Oral cavity and laryngeal cancer in patients with metabolic syndrome or type 2 diabetes
Cancerul cavităţii orale şi al laringelui la pacienţii cu sindrom metabolic sau diabet de tip 2
Abstract
Laryngeal cancer is the most common neck and head malignancy. Diabetic patients have an increased risk for developing cancer, including laryngeal and oral malignant lesions. Many factors have been involved in the relation between diabetes and carcinogenesis, such as hyperinsulinemia, hyperglycemia, increased oxidative stress, alterations in lipids levels and metabolism, and inflammation. The treatment options of oral cavity and laryngeal cancer include surgery, radiation therapy, chemotherapy, targeted therapy and immunotherapy. Metformin therapy is the most commonly used treatment in case of type 2 diabetes mellitus (T2DM). The impact of metformin use in diabetic patients with head and neck cancer is not currently fully understood. Several studies suggest that patients undergoing treatment with metformin had a lower incidence of head and neck squamous cell carcinoma and decreased rates of recurrence and metastasis. In contrast, other studies have not shown benefits of metformin therapy on the outcomes of head and neck cancer in patients with T2DM. Numerous other studies are needed to confirm the metformin benefit in oral cavity and laryngeal cancer.Keywords
oral cavity and laryngeal cancerdiabetesmetforminRezumat
Cancerul de laringe este cea mai frecventă formă de cancer în zona capului şi gâtului. Pacienţii diabetici prezintă un risc crescut de a dezvolta cancere, inclusiv cancer laringian sau leziuni maligne la nivelul cavităţii orale. Mulţi factori au fost implicaţi în relaţia dintre diabet şi carcinogeneză, cum ar fi hiperinsulinemia, hiperglicemia, stresul oxidativ crescut, alterarea metabolismului şi a nivelului lipidic şi inflamaţia. Opţiunile terapeutice ale cancerului de cavitate bucală şi laringian includ chirurgie, radioterapie, chimioterapie, terapii ţintite şi imunoterapie. Terapia cu metformină este cea mai frecventă formă de tratament al pacienţilor cu diabet zaharat de tip 2 (DZT2). Impactul utilizării metforminei la pacienţii diabetici cu cancer la cap şi gât nu este pe deplin înţeles în prezent. Unele studii sugerează că pacienţii care urmează tratament cu metformină au avut o incidenţă mai mică a carcinomului scuamos în zona capului şi gâtului şi o rată mai scăzută de recurenţe şi metastaze. În schimb, alte studii nu au arătat beneficii ale terapiei cu metformină la pacienţii cu DZT2 şi cancer în zona capului şi gâtului.Cuvinte Cheie
cancer de cavitate bucală şi laringediabetmetforminăIntroduction
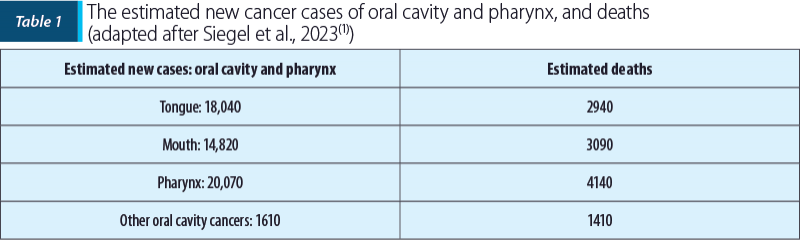
Laryngeal cancer is the most common neck and head malignancy. In 2023, Siegel et al. published in CA: A Cancer Journal for Clinicians the estimated new cancer cases and deaths in the United States of America, including malignant pathology of interest of oral cavity and pharynx(1). The estimated new cancer cases and deaths are presented in Table 1.
