Carcinomul nazofaringial avansat şi evoluţia cu tratamentul medical
Advanced nasopharygeal carcinoma with emphases to medical treatment
Abstract
Nasopharyngeal carcinoma is one of the most common of head and neck cancers. Most nasopharyngeal carcinoma have a histology cancers of squamous carcinoma. This paper presents the recommendations concerning diagnosis and treatment of ESMO this cancer, and recent studies have brought to the fore the therapy based on immunotherapy of PD1 inhibitors. Thus, the study with pembrolizumab led to superior results with current treatments and to maintaining these results over a longer period of time compared with chemotherapy.
Keywords
nasopharyngeal carcinomachemotherapyimmunotherapytherapeutic managementRezumat
Cancerul nazifaringian este unul din cele mai frecvente cancere din sfera cancerelor de cap şi gât. Cele mai multe cancere nazofaringiene au o histologie de carcinoma scuamos. Această lucrare prezintă recomandările ESMO privind diagnosticul şi tratamentul acestui tip de cancer şi ultimele studii care au adus în prim plan imunoterapia, bazată pe inhibitorii de PD1. Astfel, studiul cu pembrolizumab a dus la rezultate superioare tratamentelor actuale şi la menţinerea acestor rezultate pe o perioadă mai lungă de timp comparative cu chimioterapia.
Cuvinte Cheie
carcinomul nazofaringianchimioterapieimunoterapiemanagement terapeuticIntroduction
Nasopharyngeal carcinoma has its origin in the epithelium of the nasopharynx and is a rare tumor in most parts of the world. The main symptoms are neck swelling, nasal obstruction, and epistaxis, which are the most common.
The etiology of this cancer is multifactorial: genetic susceptibility, exposure to carcinogens, and prior infection with the Epstein-Barr virus (EBV)(1).
Nasopharyngeal cancer is included in head and neck cancers. More than 90% of these types of cancers are of squamous cell histology and originate in the lip/oral cavity, nasopharynx, oropharynx, hypopharynx and the larynx. Head and neck cancer are the sixth most common cancer type worldwide and there has been a significant increase in the global incidence of squamous cell carcinoma of the head and neck over the past decade. ESMO estimated that at present, more than 650,000 new cases of squamous cell carcinoma of head and neck (SCCHN) are diagnosed each year worldwide. In Europe alone, it is estimated that there are approximately 143,000 new cases and more than 68,000 deaths due to the disease each year. A percent of 85% of SCCHN appear after tobacco use and frequent and heavy consumption of alcohol together(2).
Other risk factors: recently, human papillomavirus (HPV) infection has emerged as a novel risk factor, and its prevalence in oropharyngeal carcinomas (OPCs) is growing in the United States, Canada, Western Europe, and Australia. HPV infection has been reported in 45–90% of OPCs, and the high-risk HPV16 subtype is the most prevalent(3).
Other risk factors are: UV light exposurefor cancer of the lips, occupational exposures. Nasal cancer has been associated with wood dust exposure, and squamous cell cancer of the maxillary sinus has been linked to nickel exposure. Petroleum exposure may be associated with pharyngeal cancer, but the relationship has not been proven.
An important risk factor is radiation exposure in particular for thyroid cancer and has been associated with cancer of the salivary glands.
Viruses are also an important risk factor. There is a strong correlation between EBV exposure and the appearance of nasopharyngeal cancer. The etiologic role of HPV in oropharyngeal cancer is supported by a lot of evidence. “A case-control study of 100 patients with squamous cancer of the oropharynx documented that HPV DNA type 16 was found in 72% of tumor specimens. Further, 64% of the patients had antibodies to HPV-16 oncoproteins. Oral infection with HPV increased the risk of oropharyngeal cancer with an odds ratio of 14.6 (95% CI, 6.3-36.6). This risk was independent of tobacco and alcohol use. These patients appear to have a better outcome following surgery, radiotherapy, and chemotherapy”(4).
