Biochemical pathology of substance P in neuropsychiatry
Patologia biochimică a substanţei P în neuropsihiatrie
Abstract
Biological, chemical and biochemical processes are always the basis of the initiation of a complex cascade of neuropathological events from which symptoms, syndromes and their clinical manifestations derive. Regarding the neurobiology and neurochemistry of some neuropeptides (in the present exposition, substance P) with implications in psychiatry and neurology, the specialized literature from us has been, somehow, scarce in the last decades. This article points out the conclusions of some previous studies regarding the neuropathology of substance P. Last but not least, the relevance of these studies brings, of course, an important contribution to the design and development of future treatment modalities.Keywords
neuropsychiatryneuropeptideneurobiochemistrysubstance PimpactRezumat
Procesele biologice, chimice şi biochimice sunt întotdeauna baza iniţierii unei cascade complexe de evenimente neuropatologice din care derivă simptomele, sindroamele şi manifestările clinice ale acestora. În ceea ce priveşte neurobiologia şi neurochimia unor neuropeptide (în expunerea de faţă, substanţa P) cu implicaţii în psihiatrie şi neurologie, literatura de specialitate de la noi a fost, cumva, vitregă în ultimele decenii. Acest articol evidenţiază punctat concluziile unor studii anterioare referitoare la neuropatologia substanţei P. Nu în ultimul rând, relevanţa acestor studii are, desigur, o contribuţie importantă asupra proiectării şi elaborării viitoarelor ţinte terapeutice.Cuvinte Cheie
europsihiatrieneuropeptideneurobiochimiesubstanta PimpactThe biochemistry and cell biology of substance P. Neuropeptides
Substance P (SP) is an undecapeptide which appertains to the neurokinin (tachykinins; TKs) family of neuropeptides. SP-containing neurons are extensively distributed in the peripheral and the central nervous system (CNS), where they are found among other neuroanatomical structures, in the mid-brain and the basal ganglia, the hypothalamus and the limbic system, as well as the hippocampus and amygdala(1). SP is interacting with other neurotransmitters by exerting neuromodulatory effects. Co-localizations with serotonin in the raphe nucleus(2), with dopamine in the midbrain and striatum, and with corticotropin-releasing hormone (CRH) in the hypothalamus(3) prove the initiation and development of some neurophysiological processes that are the basis of these effects. SP shows its effects by binding to the neurokinin-1 receptor (NK1R), which can be adequately blocked by specific NK1R antagonists(4,5).
Neuropeptides are generated from a gene that expresses its mRNA and expresses it by RNA splicing. This implies the transcription of an mRNA from a given gene, in the nucleus(6). In this way, that neuropeptide is “prepared” for axonal transport by clustering into secretory vesicles within the Golgi complex. During the transportation process, the precursor protein is processed by specific cleavage enzymes into the active and inactive defragmented peptides.
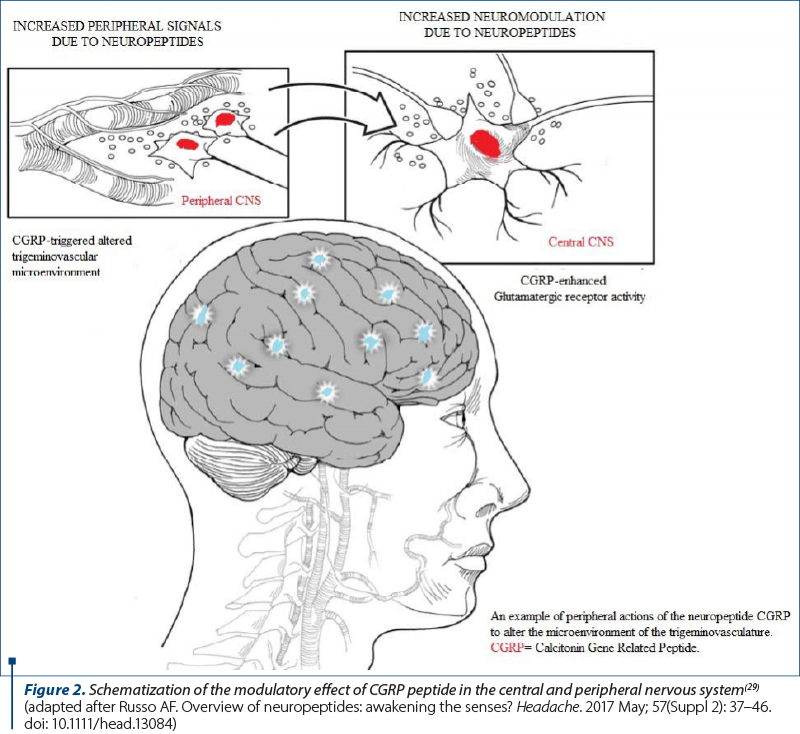
The responsible peptides in controlling pituitary secretion are synthetized in the hypothalamus. Hypothalamic releasing and inhibiting factors are released in the secretory neurons corresponding to the third ventricle, which onwards the projections to the median eminence where they meet and release peptides into the hypothalamohypophysial portal circulatory network(7). The colocalization of neuropeptides inside the classical neurotransmitter circuits denotes an interaction between these systems, and the potentiation of norepinephrine (a monoamine neurotransmitter) by neuropeptide Y is well initiated, wherefore neuropeptide Y is known to reduce stress reaction driven by norepinephrine. There