The incidence of laryngeal cancer in patients with metabolic syndrome or type 2 diabetes mellitus (T2DM) has been evaluated in numerous studies, because the two pathologies are associated with numerous complications or comorbidities, including several specific forms of cancer. A prospective study in which there were enrolled 1,058,831 subjects in 1982 (without cancer at baseline) was followed-up for mortality 26 years later. Regarding the risk of mortality among women, diabetes was associated with higher risk of death from cancers of the liver, pancreas, endometrium, colon and breast, while among men, diabetes was associated with a higher risk of death from cancers of the breast, liver, oral cavity and pharynx, pancreas, bladder and colon(2).
This article is a brief review of association of oral cavity and laryngeal cancer with metabolic syndrome or T2DM, based on articles published on Google Academic and PubMed.
The risk of oral cavity and pharyngeal cancer among patients with metabolic syndrome and T2DM
The many possible risk factors involved in the occurrence of oral cavity and pharyngeal cancer are: smoking, alcohol, radiation, immune dysfunction with impaired immune response and proinflammatory state(3). Patients with T2DM have an increased risk for developing cancer, including oral malignant lesion. The pathogenesis of oral cancer associated with diabetes remains to be elucidated. Many factors have been involved in the relation of diabetes and carcinogenesis, as well as hyperinsulinemia, hyperglycemia, increased oxidative stress, alterations in lipids levels and metabolism, or inflammation.
Hyperinsulinemia can generate the initiation and progression of the process of malignancy directly or indirectly through the involvement of insulin-like growth factor 1 (IGF-1), showing mitogenic and antiapoptotic properties(4).
Hyperglycemia can promote deoxyribonucleic acid (DNA) damage by increased advanced glycation end products (AGEs) and alteration of polyol pathway(5-7).
Increased oxidative stress characteristic of diabetes can generate DNA damage which is involved in mutational changes in oncogenes(5).
Dyslipedemia. Several studies have demonstrated that the alterations in lipids levels and metabolism are related to carcinogenesis and cancer metastasis(8,9). In an article published in 2011 in the Open Cardiovascular Medicine Journal entitled “Malignancy-associated dyslipidemia”, Bielecka-Dąbrowa et al. mentioned that the lipid peroxidation product, malondialdehyde, may cross-link DNA(10).
Inflammation is an associated condition with metabolic syndrome and T2DM. The chronic inflammatory status augments the expression of receptor for AGEs (RAGE). Several studies have reported the involvement of RAGE activation in cancer promotion(11,12). Shun-Yao et al. appreciates that the accumulation of AGEs due to diabetes promotes the progression of oral cancer and decreases the survival rate(13).
Epidemiology of oral cancer
and pharyngeal cancer in patients
with metabolic syndrome and T2DM
Gong et al. performed a meta-analysis to explore the association between the risk of precancerous lesions and oral cancer in patients with T2DM. The analysis of 13 studies revealed that, compared with subjects without diabetes mellitus, the patients with T2DM had a significantly increased incidence of oral cancer and mortality; the same meta-analysis revealed an elevated risk of precancerous lesions development(14).
Tseng investigated the trends of oral cancer in the Taiwanese population and the association with diabetes. The study revealed a possible increased risk of oral cancer in diabetic patients. In an unadjusted model, the diabetes status and the duration of the condition were significantly associated with oral cancer. In contrast, after multivariable adjustment, the association was insignificant(15). In a retrospective study using Taiwan’s Longitudinal Health Insurance Database, Tseng et al. assessed the risk of head and neck cancer in 89,089 patients newly diagnosed with diabetes compared to subjects without diabetes. The results of the study showed that diabetes is associated with an increased risk of developing head and neck cancer, respectively of the cavity, oropharyngeal cancer and nasopharyngeal carcinoma(16).
A retrospective study conducted in Hungary by Végh et al. evaluated the presence of diabetes and impaired fasting glucose in 758 patients with confirmed oral cancer. The results were compared to collected medical information for 14 years. Fourteen years earlier, diabetes was diagnosed in 14.5% and impaired fasting glucose in 9.7% of patients with oral cancer. In the last 14 years, the prevalence of T2DM was found to be 25.9%, and the prevalence of impaired fasting glucose was 20.6%. The authors concluded that the prevalence of T2DM and impaired fasting glucose increased significantly among oral cancer in 14 years(17).
