Managementul tratamentului şi urmărirea pacienţilor cu cancer de sân în perioada pandemiei de COVID-19
Breast cancer treatment and follow-up management during COVID-19 pandemic
Abstract
Breast cancer is the leading cause of cancer in women worldwide and the second cause of cancer in both sexes. In Romania, it is the leading cause of cancer, according to Cancer Country Profile 2020. Some breast cancer treatments – including chemotherapy, targeted therapies and radiation – can weaken the immune system. People with weakened immune systems or lung problems due to radiotherapy pneumonitis or with lung metastases have a much higher risk of complications if they are infected with SARS-CoV-2. On the 20th of April 2020, there were reported 2,444,209 coronavirus cases and 167,986 deaths. There are various recommendations amidst COVID-19 pandemic on the diagnostic and treatment of patients with different kinds of solid tumors. Almost all professional societies try to identify the optimal protocols for cancer facilities, to minimize the risk of SARS-CoV-2 infection in both inpatient and outpatient settings. Due to the coronavirus pandemic, routine checkups, elective, unnecessary surgeries and other procedures have been cut back or delayed to help minimize exposure to the virus which has already infected more than 2.4 million people. The recommendations were divided according to the phase of pandemic manifestations on different countries and according to priority. In this review, we want to reinforce and bring together all recommendations made by different societies involved in the treatment management of breast cancer patients, because this pandemic led to new ways of dealing cancer patients.Keywords
breast cancermanagementcovid-19Rezumat
Cancerul mamar ocupă locul întâi în privinţa tipurilor de neoplazie întâlnite la sexul feminin la nivel global, fiind a doua cauză de cancer indiferent de sex. În România este prima cauză de cancer, conform datelor raportate de Cancer Country Profile 2020. Unele tratamente utilizate în cancerul mamar – inclusiv chimioterapia, terapia ţintită şi radioterapia – pot slăbi răspunsul imunologic. Pacienţii cu reacţie imunitară deficitară, cu probleme pulmonare secundare toxicităţilor – pneumonite radice, de exemplu – sau cu metastaze pulmonare par a avea un risc mai mare de complicaţii dacă sunt infectaţi cu noul tip de coronavirus. La 20 aprilie 2020 erau raportate 2.444.209 cazuri de infecţie cu noul tip de coronavirus şi 167.986 de decese. Există o multitudine de recomandări pentru diagnosticul şi tratamentul pacienţilor oncologici în mijlocul pandemiei de coronavirus. Aproape toate societăţile profesionale încearcă să identifice protocoalele optime pentru echipele care tratează tumori solide, pentru a scădea riscul de infecţie cu SARS-CoV-2 în cazul pacienţilor spitalizaţi continuu sau al celor trataţi în ambulatoriu ori în regim de spitalizare de zi. Din cauza pandemiei, evaluările de rutină, intervenţiile chirurgicale elective, nenecesare în urgenţă, precum şi alte proceduri au fost reduse sau temporizate pentru a scădea expunerea la virus, care a afectat deja 2,4 milioane de oameni. Recomandările sunt divizate în funcţie de faza în care se află ţara respectivă în evoluţia pandemiei şi în funcţie de prioritate. În acest review urmărim să aducem la un loc recomandările diferitelor societăţi profesionale implicate în managementul pacienţilor cu cancer de sân.Cuvinte Cheie