is enough evidence concerning the involvement of neuropeptides in the pathophysiology of neuropsychiatric disorders. Neuropeptides act as neurotransmitters, neuromodulators, or neurohormones. Their control and release are not limited to synapses or axon terminals, but throughout the axon or even from dendrites(8). Almost all neuropeptide receptors are Gprotein coupled, seventransmembrane domain receptors, related to the same family of proteins as monoamine receptors. Each neuropeptide receptor is individually coupled to one type of G protein (Gs, Gi, Gq), depending on the subtype of G protein with which the receptor cooperates, which may have as consequence the stimulation or inhibition of specific second messenger pathways. Signaling pathways demand the participation of activated G protein, modulating the activity of adenylate cyclase or phospholipase C. The delivered peptides are condensed into smaller fragments and into single amino acids by specific enzymes-termed peptidases. Peptidases are bound to presynaptic/postsynaptic neuronal membrane or in solution environment in the cytoplasm and extracellular fluid, distributed globally in peripheral organs, serum and CNS. Besides, neuropeptides generally have halflives on the order of minutes. A less appreciated fact is that, over the last several decades, the number of known neuropeptides in the human brain reached over 100 distinct molecules. A probably underappreciated characteristic of neuropeptides is that, in comparison to classical neurotransmitters, neuropeptides diffuse from their point of release and hence they can react at a relatively large distance(9). Neuropeptides act as signal transducers via cell-surface receptors. Nearly all neuropeptides act at G protein coupled receptors. Neuropeptide modulation of target cells can appear by two distinct but overexposed mechanisms: within the brain→ neuromodulators, and within the periphery→ signaling molecules. Neuropeptides are capable to alter sensory perception in migraine. An eloquent example is CGRP actions in the periphery and the central nervous system (Figure 2). CGRP can deteriorate the molecular microenvironment of the trigeminal vasculature to possibly sensitize trigeminal nociceptors, as well as act in the brain to increase glutamatergic signaling of CNS neurons(10).

Substance P and the neurological pathology
Some neurological diseases are unique to the central and peripheral nervous system. It seems that substance P is involved in the pathology of some of them. Neurodegenerative disorders (NDs) such as Alzheimer’s disease (AD), Parkinson’s disease (PD), multiple sclerosis (MS), Huntington’s disease (HD) or amyotrophic lateral sclerosis (ALS) are diagnosed by clinical criteria, laboratory findings and on functional neuroimaging. The diagnostic accuracy of these diseases depends on the type of the neurodegenerative conflict. In nearly all cases, the diagnosis remains obscure/uncertain until death, and during the evolution of the disease, NDs can be slightly improved by medication but never cured. Therefore, the expansion of serum neurobiomarkers that may evaluate/predict the risk of disease, presence and progression is one of the major aspects and challenges in the research on neurodegenerative disorders, as well as in the choice of specific/alternative therapy. Neurodegenerative disorders are chronic and progressive diseases associated with selective and symmetric loss of neurons in motor, sensory or cognitive systems(11). It has been validated that TKs can exert neurotrophic effects on central neurons and neurodegenerative disorders, most probably due to the absence or depletion of TKs in selected areas of the brain and spinal cord. Substance P was admitted as the first neuroactive peptide and was the first one suggested to act as a neurotransmitter(12).