Remschmidt et al. published in 2022 in Anticancer Research a retrospective study on the link between impaired fasting glucose, diabetes and oral cancer in Austria. The results of the study showed that the alteration of carbohydrate metabolism was significantly increased among cancer patients compared with the control group(18).
Bosetti conducted a study on the association of diabetes with risk of cancers. The studies included 1468 patients with oral and pharyngeal cancer cases. The multivariate odds ratios for patients with diabetes as compared with subjects without this condition highlighted that odds ratios were significantly elevated for oral and pharyngeal cancer. The results of the study revealed that, for the cancer of the oral cavity, “the excess risk persisted over 10 years since the diagnosis of diabetes”(19).
In 2019, Kim et al. performed a retrospective study on the relationship between metabolic syndrome and laryngeal cancer. Patients with laryngeal cancer between 2009 and 2010 were retrospectively identified and tracked until 2015. During the follow-up period, 5322 patients were newly diagnosed with laryngeal cancer. The authors concluded that, in Korea, the metabolic syndrome “is significantly associated with the development of laryngeal cancer”(20). Similar results were offered by Kim et al., who investigated the association of metabolic syndrome and laryngeal cancer in the Korean population. A total of 6,757,048 subjects who received national health were analyzed and followed-up in 2018. The results of the study revealed that patients with metabolic syndrome have a significantly higher risk of laryngeal cancer compared with the subjects without this condition(21).
A meta-analysis that explores the relationship between T2DM and laryngeal cancer was conducted by Yan. Fourteen case-control studies and 13 cohort studies were included in this analysis. The conclusions of the meta-analysis are that T2DM is associated with an increased risk of head and neck cancer, and other factors involved are smoking, alcohol abuse and obesity(22).
In 2016, Gupta, Johnson and Kumar have published in Oncology data on global epidemiology of head and neck cancer. The authors used data from the GLOBOCAN 2012, Cancer Incidence in Five Continents, and World Health Organization Mortality Database and Surveillance. The results of the analysis revealed that “the estimated rates for cancers of the tonsils and pharynx among males (7.5/2.5 per 100,000 per annum) and females (2.7/0.5 per 100,000 per annum) are reported to be the highest in Western Europe, whereas these rates for cancer of the larynx among males (7.9/4.0 per 100,000 per annum) and females (0.9/0.5 per 100,000 per annum) are reported to be the highest in the Caribbean”(23).
Treatment of head and neck cancer: general recommendations and particularities
of therapy in diabetic patients
The National Comprehensive Cancer Network guidelines for head and neck cancer provides information on the lip, oral cavity, pharynx, larynx and paranasal sinuses cancers. In the 9th edition of the American Joint Committee of Cancer, it is appreciated that cervical cancer is a worldwide pandemic. “In 2018, there were an estimated 570,000 new cases and 311,000 deaths worldwide, making it the fourth most frequently diagnosed cancer and the fourth leading cause of cancer death in women”(24).
The treatment options include surgery, radiation therapy, chemotherapy, targeted therapy and immunotherapy. The impact of diabetes on the treatment of patients with head and neck cancer has been evaluated in several studies. The impact of metformin use in diabetic patients with head and neck cancer is unclear. Spratt et al. performed a multicenter study named “The influence of diabetes mellitus and metformin on distant metastases in oropharyngeal cancer”. The study included 1745 patients with oropharyngeal cancer, of which 184 patients had diabetes at the time of diagnosis, and of these, 102 were on metformin therapy, the most commonly used treatment in the case of T2DM. The study revealed that “diabetic patients not using metformin independently have significantly higher rates of distant metastases than non-diabetic patients, whereas metformin users have similar rates of distant metastases to non-diabetics”(25). A systematic review published in 2015 by Rego et al. analyzed information provided in the literature on the potential effect of metformin on head and neck squamous cell carcinoma. Reviews and meta-analyses revealed that patients undergoing treatment with metformin had a lower incidence of head and neck squamous cell carcinoma and “that individuals taking metformin had decreased rates of locoregional recurrence and metastasis and improved overall survival and disease-free survival rates”(26). Stokes et al. evaluated the survival impact of metformin in head and neck cancer. The study included 1646 patients diagnosed with head and neck cancer from 2008 to 2011. Of these, 378 patients were diagnosed with diabetes and 124 were taking metformin. The results of the study revealed that metformin improved cancer-specific survival(27).