Staging
For diagnostic and staging of nasopharyngeal cancer we must have an H&N examination and: laryngoscopy, examination under anesthesia with endoscopy, laboratory evaluation (EBV DNA, anticapsid antibodies, and serum immunoglobulin G are tumor markers for nasopharyngeal carcinomas). Posteroanterior and lateral chest X-rays should be obtained in all adult patients to eliminate the possibility of occult lung metastasis or a second primary.
The CT may highlight the extent of disease and the presence and extent of lymphatic involvement. An MRI may provide accurate information regarding the size, location, and soft-tissue extent of tumor. Positron emission tomography (PET) is useful in both primary and recurrent squamous cell carcinomas of the head and neck. Biopsies of the primary tumor often can be performed in an outpatient setting.
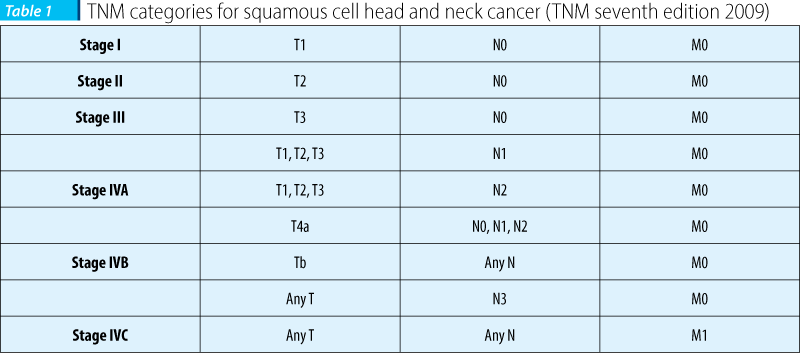
TNM staging
See Table 1.
Treatment
The treatment is multidisciplinary schedule that should be established of the first presentation of the patient. In early stages (I-II), either conservative surgery or radiotherapy (external radiotherapy or brachytherapy) give similar loco-regional control. However, this is based only on retrospective studies as there are no randomized trials available for reference. Modern radiotherapy treatment should include 3D conformal radiation therapy or intensity-modulated radiation therapy (IMRT).
Standard options for locally advanced stages III and IV tumours are: surgery including reconstruction plus postoperative radiotherapy and, for those patients found at surgery to have high-risk features (nodal extracapsular extension and/or R1 resection), post-operative chemoradiotherapy (CRT) with single-agent platinum. Combined concomitant chemoradiation is also the standard treatment in non-resectable patients.
Radiotherapy given concomitantly with cetuximab has demonstrated a higher response rate, longer disease-free progression and longer overall survival versus radiotherapy alone. There is no formal comparison between the combination of radiotherapy with cisplatin or cetuximab. In this context, the therapeutic decision is difficult to take. However, it should be considered that results of concomitant chemoradiation are based on thousands of patients, that the combination is associated with significant toxicity and that its efficacy in the elderly population is questionable. On the other hand, results of cetuximab + radiation proved to be less toxic and the benefit in the elderly subgroup is also questionable.
The role of induction chemotherapy (ICT) has been reconsidered since the introduction of taxane-platinum-based (TPF) combinations that have proved to be superior to platinum–fluorouracil PF schedule in loco-regionally advanced disease . However, at present, induction chemotherapy is not seen as a standard treatment(5).
New treatment of head and neck cancer
PD-1 immunotherapy. Nearly one-quarter of patients with advanced head and neck cancer responded to treatment with pembrolizumab (Keytruda, Merck), according to the KEYNOTE012 trial; there were 28 partial responses and one complete response. The overall response rate is double that of cetuximab (Erbitux, Bristol Myers-Squibb/Eli Lilly), which is the only targeted therapy for these patients.