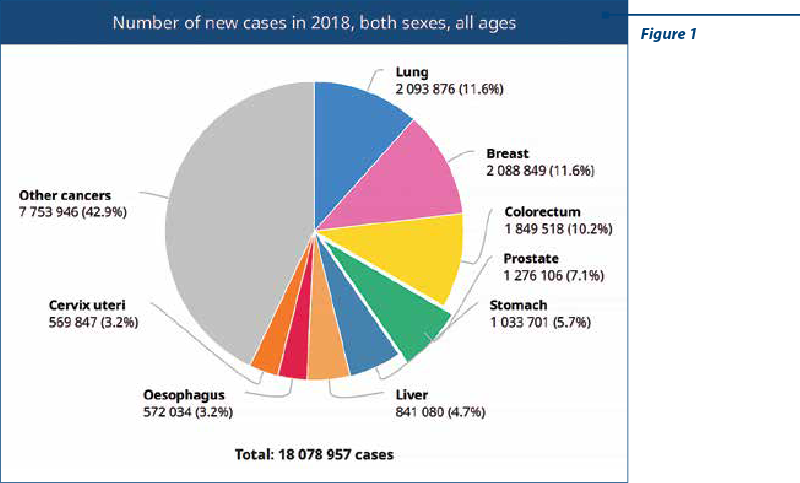
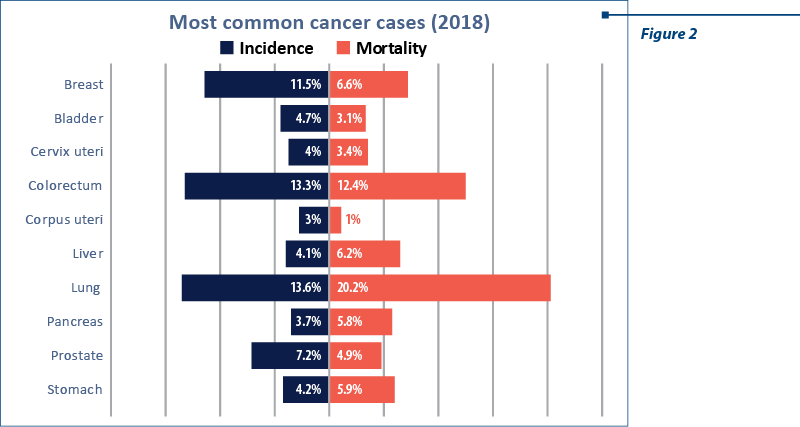
cancer de sânmanagementcovid-19Breast cancer is the leading cause of cancer in women worldwide and the second cause of cancer in both sexes (Figure 1)(1). In Romania, it is the leading cause of cancer, according to Cancer Country Profile 2020 (Figure 2)(2).
Some breast cancer treatments – including chemotherapy, targeted therapies and radiation – can weaken the immune system(3). People who have weakened immune systems or lung problems due to radiotherapy pneumonitis or with lung metastases have a much higher risk of complications if they are infected with SARS-CoV-2. For most people, the immune system recovers within a couple of months after completing these treatments, so those who have been treated for breast cancer in the past don’t necessarily have a higher risk of severe illness(4).
On the 20th of April 2020, there were reported 2,444,209 coronavirus cases and 167,986 deaths(5).
There are various recommendations amidst COVID-19 pandemic on the diagnostic and treatment of patients with different kinds of solid tumors. The ethical principle that should guide our medical decisions should be primum non nocere, respecting patient’s wish and trying to do what is best for him/her with fair allocation of resources.
Unanswered questions – such as: Do cancer patients have a higher risk of contracting the infection or of developing severe infection?, Which is the best approach for the treatment of cancer patients in the middle of COVID-19 outbreak? or What therapies can be delayed and what would be the impact on survival? – remain.
We looked at different medical societies recommendations and at recently published data, and we want to evaluate and reinforce the recommendations on breast cancer patients’ management.
Almost all professional societies try to identify the optimal protocols for cancer facilities, to minimize the risk of SARS-CoV-2 infection in both inpatient and outpatient settings. The Romanian National Society of Medical Oncology wrote also a statement reported on the health ministry page(7).
In a retrospective cohort study published in Annals of Oncology, Zhang et al. described the characteristics and outcomes of 28 patients with COVID-19 and cancer from three hospitals in Wuhan, China(6), and showed that cancer patients were at high risk for severe events and mortality.
William K. Oh, MD at the Icahn School of Medicine at Mount Sinai, recommended caution in interpreting findings reported from China due to a 1.7-fold increased prevalence than that from the Chinese population of the same age observed in American population, and the mortality rate in these patients is more than 10 times higher in the population from New York than that reported in all COVID-19 infected patients in China(6).
