centers will pave the way and provide the material basis for the functioning of other screening programs, with colorectal cancer already being a requirement of the European Union, and with other lung and prostate cancer programs currently under evaluation. The grouping under the same institutional roof of several specialties will make possible the multidisciplinary approach to the case, constantly recommended by the EU and impossible to achieve in the current system in which theoretically all information is centralized at primary medicine level. In addition, primary prevention through HPV vaccination could gain promotion and confidence, an appropriately administered location and genuine records of vaccinated people, able to statistically support the reporting of adverse reactions and efficacy.
The immediate national strategy must include cervical cancer screening, breast cancer screening and screening for genetic cancer, with sustained efforts being needed in the field of early detection of neoplastic disease. The national cervical cancer screening program needs major revision required by the integration of the primary HPV test, the possibility of performing HPV reflex, both of which have repercussions on the age of onset of screening and on the interval between testing and vaccination that must cover an age range longer than the current one. The breast cancer screening program must be created and put into function as soon as possible. Mammography performed every two years from 45-75 years old is the primary intervention that will bring on a major decrease in the mortality of women in Romania in the next period. The screening for endometrial cancer of asymptomatic women with a genetic diagnosis of Lynch syndrome (HNPCC)/Lynch syndrome family history (HNPCC)/family aggregation of colorectal or endometrial cancer without a genetic diagnosis of Lynch syndrome (HNPCC) consists of endometrial biopsy performed annually from the age of 30-35 years old or 5-10 years before the earliest age of diagnosis of a form of Lynch syndrome-related cancer (HNPCC) in the family. Performing transvaginal ultrasound examination is complementary to endometrial biopsy and should be performed annually, starting from the same age. For asymptomatic women with a family history of Lynch syndrome or with familial aggregation of colorectal or endometrial cancer without a genetic diagnosis of Lynch syndrome (HNPCC), genetic consultation and genetic testing of MMR gene mutations are recommended. For patients with Cowden syndrome, the increased risk of endometrial cancer that they have justifies endometrial biopsy screening starting at the age of 35-40 years old or five years earlier than the age of diagnosis of the first case of endometrial cancer in the family and annual transvaginal ultrasound in postmenopausal.
Defining of target population
n
The population of both sexes aged between 11 and 17 years old for the purpose of HPV vaccination.
n
Women aged 25-64 years old, insured and uninsured, who do not have a confirmed diagnosis of cervical cancer, are asymptomatic, have no history suggestive of cervical cancer pathology, in view of cervical cancer screening through primary HPV testing or Babeş-Papanicolaou cytology, with reflex testing of positive cases.
n
Women aged 45-75 years old, insured and uninsured, asymptomatic, who do not have a confirmed diagnosis of breast cancer, in order to screen for breast cancer by digital mammography.
n
Women of any age, insured or uninsured, carriers of BRCA gene mutations or with confirmed Lynch/Cowden syndromes, and their first-degree relatives, for breast and endometrial cancer screening by mammography, endometrial biopsy and genetic testing, respectively.
In order to implement the proposed changes, there is a clear need for very extensive changes of the legislation, infrastructure, as well as a massive mobilization of human and financial resources that must be included in the public policies to be developed on medium and long term.
The tests that the screening center must perform are: primary HPV test, Babeş-Papanicolaou cytology, colposcopy, histological examination with immunohistochemistry of biopsy specimens, digital mammography, ultrasound, immunohistochemical tests aimed to loss or structural alteration of proteins encoded by MMR genes, PCR tests for the presence of microsatellite instability (MSI) and the presence of mutations of PTEN or BRCA genes 1 and 2.
The material base must have its own certified laboratory that includes equipment dedicated to the histopathological examination of paraffin, sections of ice and immunohistochemistry and genetics laboratory equipped with PCR, gynecology offices equipped with gynecological tables, colposcopes and ultrasound devices, along with radiology equipment for digital mammography. The most important investment is in the technical field of artificial intelligence, the computerized system of the National Screening Registry having to ensure the secure database of patients, the retrieval and storage of information from the laboratories and equipment of the center, the connection with the databases of the health units designated to take over, diagnose and treat positive cases at screening and, last but not least, the connection with the National Cancer Registry and the National Registry of Rare Diseases.
The human resources dedicated to the center must include specialists in obstetrics and gynecology, imagistics, pathological anatomy, laboratory medicine and medical genetics, nurses trained in the collection of cervical samples, radiology nurses, medical genetics laboratory assistants, and staff dedicated to the call/recall programming center, able to ensure the operationality of the National Screening Register and its connections with the population record, the National Cancer Registry and the National Register of Rare Diseases.