In contrast, a retrospective study in which there were included 1679 patients diagnosed with squamous carcinoma of larynx, hypopharynx and nasopharynx, conducted between 2007 and 2012, in Canada, did not highlight a survival advantage in patients on treatment with metformin at the time of diagnosis(28). A retrospective review published in 2019 by Lee et al. evaluated the effect of treatment with metformin on the outcomes of head and neck cancer in patients with T2DM. After analyzing the data, the authors concluded that “no association between metformin use and oncologic outcomes were observed”(29).
Conclusions
The diabetic patients have an increased risk for developing cancer, including laryngeal and oral malignant lesion. The treatment options of oral cavity and laryngeal cancer include surgery, radiation therapy, chemotherapy, targeted therapy and immunotherapy. Metformin therapy is the most commonly used treatment in T2DM. The impact of metformin use in diabetic patients with head and neck cancer is unclear. Numerous other studies are needed to confirm the metformin benefit in oral cavity and laryngeal cancer.
Conflict of interest: none declared
Financial support: none declared
This work is permanently accessible online free of charge and published under the CC-BY.
Bibliografie
- Siegel RL, Miller KD, Wagle NS, Jemal A. Cancer statistics, 2023. CA Cancer J Clin. 2023;73(1):17-48.
- Campbell PT, Newton CC, Patel AV, Jacobs EJ, Gapstur SM. Diabetes and Cause-Specific Mortality in a Prospective Cohort of One Million U.S. Adults. Diabetes Care. 2012;35(9):1835–1844.
- Verhulst MJL, Loos BG, Gerdes VEA, Teeuw WJ. Evaluating All Potential Oral Complications of Diabetes Mellitus. Front Endocrinol (Lausanne). 2019;10:56.
- Abudawood M. Diabetes and cancer: A comprehensive review. J Res Med Sci. 2019;24:94.
- Lee SC, Chan JC. Evidence for DNA damage as a biological link between diabetes and cancer. Chin Med J (Engl). 2015;128(11):1543–1548.
- Stopper H, Schinzel R, Sebekova K, Heidland A. Genotoxicity of advanced glycation end products in mammalian cells. Cancer Lett. 2003;190(2):151-156.
- McCarthy MI. Genomics, type 2 diabetes, and obesity. N Engl J Med. 2010;363(24):2339-2350.
- Alexopoulos CG, Blatsios B, Avgerinos A. Serum lipids and lipoprotein disorders in cancer patients. Cancer. 1987;60(12):3065–3070.
- Simo CE, Orti LA, Sena FF, Contreras BE. Blood cholesterol in patients with cancer. Ann Med Intern. 1998;15:363–366.
- Bielecka-Dąbrowa A, Hannam S, Rysz J, Banach M. Malignancy-associated dyslipidemia. Open Cardiovasc Med J. 2011;5:35-40.
- Riichiro A, Sho-ichi Y. AGE-RAGE system and carcinogenesis. Curr Pharm Des. 2008;14(10):940–945.
- Christoffer G, Astrid R, Moritz D, Julia N, Gerhard F, Karin M-D, Alexander E, Bernd A, Bierhaus Angelika NPP. RAGE signaling sustains inflammation and promotes tumor development. J Exp Med. 2008;205(2):275–285.
- Shun-Yao K, Hshin-An K, Tzong-Ming S, Tzong-Cherng C, Chen H-I, Yi-Ting C, Yu Y-H, Yang S-H, Shu-Shing C. Advanced glycation end products influence oral cancer cell survival via Bcl-xl and Nrf-2 regulation in vitro. Oncol Lett. 2017;13(5):3328–3334.