Due to coronavirus pandemic, routine checkups, elective, unnecessary surgeries and other procedures have been cut back or delayed to help minimize exposure to the virus that has already infected more than 2.4 million people.
The recommendations were divided according to phase of pandemic manifestations on different countries and according to priority.
The COVID-19 Pandemic Breast Cancer Consortium (CPBCC)(8) divided recommendations according to phase of pandemic manifestations that different countries experience at different moments in time, phase I meaning that hospitals have few COVID-19 patients, their resources are not exhausted, they have intact ICU ventilator capacity, and the COVID-19 trajectory is not in rapid escalation phase. In phase II, cancer and general hospitals have many COVID-19 patients, ventilator capacity and intensive care unit (ICU) resources are limited or the SARS-CoV-2 severe infection trajectory is in a rapidly escalating phase. Phase III is characterised by rolling out hospital resources to COVID-19 patients and no ventilator or ICU capacity(8).
CPBCC 2020 guidelines refer only to surgery and recommend in phase I (semiurgent) to be addressed to surgery cases that need to be operated as soon as possible, and include patients which, if delaying surgery, their breast cancer-related survival will be affected:
-
patients who finished neoadjuvant treatment (neoadjuvant chemotherapy and targeted therapy);
-
patients with clinical stage T2 or N1 estrogen receptor (ER)-positive/progesterone receptor (PR)-positive/HER2-negative tumors;
-
patients with adverse features such as triple negative or HER2-positive tumors;
-
patients with discordant biopsies likely to be malignant;
-
patients with operable malignant recurrence.
For other subgroups of patients, there could be considered alternative treatment approaches:
-
patients whose tumours can receive hormonal therapy without impacting their breast cancer-related survival – clinical stage T1N0 ER+/PR+/HER2 – and some women with clinical stage T2 or N1 ER+/PR+/HER2-;
-
patients with triple negative and HER2+ tumours who could start neoadjuvant therapy prior to surgery;
-
patients with inflammatory and locally advanced breast cancers who should receive neoadjuvant therapy prior to any surgery.
-
In phase II, CPBCC 2020 guidelines recommend restricting surgery to patients whose survival is likely to be compromised if their procedure is not performed within the next few days, and include the following cases:
-
incision and drainage of breast abscess;
-
evacuation of a hematoma;
-
revision of an ischemic mastectomy flap;
-
revascularization/revision of an autologous tissue flap (however, autologous reconstruction should be deferred).
Mainly, here are cases that are considered priority A according to Pandemic Planning Clinical Guidelines for Patients with Cancer, from Ontario Health Cancer Care.
In phase III, guidelines recommend restricting surgery to patients whose survival is likely to be compromised if their procedure is not performed within the next few hours and are similar to recommendations seen in phase II (evacuation of a hematoma, revision of an ischemic mastectomy flap, revascularization/revision of an autologous tissue flap)(13).
At the “Prof. Dr. Alexandru Trestioreanu” Institute of Oncology Bucharest, there are recommendations to treat with priority:
-
patients with triple negative breast cancer who have finished their neoadjuvant treatment that cannot be continued and no radiotherapy is recommended;
-
patients with negative biopsies, but with suspicion on clinical exams and radiological exams of malignancy;
-
local recurrences.
Cases that can be postponed:
-
patients with HER2-positive breast cancer who finished their neoadjuvant chemotherapy and can continue neoadjuvant treatment with anti-HER2 agents;
-
patients with hormone receptor positive who have finished their neoadjuvant treatment and can continue with endocrine therapy;
-
patients with breast cancer confirmed by biopsy can take into consideration neoadjuvant therapy (especially endocrine therapy) for hormone receptor positive breast cancer;
-
patients with indication of radiotherapy can receive it preoperatively.
Cases that should be postponed:
-
patients with benign lesions;
-
premalignant lesions;
-
prophylactic surgery;
-
sentinel lymph nodes after breast cancer surgery;
-
cTisNo;
-
reexcision for clear margins;
-
breast reconstruction;
-
tumors that respond well to neoadjuvant treatment;
-
locally advanced or inflammatory breast cancer.
