Purpose. Our objectives are to analyze the prevalence of venous thrombosis (portal vein, hepatic veins, and inferior vena cava) in hepatocellular carcinoma (HCC) in correlation with the tumoral type (nodular, multicentric or infiltrative), tumoral markers and chronic liver disease etiology. Materials and method. We conducted a retrospective study that included all the patients admitted in the Fundeni Clinical Institute with the diagnosis of HCC who had imaging studies, within a three-year interval (2018-2020), out of which we selected cases in which HCC associated tumoral venous thrombosis (723 patients), analyzing tumoral aspects and their vascular implications. Results. The patients with infiltrative type of HCC had the strongest association with tumoral venous thrombus (72.72% of cases). Malignant invasion of the portal vein was detected in 17.24% of our patients. Hepatic veins are less commonly invaded by HCC, and the tumoral thrombosis was found in 6.89% of the studied cases. Conclusions. Tumoral thrombosis is one of the most common complications of advanced HCC, occurring most often in the portal vein, and more rarely in the hepatic veins, with extension into the inferior vena cava and the right atrium.
Imagistica trombozei venoase tumorale din carcinomul hepatocelular – esenţialul
Imaging of tumoral venous thrombosis in hepatocellular carcinoma – the essentials
First published: 24 martie 2021
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/OnHe.5.1.2021.4576
Abstract
Rezumat
Obiectiv. Scopul acestei lucrări este de a analiza prevalenţa trombozei tumorale (la nivelul venei porte, venelor hepatice şi al venei cave inferioare) la pacienţii cu carcinom hepatocelular (CHC), în corelaţie cu date despre tipurile de dezvoltare tumorală (nodulară, multicentrică sau infiltrativă), markerii tumorali (alfa-fetoproteina) şi etiologia bolii hepatice cronice. Materiale şi metodă. Am efectuat un studiu retrospectiv care a cuprins toţi pacienţii internaţi în Institutul Clinic Fundeni cu diagnosticul de carcinom hepatocelular, investigaţi imagistic în perioada 2018-2020 (trei ani), din care am selectat cazurile în care CHC a asociat tromboză tumorală venoasă (723 de pacienţi), analizând aspectele tumorale şi implicaţiile vasculare. Rezultate. Pacienţii cu CHC infiltrativ au avut cea mare incidenţă de tromboză venoasă tumorală (72,72% din cazuri). Invazia malignă a venei porte a fost decelată la 17,24% dintre pacienţii noştri. Venele hepatice au fost mai puţin invadate de CHC, aspectul de tromboză tumorală fiind întâlnit în 6,89% din cazurile studiate. Concluzii. Tromboza tumorală este una din cele mai comune complicaţii ale CHC avansat, apărând mai frecvent în vena portă, mai rar în venele hepatice, cu extensie în vena cavă inferioară şi atriul drept.
Introduction
Hepatocellular carcinoma (HCC) is the fifth most common cancer in the world and the third most prevalent cause of tumor-related death(1), occurring most often in patients with preexisting chronic liver disease. Imaging – especially computed tomography (CT) and magnetic resonance imaging (MRI) – plays an important role in the detection and staging of HCC. The most common imaging characteristics of HCC on both contrast CT and MRI is a lesion with arterial phase hyperenhancement and washout in the portal venous/delayed phase (seen in approximately 90% of lesions)(4,5).
Objectives
Our objectives are to analyze the prevalence of tumoral venous thrombosis – portal vein (PV), hepatic veins (HV) and inferior vena cava (IVC) – in hepatocellular carcinoma in correlation with the type of the tumor (nodular, multicentric or infiltrative), with tumoral markers and different etiologies of chronic liver disease.
Materials and method
We conducted a retrospective study, analyzing the imaging studies performed within our department, during a three-year interval (from 1.01.2018 to 30.12.2020). Within this period, there have been 4892 admissions with the diagnosis of HCC, out of which 723 patients (507 males and 215 females; mean age: 65 years old) were evaluated in our department by multislice (MS) CT, using a multiphase dynamic acquisitions (unenhanced and enhanced CT in arterial, portal and late phase), together with sagittal and coronal multiplanar reconstructions (MPR) and/or by magnetic resonance imaging (MRI) with liver specific contrast i.v. injection (Gd-EOB-DTPA) using the same acquisitions phase after contrast material (CM) i.v. injection like in CT evaluation, together with the hepatobiliary phase realized 20 minutes after the beginning of the specific CM injection. We analyzed the prevalence of tumoral thrombosis in these 723 patients and correlated the data with the type of tumoral vein thrombus (PV, HV and IVC), with tumor growth pattern, chronic liver disease and tumoral markers.