- Gong Y, Wei B, Yu L, Pan W. Type 2 diabetes mellitus and risk of oral cancer and precancerous lesions: a meta-analysis of observational studies. Oral Oncol. 2015;51(4):332–340.
- Tseng CH. Oral cancer in Taiwan: is diabetes a risk factor?. Clin Oral Invest. 2013;17(5):1357–1364.
- Tseng KS, Lin C, Lin YS, Weng SF. Risk of head and neck cancer in patients with diabetes mellitus: a retrospective cohort study in Taiwan. JAMA Otolaryngol Head Neck Surg. 2014;140(8):746–753.
- Végh D, Bányai D, Hermann P, Németh Z, Ujpal M. Type-2 diabetes mellitus and oral tumors in Hungary: a long-term comparative epidemiological study. Anticancer Res. 2017;37(4):1853-1857.
- Remschmidt B, Pau M, Gaessler J, Zemann W, Jakse N, Payer M, Végh D. Diabetes mellitus and oral cancer: A retrospective study from Austria. Anticancer Res. 2022;42(4):1899–1903.
- Bosetti C, Rosato V, Polesel J, Levi F, Talamini R, Montella M, Negri E, Tavani A, Zucchetto A, Francceschi S, Carrao G, La Vecchia C. Diabetes mellitus and cancer risk in a network of case-control studies. Nutr Cancer. 2012;64(5):643–651.
- Kim SY, Han KD, Joo YH. Metabolic Syndrome and Incidence of Laryngeal Cancer: A Nationwide Cohort Study. Sci Rep. 2019;9(1):667.
- Kim HB, Kim GJ, Han KD, Joo YH. Changes in metabolic syndrome status and risk of laryngeal cancer: A nationwide cohort study. PLoS One. 2021;7;16(6):e0252872.
- Yan P, Wang Y, Yu X, Liu Y, Zhang ZJ. Type 2 diabetes mellitus and risk of head and neck cancer subtypes: a systematic review and meta-analysis of observational studies. Acta Diabetol. 2021;58(5):549–565.
- Gupta B, Johnson NW, Kumar N. Global epidemiology of head and neck cancers: a continuing challenge. Oncology. 2016;91(1):13–23.
- Olawaiye AB, Baker TP, Washington MK, Mutch DG. The new (Version 9) American Joint Committee on Cancer tumor, node, metastasis staging for cervical cancer. CA Cancer J Clin. 2021;71(4):287–298.
- Spratt DE, Beadle BM, Zumsteg ZS, Rivera A, Skinner HD, Osborne JR, Garden AS, Lee NY. The influence of diabetes mellitus and metformin on distant metastases in oropharyngeal cancer: a multicenter study. Int J Rad Oncol Biol Phys. 2016;94(3):523–531.
- Rego DF, Pavan LM, Elias ST, De Luca Canto G, Guerra EN. Effects of metformin on head and neck cancer: A systematic review. Oral Oncology. 2015;(5):416–422.
- Stokes WA, Eguchi M, Amini A, Hararah MK, Ding D, McDermott JD, Bradley CJ, Karam SD. Survival impact and toxicity of metformin in head and neck cancer: An analysis of the SEER-Medicare dataset. Oral Oncol. 2018;84:12-19.
- Quimby AE, Lebo NL, Griffiths R, Hall S, Dimitroulakos J, Johnson-Obaseki S. Does metformin usage improve survival in head and neck squamous cell carcinoma? A population-based study. J Otolaryngol Head Neck Surg. 2018;47(1):74.
- Lee DJ, McMullen CP, Foreman A, Huang SH, Lu L, Xu W, de Almeida JR, Liu G, Bratman SV, Goldstein DP. Impact of metformin on disease control and survival in patients with head and neck cancer: a retrospective cohort study. J Otolaryngol Head Neck Surg. 2019;48(1):3.