Radiotherapy (RT) is an important treatment for breast cancer, but sometimes there is a minimal locoregional benefit for some patients and no survival benefit for others. Radiotherapists should take into consideration to minimize infectious risk without compromising oncologic outcomes. During COVID-19 crisis, radiotherapy should be reconsidered where there is a limited benefit. Radiotherapist should take into consideration omitting RT whenever appropriate, delaying or abbreviating RT whenever appropriate.
1. Omit RT for patients 65 years old and over (or younger, with relevant comorbidities, with invasive breast cancer that are up to 30 mm with clear margins, G 1-2, oestrogen receptor [ER] positive, human epidermal growth factor receptor 2 [HER2] negative and node negative, who are planned for treatment with endocrine therapy)(14).
2. Deliver RT in 5 fractions for all patients requiring RT with node negative tumours that do not require a boost. Options include 28-30 Gy in once weekly fractions over 5 weeks or 26 Gy in 5 daily fractions over one week as in the FAST and FAST Forward trials, respectively(15-17).
3. Boost RT should be omitted to reduce fractions and/or complexity in the vast majority of patients unless they are 40 years old and under. An example of a significant risk factor is the presence of involved resection margins where further surgery is not possible. Any boost should be either simultaneous and integrated to minimize fractions if resource permits or hypofractionated sequential – e.g., 12 Gy in 4 fraction over 4 days(18).
4. Nodal RT can be omitted in postmenopausal women requiring whole breast RT following sentinel lymph node biopsy and primary surgery for T1, ER positive, HER2-negative G1-2 tumours with 1e2 macrometastases(19).
5. Moderate hypofractionation should be used for all breast/chest wall and nodal RT – e.g., 40 Gy in 15 fractions over 3 weeks. The use of moderate hypofractionation is already the standard of care in many countries and in the altered risk benefit context of a pandemic should be strongly considered in patients with breast reconstruction(20-23).
There were reported some strategies recommended for patients who are diagnosed with COVID-19 whilst on radiotherapy: convert remaining dose to hypofractioned regimens, minimize radiotherapy treatment break during infection treatment and recommence RT only after respiratory symptoms are resumed, with a multidisciplinary input.
The systemic treatment for breast cancer can be divided by stage of disease, early and advanced/metastatic, neoadjuvant, adjuvant and systemic treatment in metastatic setting and by type of treatment related to imunohistochemistry breast cancer subtype. Endocrine therapy for hormone receptor positive, HER2-negative or positive breast cancer, anti-HER2 therapy for HER2-positive breast cancer, chemotherapy for triple negative disease or advanced disease or neoadjuvant treatment or metastatic endocrine resistant.
By adopting these recommendations, where RT is minimized and targeted to those with the highest risk of relevant breast recurrence, we aim to protect our patients and health care professionals from potential exposure to SARS-CoV-19, as well as reducing the workload for health care providers and/or infrastructure at the moments that resources face strain due to the pandemic(24).
Endocrine therapy (ET) for hormone receptor positive breast cancer
Numerous endocrine agents are available for the treatment of hormone receptor positive breast cancer: estrogens, androgens, progestins, antiestrogens (selective estrogen receptor modulators [SERMs] and selective estrogen receptor downregulators [SERDs]), aromatase inhibitors, gonadotropin-releasing hormone (GnRH) analogs, antiprogestins and antiandrogens.
Endocrine therapies frequently used are: SERMs – tamoxifen; aromatase inhibitors – anastrozole, letrozole, exemestane; SERDs – fulvestrant; GnRH analogues – gosereline. There are three main ways in which hormone therapy is used to treat hormone-sensitive breast cancer: adjuvant therapy for early-stage breast cancer(25), treatment of advanced or metastatic breast cancer(26), neoadjuvant treatment of breast cancer(27).
Adjuvant ET options in postmenopausal women include tamoxifen and aromatase inhibitors (AI). Aromatase inhibitors result in better disease-free survival (DFS), but no overall survival (OS) clinical meaningful with a different safety profile. Premenopausal patients may be treated with tamoxifen alone or an association of ovarian suppression function (OSF) with tamoxifen or an aromatase inhibitor. The association of OSF and AI should be considered in higher-risk cases.