Results
In our study, out of 723 evaluated patients with HCC, 175 (24.20%) presented with tumor thrombus of the portal or hepatic venous system (hepatic veins, inferior vena cava, and right atrial invasion). Venous drainage evolves during hepatocarcinogenesis from hepatic veins (cirrhotic/dysplastic nodules and early HCCs) to portal veins (progressed HCCs).
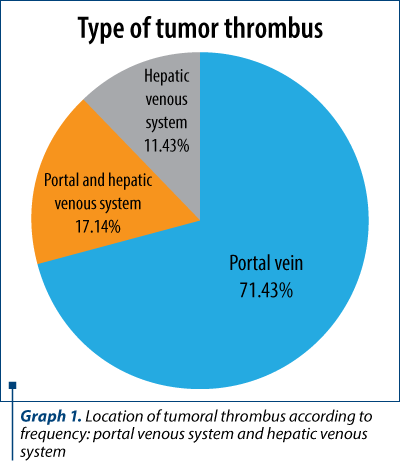
Tumoral venous thrombosis pattern. In Graph 1 we present the most common venous tumor thrombus type found in our study.
The malignant invasion of the PV is a frequent complication that occurred in up to 17.24% of our patients. The imaging characteristics of malignant thrombus are presented in Figure 1 and Figure 2:
-
direct extension of a parenchymal tumor lesion into the adjacent vessel;
-
similar CT attenuation coefficient and signal intensity of the tumoral thrombus compared with the primary liver tumor (HCC);
-
presence of arterial enhancing due to neovessels within the thrombus – “threads and streaks” appearance, in association with the dilatation of the thrombosed main PV frequently more than 23 mm;
-
the diffusion weighted imaging (DWI) must be always analyzed in correlation with the morphologic sequences pre- and post-contrast, because acute bland thrombus can also have diffusion restriction.


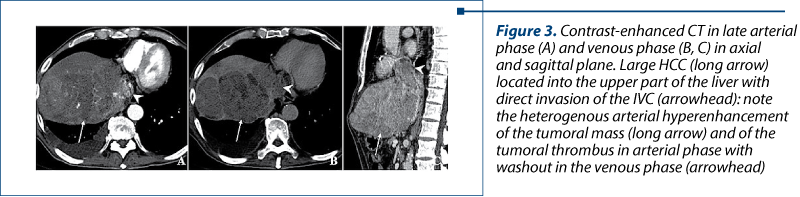
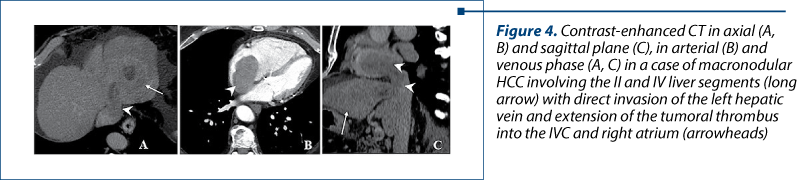
Hepatic veins are less commonly invaded by HCC; the tumoral thrombosis was found in 6.89% of our cases. In advanced disease, the tumor thrombus can propagate into the lumen of the IVC and even into the right atrium, increasing the chance of pulmonary embolism and distant metastases. Out of all the patients investigated, 6 (0.82%) had atrial invasion. The imaging characteristics of tumoral thrombosis involving the HV are like PV tumoral thrombosis, the expansion and arterial enhancement being the main characteristics (Figure 3 and Figure 4).

Chronic liver disease. In our study, the most common type of associated liver disease was chronic hepatitis C infection, followed by alcoholic hepatitis and chronic hepatitis B infection (Graph 2).
We analyzed the prevalence of tumoral thrombosis according to the main types of chronic liver disease. The most frequent association was with mixed alcoholic and viral liver disease (39.47%; 15 out of 38 patients), followed by HBV infection (28.84%; 30 out of 104 patients), then HCV infection (23.76%; 72 out of 303 patients), alcoholic nonviral (20.17%; 23 out of 114 patients) and HBV HDV infection (16.32%; 16 out of 98 patients).