Under these circumstances and taking into account the screening programs designed to be implemented and the forecasted level of coverage, I believe that there is a need for coordination of efforts on the part of all stakeholders, in order to improve access to primary and secondary prevention programs, in screening centers that are coherently organized and have feasible indicators to evaluate the activity and performance, which necessarily include a better degree of coverage for the population at risk. In the absence of coordinated actions, the health gap between Romania and the EU will increase, which is not in our country’s favor.
Romania should use the existing opportunities to improve its screening program in the years to come. Due to similar problems in Central and Eastern Europe, specific EU support for members in this geographical area could help minimize differences in cervical and breast cancer mortality between EU members.
Conclusions
The development of modern medicine from the point of view of the technical and informational possibilities, of integrating the individuals in a program that involves monitoring, screening and prophylaxis of their health, the development of the concept of interdisciplinary collaboration in the approach of various pathologies also imposes, in the cases of genital and breast cancers, the need to update the plan of detection and monitoring of preneoplastic conditions, of the high-risk population, of preclinical forms, as well as a shift to a higher level of the therapeutic approach in terms of the application of scientifically proven knowledge accumulated over the past few years. Currently, the very real possibility of the relationship of gynecology with anatomopathological, genetics, immunohistochemistry, imaging and oncology represents a consistent motivation for trying to develop algorithms of early diagnosis with certain therapeutic and prognostic clinical repercussions for genital and breast malignant pathology in the future.
From my professional maturity, I believe that the development of the Romanian society and of the medical education in the last 30 years will allow the medical system to repolarize from the area of medical care of advanced cases of the disease to the area of primary and secondary prevention, in accordance with the global efforts, increasing the duration and the quality of life of individuals in populations whose health will be so greatly improved.
Conflict of interests: The authors declare no conflict of interests.
Bibliografie
-
European Council. Council recommendation of 2 December 2003 on cancer screening (2003/878/EC). Off J Eur Union. 2003;327:34–38.
-
Ponti A, Anttila A, Ronco G, Senore C. Cancer Screening in the European Union. Report on the implementation of Council Recommendation on Cancer Screening. Brussels: European Commission, 2017. Available at: https://ec.europa.eu/health/sites/default/files/major_chronic_diseases/docs/2017_cancerscreening_2ndreportimplementation_en.pdf (accesed on 21 March, 2021)
-
Furtunescu F, Bohîlţea RE, Voinea S, Neacşu A, Pop CS. Breast cancer mortality gaps in Romanian women compared to EU after ten years of accession – Is breast cancer screening a priority for action in Romania? Experimental And Therapeutic Medicine. 2021;21:268-273. DOI: 10.3892/etm.2021.9699.
-
Lynge E, Tornberg S, von Karsa L, Segnan N, van Delden JJM. Determinants of successful implementation of population-based cancer screening programmes. European Journal of Cancer. 2012;48:734-748.
-
Forbes C, Jepson R, Martin-Hirsch P. Interventions targeted at women to encourage the uptake of cervical screening. Cochrane Database Syst Rev. 2002;(3):CD002834.
-
Yabroff KR, Mangan P, Mandelblatt J. Effectiveness of interventions to increase Papanicolaou smear use. J Am Board Fam Pract. 2003;16(3):188.
Articole din ediţiile anterioare
Screeningul cancerului de col uterin în România: este timpul pentru o schimbare
Cervical cancer is one of the most frequent female cancers in the world, mainly in the lower-resource countries. It is preventable through vaccinat...
Infecţiile ToRCH – toxoplasmoza, rubeola, citomegalovirusul şi virusul herpes simplex - abordare şi management
ToRCH – toxoplasmoza, rubeola, citomegalovirusul (CMV) şi virusul herpes simplex (HSV) – sunt cele mai frecvente cauze ale infecţiilor congenitale ...
Cunoaştere şi conştientizare privind virusul papiloma uman (HPV) în rândul adulţilor tineri – un studiu transversal
Istoric. Infecţia cu papilomavirusul uman (HPV) este una dintre cele mai frecvente boli cu transmitere sexuală şi este incriminată pentru proporţii...
Nivelurile beta gonadotropinei corionice umane şi ale proteinei plasmatice A asociate sarcinii – markeri târzii de implantare şi placentaţie anormale. Comparaţie între sarcinile spontane cu feţi unici şi cele obţinute prin tehnici de reproducere uman
During late first trimester, between 11-13 weeks of gestation, it is recommended for all pregnant women to have a screening for Down syndrome and f...