ET doesn’t affect patient immunity and can be safely administered, when indicated, in amidst COVID-19 pandemic.
CPBCC supports that LHRH agonists may be given with long acting, every 3 month dosing, to reduce patient visits or, alternatively, home administration of LHRH agonists by patient or visiting nursing may be considered in adjuvant or metastatic setting (e.g., tamoxifen, aromatase inhibitors) and can be safely continued. Fulvestrant should have no effect on immune function, but requires monthly clinical administration.
Neoadjuvant endocrine therapy according to CPBCC (B priority), based on randomized trials, and preoperative treatment with an aromatase inhibitor may offer clinical benefit over tamoxifen in postmenopausal women(27). For premenopausal women, LHRH agonists should be used, and aromatase inhibitors are preferred over tamoxifen. Home administration of LHRH agonists by patient or visiting nurse may be considered where this is an option.
For symptomatic and asymptomatic infected breast cancer patients who receive treatments for SARS-CoV-2 infection, drug-drug interactions should be taken into consideration(28).
Drugs recommended to be used by the therapeutic protocol in adult patients of the “Prof. Dr. Matei Balş” National Institute of Infectious Diseases contain drugs such as: Kaletra® (lopinavir/ritonavir) for asymptomatic patients, Tamiflu® in association with lopinavir/ritonavir in mild symptomatic patients without pneumonia, the association of Plaquenil® for medium symptomatic cases and the association of methylprednisolon for severe, critical cases. There could be used remdesivir (compassionate use), RoActemra® (tocilizumab) in severe forms. Kaletra® can be replaced with Rezolsta® (DRV+cobicistat), but there are no current results for Rezolstra®.
There are highly likelihood significant or life-threatening interactions between ritonavir and tamoxifen or anastrozole, and it is recommended to use alternative drugs due to increase in QTc interval and the risk of torsades de pointes. When we use exemestane with lopinavir, there is a recommendation for monitorization, due to increasing levels of exemestane and to an increased risk of fatigue, hypertension and nausea. Letrozol levels are also increased by lopinavir and there is an increased risk of back pain and bone pain. There are no interactions between fulvestrant and Kaletra®.
Tamiflu® does not interact with any of the endocrine treatment used in breast cancer.
The levels of active metabolite of tamoxifen can be reduced when taken concomitantly with hydroxycloroquine and there is a trend of increase in transaminase levels. There is no interaction between any aromatase inhibitor or fulvestrant and Plaquenil®.
Methylprednisolone could slightly increase the levels of tamoxifen’s active metabolite and there is a minor risk of long QT syndrome. The levels of exemestane and letrozole could be slightly decreased by methylprednisolone. The association of methylprednisolone and anastrozole can lead to the increase of immunosuppression and fluid-electrolyte disturbances, due to increased levels of methylprednisolone. There are no drug-drug interactions between fulvestrant and methylprednisolone.
Tocilizumab can decrease the levels of tamoxifen’s active metabolite and increase the levels of exemestane and letrozole. No interactions between fulvestrant, anastrozole and tocilizumab have been reported(28).
We should be cautious when recommending to our patients to continue endocrine therapy if they are on treatment for SARS-CoV-2 infection, but we must keep in mind also the results reported by Group 1-98 Trial on the impact of DFS on endocrine treatment adherence.
Anti-HER2 treatment for HER2-positive breast cancer
About 20% of breast cancer are HER2-positive and they are characterised by aggressive behaviour and poor prognosis. Commonly used anti-HER2 drugs are: trastuzumab, pertuzumab, TDM-1, neratinib, lapatinib. By 1998, the results of phase 3 clinical trials showed that patients with HER2-positive breast cancer treated with trastuzumab and chemotherapy survived better than patients treated with chemotherapy alone. In 2012, FDA approved pertuzumab as a treatment for women with HER2-positive metastatic breast cancer to be used in combination with trastuzumab and docetaxel.