In the current study, the overall response rate can be extrapolated with that seen with immunotherapy in lung cancer ( Stephen Liu, MD). NCCN and ESMO guidelines recommend initial treatment with platinum-based doublet chemotherapy with or without cetuximab. Second-line options include methotrexate, docetaxel, and cetuximab. It is known that only 10% to 13% of patients respond to single-agent cetuximab.
Pembrolizumab is also effective in HPV-negative and HPV-positive patients. “In their study, dr. Seiwert and colleagues found overall response rates with pembrolizumab of 27.2% in the 81 HPV-negative patients and 20.6% in the 34 HPV-positive patients.”
“Importantly, patients were not selected for the study on the basis of PD-L1 status. This biomarker predicts response to PD-1/PD-L1 immunotherapies such as pembrolizumab”.
The most common adverse events of any grade for pembrolizumab: fatigue (15.2%), hypothyroidism (9.1%), and rash (7.6%). Adverse events of grade 3 or higher only occurred in 9.8% of patients. Two patients developed treatment-related pneumonitis of grade 3 or higher, and one developed treatment-related colitis of grade 3 or higher.The adverse-event profile is better than that of current standard treatment (dr. Masters(6)).
MMR (miss mash repair) gene deficiency appears to be a predictor of response to pembrolizumab, and it’s very encouraging that the responses in MMR-deficient tumors thus far have been long-lasting. The data resulted from colon cancer study.
The immune-related objective response rate and the immune-related progression-free survival at 20 weeks in MMR-deficient colorectal cancer were 40% and 78%, respectively, and for MMR-deficient other cancers the rates were 71% and 67%, respectively.
High Somatic Mutation Loads Predictive
“Whole exome sequencing revealed an average of 1782 somatic mutations per tumor in MMR-deficient tumors compared with only 73 in MMR-proficient tumors (P = .0015). The high somatic mutation loads were associated with progression-free survival (P = .02). «The 1700 mutations is like putting a red flag on a cancer cell and saying to the immune system «here I am», and now allows the immune system to view these cancer cells as foreign intruders” (dr. Schuchter(7)).
Bibliografie
1. A. Psyrri; T. Rampias; J. B. Vermorken, The Current and Future Impact of Human Papillomavirus on Treatment of Squamous Cell Carcinoma of the Head and Neck
Ann Oncol. 2014;25(11):2101-2115.
2. Eugene A. Chu, MD; Julie M. Wu, MD; David E. Tunkel, MD; Stacey L. Ishman, MD
Nasopharyngeal Carcinoma: The Role of the Epstein-Barr Virus, July 16, 2008.
3. A. Psyrri; T. Rampias; J. B. Vermorken, The Current and Future Impact of Human Papillomavirus on Treatment of Squamous Cell Carcinoma of the Head and Neck,
Ann Oncol. 2014;25(11):2101-2115.
4. John Andrew Ridge, Ranee Mehra, Miriam N. Lango, and Steven Feigenberg, Head and Neck Tumors, May 01, 2014 Cancer Management - See more at: http://www.cancernetwork.com/cancer-management/head-and-neck-tumors#sthash.njSRc2Tg.dpufhttp://www.cancernetwork.com/head-and-neck-tumors
5. V. Grégoire, J.-L. Lefebvre, L. Licitra, E. Felip. On behalf of the EHNS–ESMO–ESTRO Guidelines Working Group, Squamous cell carcinoma of the head and neck: EHNS–ESMO–ESTRO Clinical Practice Guidelines for diagnosis, treatment and follow-up. http://www.esmo.org/Guidelines-Practice/Clinical-Practice-Guidelines/Head-and-Neck-Cancers/Squamous-Cell-Carcinoma-of-the-Head-and-Neck
6. Nick Mulcahy. Drug Doubles Responses in Head and Neck Cancer, May 29, 2015.
http://www.medscape.com/viewarticle/845640?src=wnl_edit_cfna&uac=127663DJ
7. American Society of Clinical Oncology (ASCO) 2015 Annual Meeting: Abstract LBA 100. Presented May 30 2015.