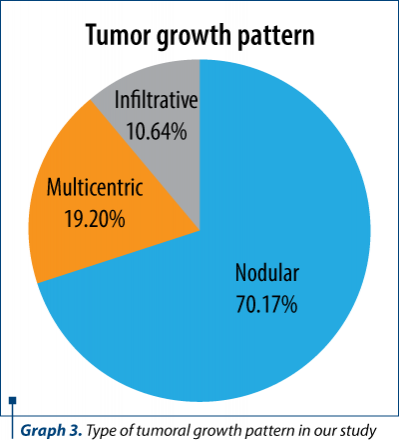
Tumor growth pattern. Macroscopically, HCC can be divided into three main growth patterns: nodular, massive/multifocal, and infiltrative. In our study, the most common type of tumor development was nodular. Graph 3 represents the prevalence of tumoral growth patterns in our study.
We analyzed the prevalence of tumoral thrombosis according to type of tumor growth (Graph 4), the patients with infiltrative type having the strongest association with tumor thrombus (72.72%; 56 out of 77 patients), followed by multifocal (35.97%: 50 out of 139 patients) and nodular (13.60%; 69 out of 507 patients).
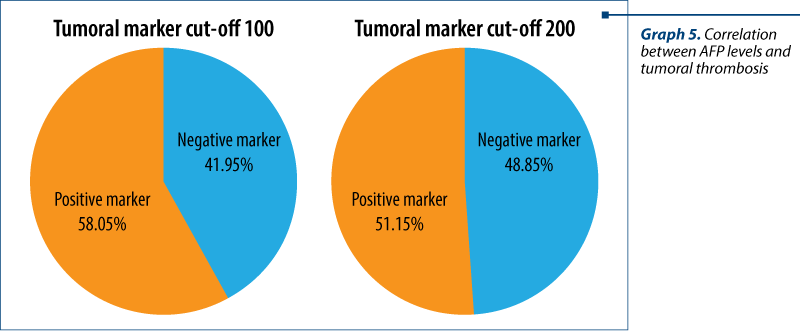
a-fetoprotein (AFP). AFP is a tumor marker discovered in 1963 that is widely used in the surveillance of cirrhosis and in the diagnosis of hepatocellular carcinoma(11). In our study, we have investigated the prevalence of high AFP levels in patients with tumoral thrombosis using two cut-off values of 200 ng/ml and 100 ng/ml, with correlations shown in Graph 4.
Discussion
The frequent implication of the portal venous system by a tumoral thrombus in HCC was reported in literature(4,6,7), also the strong association between the infiltrative type of HCC and venous invasion is consistent with other articles(2,13,15).

Patients with liver cirrhosis often develop thrombosis within venous structures(4), most frequently involving the portal venous system; adding to that the inherent hypercoagulable state of cancer patients, it means that it is important to differentiate between cruoric thrombus and malignant involvement of venous structures. Macrovascular invasion is a contraindication for curative treatment such as resection or liver transplant(1,6). Imaging is most frequently used for the noninvasive diagnosis of tumoral thrombus, because percutaneous biopsy poses a risk of bleeding especially in cirrhotic patients and may give false results when adjacent HCC mass is biopsied instead of the luminal thrombus(6). Most of the patients with HCC have a history of underlying cirrhosis or chronic liver disease(2). In our study, the most common type of associated liver disease was chronic hepatitis C infection. According to the EASL practical guidelines of HCC, the efficacy regarding AFP as a serological test in surveillance is suboptimal, because flares of HBV or HCV infections may also cause high levels of AFP and, also, only a small proportion of tumors at an early stage present with abnormal AFP levels(12). The correlation with AFP levels is still debated in literature and cannot be used as a sole diagnostic tool(12). Alcohol has a synergistic effect on preexisting viral chronic liver disease(14) that may explain why this group had the strongest association with a more aggressive behaviour and tumoral thrombus. In cirrhosis, there is a well-recognized pathway from regenerative nodule to dysplasia and ultimately HCC, this is why screening these patients is very important and, according to the EASL-EORTC recommendations, the most effective screening tool is ultrasonography performed every six months in high-risk patients. Imaging plays a critical role in the diagnosis and management of HCC because, unlike other types of cancers, it can frequently establish the diagnosis noninvasively, without the need for pathologic confirmation prior to treatment using the EASL-EORTC diagnostic criteria, which apply only in cirrhotic patients. Some patients were regularly evaluated and treated in our clinic and eventually developed venous invasion, while other had an aggressive tumor with venous invasion as an initial presentation. In our study, the most common type of tumoral thrombosis was of the portal venous system. HCV infection was the most frequent chronic liver disease found, but the mixed (viral and alcoholic) type of disease was the most strongly associated with tumoral thrombus. The infiltrative type of tumoral growth is the most common form to be linked to venous invasion, followed by multicentric type.