Trastuzumab and pertuzumab are unlikely to affect the immune function and should be safe for patients, therefore CPBCC recommends to continue those treatments.
Centers for Disease Control and Prevention (CDC) recommend reducing the course of adjuvant trastuzumab treatment from 12 months to 6 months, to give pertuzumab plus trastuzumab for neoadjuvant therapy, adjuvant therapy, locally recurrent or metastatic disease without chemotherapy in order to reduce the risk of neutropenia. In normal times, cardiac evaluation is recommended to be done at 3-month interval during the active treatment with anti-HER2 therapies. The delay of cardiac evaluation in patients without cardiac symptoms and with advanced disease status imagistic evaluation is recommended in COVID-19 pandemic.
CPBCC 2020 states that anti-HER2 antibody treatment in metastatic setting may reasonably be liberalized to longer intervals (e.g., 4 weeks).
Lower priority: antibody treatment (i.e., trastuzumab, pertuzumab) for metastatic, HER2-positive breast cancer beyond two years of maintenance in patients with minimal disease burden (followed-up for progression every 3-6 months).
In stage 1 HER2-positive breast cancers, clinicians may substitute trastuzumab-DM1 instead of paclitaxel/trastuzumab for patient safety or convenience based on randomized trial data, as recommended by CPBCC 2020.
When checking the drug-drug interaction sites and ONCOassist, we found that lapatinib should never be used concomitantly with ritonavir or tocilizumab due to increase in QTc interval, neutropenia and elevated LFT risk. No interactions were found between Tamiflu® and lapatinib. The patients should inform about the treatment with trastuzumab if they have to receive hydroxycloroquine or tocilizumab, due to the risk of cardiac-related toxicity. If a patient is on TDM1 and receives Kaletra®, he has a higher risk of increase in LFT(28).
Chemotherapy
Chemotherapy is the foundation stone of triple negative breast cancer (TNBC) treatment. There are a lot of chemotherapy regimens used in neoadjuvant, adjuvant and metastatic settings, not only in TNBC. Chemotherapy can cause neutropenia that lead cancer patients to risk of infection with SARS-CoV-2 and to risk of developing severe forms of infection, with high mortality rate.
As Chemotherapy Clinical Reference Group states, we should give prophylactic daily granulocyte-colony stimulating factor (G-CSF) or a biosimilar PEGylated G-CSF to prevent neutropenia and reduce admissions.
After an assessment of the risks and benefits for the patient, consider stopping:
-
later-line palliative treatment to reduce the need for admission;
-
adjuvant therapy for low-risk patients (for example, those with breast, lung or colorectal cancer) to reduce the need for immunosuppressive therapy.
Neoadjuvant chemotherapy is the first step in the multimodal treatment for locally advanced breast cancer.
Priority A according to CPBCC 2020 are represented by initiation and continuation of neoadjuvant/adjuvant chemotherapy for triple negative and HER2-positive breast cancer; in metastatic setting use, in early line chemotherapy, is likely to improve outcomes and is recommended to be initiated/continuated.
The COVID-19 Pandemic Breast Cancer Consortium includes as priority B modifications of chemotherapy schedules so as to reduce clinical visits (for instance, using 2- or 3-week dosing instead of weekly dosing for selected agents when appropriate). Patients should receive G-CSF growth factor support to minimize neutropenia, while dexamethasone use should be limited as appropriate to reduce immunosuppression – as priority A.
Lower priority according to CPBCC is later-line palliative chemotherapy that is less likely to improve outcomes.
The CDC recommendations are to switch to oral capecitabine from intravenous taxanes with anti-HER2 therapies for metastatic disease to reduce the risk of neutropenia and substitute albumin-bound paclitaxel (Abraxane®) for paclitaxel or docetaxel to reduce toxicity and potential for admission.
Patients with SARS-CoV-2 infection should inform infection disease specialist about chemotherapy and administration of GM-CSF. Medical oncologist has to teach patients to inform all other specialists about their treatment.