Multiple sclerosis
Multiple sclerosis is a neurological disorder characterized by focal inflammation, demyelination and gliosis in the grey matter of the CNS. Innate and adaptive immune response are the key in the development of MS, but it is pathologically impacted by T cells. Early studies indicated that substance P can play an essential role in activating autoimmune T cells by increasing proinflammatory cytokine production, which can up-regulate SP and NK1R expression, determining antigen-presenting cells to produce more T cell stimulatory cytokines(13). Substance P can increase the permeability of blood-brain barrier by cross-reacting with NK1R on endothelial cells that generates an inflow of immune mediators and activation of the T cells to the CNS, which can interact with astrocytes and oligodendroglia, advancing to further release of cytokines and substance P.
Huntington’s disease and amyotrophic lateral sclerosis
Huntington’s disease and amyotrophic lateral sclerosis have been studied for alterations in substance P levels. HD is an autosomal dominant neurogenetic disorder caused by the decompensation and degeneration of some areas of the brain, mainly in the ganglia basal region. Moreover, in this disease, there is a decrease in the number of neurotransmitters, like substance P, especially in the CNS and basal ganglia. In other regions of the brain, such as the thalamus and hypothalamus, there is no significant difference in the concentration of SP between patients and healthy groups(14). Several surveys have shown that substance P could increase the release of striatal dopamine by sending a projection to the CNS, and SP containing striatal neurons can have a feedback loop with dopaminergic input(15).
Amyotrophic lateral sclerosis is another neurodegenerative disorde that is associated with changes in the substance P levels in the central nervous system. Anyhow, studies on this disease are limited. There are consecrated studies showing that the level of substance P as a biomarker may be altered in this illness. Cerebrospinal fluid biomarkers such as SP could represent neuronal cell loss at an early stage of the disease. Recent reports highlighted that the concentration of substance P in the cerebrospinal fluid of ALS patients was higher than in control groups(16). Other designed studies indicate that the distribution of NK1R was detected in the spinal cord of ALS patients. Also, they reported a significant reduction in SP binding, especially in the ventral horn, being associated with the degeneration of motor neurons. All these suggested that the localization of receptors of substance P to the ventral horn motor neurons in the spinal cord can attest the role of SP in the function of motor neurons in the disease.
Parkinson’s disease
Parkinson’s disease is a progressive neurodegenerative disorder that is characterized by the degeneration/loss of dopaminergic neurons in the substantia nigra pars compacta(17). Other disorders due to the degeneration of dopaminergic neurons are represented by inflammation, blood-brain barrier dysfunction, oxidative stress, glutamate cytotoxicity and mitochondrial dysfunction. Other research studies have shown that the SP level is decreased in substantia nigra pars compacta and pars reticulata(18). Yet, the researchers have identified the lack of any relation between the reduction of the SP level and the severity of the disease. A consistent number of studies tried to determine the pathology of Parkinson’s disease and found that the primary incriminated factor is the loss of substance P activity in the substantia nigra, probably as a consequence of the lack of dopamine as a secondary response(19).
Substance P and the psychiatric pathology
Neuropeptides exert an impact on some physiological functions, such as social cognition, thermoregulation, sleep, appetite, thirst, sex and others. These are the functions that get affected in psychiatric illnesses such as depression, mood disorders, dementia, autism, schizophrenia and many more. Many previous articles have focused on per se diseases. In this second part of the article, we bring to attention some major illnesses in psychiatry in association with neuropeptides.
Depression
Plenty of neuropeptides have been implicated in depressive disorders, ranging from substance P, corticotropin-releasing hormone (CRH), thyrotropin-releasing hormone (TRH), vasopressin, neuropeptide Y and galanin. The biological effects of substance P are well chosen via the receptors NK1, NK2 and NK3. NK1 and NK3 receptors are extensively distributed in the central nervous system, forasmuch the NK2 receptor is located in the smooth muscle of the gastrointestinal, respiratory and urinary tracts, but it has also been discovered in distinct regions of the rodent CNS – prefrontal cortex and the hippocampus. It also plays a role as a pain neurotransmitter, and the administration to animals denotes behavioral and cardiovascular effects resembling the stress response and related disorders(20). It is co-localized with neoendorphins and serotonin; the serotonergic activity in the hippocampus and lateral septum was improved by NK1 antagonism. It was also observed that both depressed and post-traumatic stress disorder patients had elevated cerebrospinal fluid substance P concentrations(21). Moreover, the central administration of substance P has been found to cause anxiogenic effects in the elevated plus maze, whereas the administration of substance P antagonists attenuated social anxiety by contributing to the antidepressant effect.