As a limitation of this work, we mention that the study was a retrospective one and all the imaging reports were prone to interreader variability; also, the levels of AFP were not always available at the time of tumoral thrombus diagnosis.
Conclusions
Advanced cross-sectional imaging (CT and MRI) plays an important role in the evaluation of patients with a clinical suspicion of HCC (liver cirrhosis, high AFP), allowing to make a complete evaluation of the tumor, complications and spread. The infiltrative type of HCC followed by the multicentric type are the most common forms to be linked to venous invasion. The most common type of tumoral thrombosis involves the portal venous system.
Conflicts of interests: The authors declare no conflict of interests.
Bibliografie
- Zhu K, Chen J, Lai L, et al. Hepatocellular carcinoma with portal vein tumor thrombus: treatment with transarterial chemoembolization combined with sorafenib – a retrospective controlled study. Radiology. 2014 Jul;272(1):284-93.
- Reynolds AR, Furlan A, Fetzer DT, et al. Infiltrative hepatocellular carcinoma: what radiologists need to know. Radiographics. 2015 Mar-Apr;35(2):371-86.
- Kojiro M. Histopathology of liver cancers. Best Pract Res Clin Gastroenterol. 2005;19(1):39–62.
- Shriki JE, Seyal AR, Dighe MK, et al. CT of Atypical and Uncommon Presentations of Hepatocellular Carcinoma. Am J Roentgenol. 2015 Oct;205(4):W411-23.
- Lee JH, Lee JM, Kim SJ, et al. Enhancement patterns of hepatocellular carcinomas on multiphasic multidetector row CT: comparison with pathological differentiation. Br J Radiol. 2012; 85:e573–e5835.
- Karaosmanoglu AD, Onur MR, Uysal A, et al. Tumor in the veins: an abdominal perspective with an emphasis on CT and MR imaging. Insights Imaging. 2020 Mar 25;11(1):52.
- Choi JY, Lee JM, Sirlin CB. CT and MR imaging diagnosis and staging of hepatocellular carcinoma: part II. Extracellular agents, hepatobiliary agents, and ancillary imaging features. Radiology. 2014 Oct;273(1):30-50.
- Sneag DB, Krajewski K, Giardino A, et al. Extrahepatic spread of hepatocellular carcinoma: spectrum of imaging findings. Am J Roentgenol. 2011 Oct;197(4):W658-64.
- Smillie RP, Shetty M, Boyer AC, et al. Imaging evaluation of the inferior vena cava. Radiographics. 2015 Mar-Apr;35(2):578-92.
- Nowicki TK, Markiet K, Szurowska E. Diagnostic Imaging of Hepatocellular Carcinoma – A Pictorial Essay. Curr Med Imaging Rev. 2017;13(2):140-153.
- Farinati F, Marino D, De Giorgio M, et al. Italian Liver Cancer (ITALICA) Group Diagnostic and Prognostic Role of α-Fetoprotein in Hepatocellular Carcinoma. American Journal of Gastroenterology. 2006 Mar;101(3):524-532.
- European Association for the Study of the Liver. EASL Clinical Practice Guidelines: Management of hepatocellular carcinoma. J Hepatol. 2018 Jul;69(1):182-236. Erratum in: J Hepatol. 2019 Apr;70(4):817.
- Kneuertz PJ, Demirjian A, Firoozmand A, et al. Diffuse infiltrative hepatocellular carcinoma: assessment of presentation, treatment, and outcomes. Ann Surg Oncol. 2012 Sep;19(9):2897-907.
- Balogh J, et al. Hepatocellular carcinoma: a review. Journal of Hepatocellular Carcinoma. 2016 Oct 5;3:41-53.
- Kim JH, Lee JM, Yoon JH, et al. Portal Vein Thrombosis in Patients with Hepatocellular Carcinoma: Diagnostic Accuracy of Gadoxetic Acid-enhanced MR Imaging. Radiology. 2016 Jun;279(3):773-83.
Articole din ediţiile anterioare
Esenţialul despre imagistica prin rezonanţă magnetică multiparametrică în diagnosticul carcinomului hepatocelular
Carcinomul hepatocelular (CHC) este cea mai frecventă tumoră malignă primară a ficatului, asociată frecvent cu ciroza, cu o incidenţă crescândă la...