Oral targeting agents CDK4/6 inhibitors, mTOR inhibitors and PIK3Ca inhibitors
There is a new focus on adding targeted therapies to hormone therapy for advanced or metastatic HR-positive breast cancer for prolonging the time until chemotherapy is needed and, ideally, extend survival. Approved drugs include:
-
Palbociclib, ribociclib, abemaciclib (CDK4/6 inhibitors) and everolimus (mTOR inhibitor) – they have been approved by FDA recently for use with hormonal therapy for the treatment of advanced or metastatic breast cancer. They can be given with hormonal therapy after treatment with hormonal therapy to women with advanced or metastatic ER-positive, HER2-negative breast cancer. These combinations are also being tested to see if they can prevent a relapse after the treatment of early-stage ER-positive disease.
-
Alpelisib (PIK3Ca inhibitor) is approved to be used in combination with hormonal therapy to treat HR-positive and HER2-negative breast cancers that have a mutation in the PIK3CA gene.
The use of oral targeted agents must be weighed against the increased risk of adverse events which may increase interaction with healthcare centers and staff. Doses may be reduced to optimize tolerability and minimize treatment-related toxicities. There is a recommendation for starting the first line of endocrine therapy for metastatic disease with HR-positive and HER2-negative status without CDK4/6 inhibitors.
Due to neutropenia induced by CDK4/6 inhibitors, consider stopping CDK4/6 inhibitor if the patient has the infection with SARS-CoV-2 and needs treatment.
The addition of mTOR or PI3KCA inhibitors is not of immediate priority and should be avoided, as ESMO recommends, due to immune suppression (everolimus) or risk of diabetes (alpelisib); the risk for pulmonary side effects could be a reason to postpone these agents to later lines(12).
We should take into consideration all guidelines recommendations when we treat a breast cancer patient in the COVID-19 pandemic, and we should offer the best treatment available for our patients.
Conflict of interests: The authors declare no conflict of interests.
Bibliografie
-
GLOBOCAN 2018.
-
Cancer country profile 2020.
-
Complications of Therapy, Abeloff’s Clinical Oncology 6th Edition.
-
Zhang L, Zhu F, Xie L, Wang C, et al. Clinical characteristics of COVID-19-infected cancer pa-tients: A retrospective case study in three hospitals within Wuhan, China. Ann Oncol. 2020 Mar 26; pii: S0923-7534(20)36383-3.
-
https://www.worldometers.info/coronavirus/
-
Stenger M. Outocomes of COVID-19 Infectionin Patiens With Cancer in Whuan, China. The ASCO Post 04/17/2020.
-
http://www.ms.ro/masuri-recomandate-in-vederea-reducerii-impactului-pandemiei-covid-19/
-
COVID-19 Breast Cancer Patient Triage Guidelines (CPBCC, 2020). Medscape. Apr 08, 2020.
-
Ontario Health, Cancer Care Ontario. Pandemic Planning Clinical Guideline for Patients with Cancer. Available at: https://www.accc-cancer.org/docs/document/cancer-program-fundamentals/oh-cco- pandemic-planning-clinical-guidelines (2nd April 2020, date last ac-cessed).
-
European Society for Medical Oncology (ESMO). The ESMO-MCBS Score Card. Available at: https://www.esmo.org/guidelines/esmo-mcbs/esmo-magnitude-of-clinical-benefit-scale (date last accessed: 2nd of April 2020).
-
World Health Organization. COVID-19: Operational guidance for maintaining essential health services during an outbreak. Available at: https://www.who.int/publications-detail/covid-19-operational-guidance-for-maintaining-essential-health-services-during-an-outbreak (date last accessed: 1st of April 2020).
-
ESMO clinical practice guidelines: Breast cancer. Available at: https://www.esmo.org/guidelines/breast-cancer (date last accessed: 1st April 2020).
-
American College of Surgeons. COVID-19 Guidelines for Triage of Breast Cancer Patients. Available at: https://www.facs.org/covid-19/clinical-guidance/elective-case/breast-cancer (date last accessed: 1st of April 2020).
-
Kunkler IH, Williams LJ, Jack WJ, Cameron DA, Dixon JM. PRIME II investigators. Breast-conserving surgery with or without irradiationin women aged 65 years or older with early breast cancer (PRIME II): a randomised controlled trial. Lancet Oncol. 2015 Mar; 16(3):266e273.
-
Brunt AM, Haviland J, Sydenham M, Algurafi H, Alhasso A, Bliss P, et al. FAST Phase III RCT of Radiotherapy Hypofractionation for Treatment of Early Breast Cancer: 10-Year Results (CRUKE/04/015). IJROBP. 2018; 102(5):1603e1604.
-
Brunt AM, Wheatley D, Yarnold J, Somaiah N, Kelly S, Harnett A, et al. FAST-Forward Trial Management Group. Acute skin toxicity associated with a 1-week schedule of whole breast radiotherapy compared with a standard 3-week regimen delivered in the UK FAST-Forward Trial. Radiother Oncol. 2016; 120:114e118.
-
Brunt AM, Haviland JS, Sydenham MA, Alhasso A, Bloomfield D, Chan C, et al. OC-0595: FAST-Forward phase 3 RCT of 1-week hypofractionated breast radiotherapy: 3-year normal tissue effects. Radiotherapy and Ocology. 2018 Apr; 127. S311-S312. 10.1016/S0167-8140(18)30905-8.
-
Bloomfield DJ. Core Group facilitated by The Royal College of Radiologists. Development of postoperative radiotherapy for breast cancer: UK consensus statements e a model of patient, clinical and commissioner engagement? Clin Oncol R Coll Radiol. 2017 Oct; 29(10):639e641.
-
Haviland JS, Owen JR, Dewar JA, Agrawal RK, Barrett J, Barrett-Lee PJ, et al. START Trial-ists’ Group. The UK standardisation of breast radiotherapy (START) trials of radiotherapy hypofractionationfor treatment of early breast cancer: 10-year follow-up results of two ran-domised controlled trials. Lancet Oncol. 2013 Oct; 14(11):1086e1094.
-
https://www.nice.org.uk/guidance/ng101.
-
Whelan TJ, Pignol JP, Levine MN, Julian JA, MacKenzie R, Parpia S, et al. Long-term results of hypofractionated radiation therapy for breast cancer. N Engl J Med. 2010 Feb 11; 362(6): 513e520.
-
Leong N, Truong PT, Tankel K, Kwan W, Weir L, Olivotto IA. Hypofractionated nodal radiation therapy was not associated with increased patient-reported arm or brachial plexopath symptoms. Int J Radiat Oncol Biol Phys. 2017 Dec 1; 99(5):1166e1172.
-
Clinical Oncology 32 (2020) International Guidelines on Radiation Therapy for Breast Cancer During the COVID-19 Pandemic) 279e281 National comprehensive cancer network. Corona-virus Disease 2019 (COVID-19) Resources for the Cancer Care Community. Available at: https://www.nccn.org/covid-19/ (date last accessed: 1st of April 2020).
-
Cuzick J, Sestak I, Baum M, et al. Effect of anastrozole and tamoxifen as adjuvant treatment for early-stage breast cancer: 10-year analysis of the ATAC trial. Lancet Oncology. 2010; 11(12):1135–1141.
-
Mouridsen H, Gershanovich M, Sun Y, et al. Phase III study of letrozole versus tamoxifen as first-line therapy of advanced breast cancer in postmenopausal women: analysis of survival and update of efficacy from the International Letrozole Breast Cancer Group. Journal of Clinical Oncology. 2003; 21(11):2101–2109.
-
Chia YH, Ellis MJ, Ma CX. Neoadjuvant endocrine therapy in primary breast cancer: indications and use as a research tool. British Journal of Cancer. 2010; 103(6):759–764.
-
ONCOASSIST Decision Support System
-
Chirgwin JH, Giobbie-Hurder A, Coates AS, et al. Treatment Adherence and Its Impact on Disease-Free Survival in the Breast International Group 1-98 Trial of Tamoxifen and Letrozole, Alone and in Sequence. Journal of Clinical Oncology. 2016; 34:21, 2452-2459.