Schizophrenia
Schizophrenia is a psychiatric disorder affecting approximately 0.5% to 1% of the population in their lifetime(22). Psychosis normally occurs in the late teenage years or early adulthood, between 18 and 25 years old (23). Even though the cause underlying this mental illness remains to be clarified, several biochemical effectors have been proposed, including dysregulations in oligodendrocytes(24), N-methyl-D-aspartate (NMDA) signaling, or dopaminergic transmission. More than 100 neuropeptides, including substance P, have been characterized and many of these have been involved in schizophrenia. Neuropeptides are intensively identified than classic neurotransmitters to limbic structures incriminated in severe mental illness. Psychosis without schizophrenia, such as affective disorder and unspecified functional psychosis, displayed higher substance P concentrations(25). An immunocytochemical study also did not spot any changes in substance P in the basal ganglia in six schizophrenia patients compared with unaffected controls(26).
Addiction
A few research studies come up by mentioning new findings about certain features of substance P in promoting, initiating and entertaining neurobiochemical reactions to alcohol and other addictive substances. Studies have suggested that neuropeptide systems may inflect excessive alcohol intake, based on evidence that ethanol ingestion and energy balance modulates substance P in specific brain areas, as well as numerous neuropeptide systems, and determining the responsivity of these systems similarly as ethanol intake(27). Substance P in the hypothalamus can act locally or activate in other regions by linking with other systems, such as the mesolimbic dopamine reward system which “supervise” the defensively compensation of drugs consumption.
Anxiety disorders
We have already seen that, among other neuropeptides, substance P can be involved in numerous neurophysiological and pathological processes, including stress and anxiety related behaviors. According to this, brain regions that are thought to be involved in anxiety regulation contains SP and its specific NK1 receptors. Substance P concentration in different brain areas alters with the vulnerability of stressful stimulus by affecting NK1 receptor binding. SP is released in reply to a stressor, generating anxiogenic effects via the activation of hypothalamic-pituitary-adrenal (HPA) axis, by cortisol release. Cortisol release, by the activation of HPA axis, it is nothing but the indirect anxiogenic effect of substance P(28). At the time that SP concentration is high-founded in stressful circumstances, certain studies have revealed that the pharmacological antagonism or genetic deficiency in NK-1 receptors has as a consequence the anxiolytic response, becoming in this way a suitable therapeutic agent for the treatment of stress-related and anxiety-related disorders.
Conclusions
The normal and pathological neurochemical effects of substance P do not occur singularly. They are always promoted and trained by activating the brain areas where they are located, then by the stimuli and their nature that cause them, as well as by other neuropeptides which can sometimes have a neuromodulatory and not necessarily a destructive role. The data from the specialized literature on this topic are not very many, and in our country, extremely few. For this reason, the essential biochemical mechanisms and, together with them, the most important neurological and psychiatric pathologies caused by substance P were exposed in the article, summarizing the impact of these molecules on individual neuropsychiatric illnesses.
Conflict of interest: none declared
Financial support: none declared
This work is permanently accessible online free of charge and published under the CC-BY.

Bibliografie
-
Pioro EP. Distribution of substance P and enkephalin immunoreactive neurons and fibers. In: Paxinos G (ed), The Human Nervous System. San Diego, New York, Academic Press, 1990, pp 1051–1094.
-
Sergeyev V, Hokfelt T, Hurd Y. Serotonin and substance P co-exist in dorsal raphe neurons of the human brain. Neuroreport. 1999;10:3967–3970.
-
Otsuka M, Yoshioka K. Neurotransmitter functions of mammalian tachykinins. Physiol Rev. 1993;73:229–308.
-
Kramer MS, Cutler N, Feighner J, Shrivastava R, Carman J, Sramek JJ, Reines SA, Liu G, Snavely D, Wyatt-Knowles E, Hale JJ, Mills SG, MacCoss M, Swain CJ, Harrison T, Hill RG, Hefti F, Scolnick EM, Cascieri MA, Chicchi GG, Sadowski S, Williams AR, Hewson L, Smith D, Rupniak NM. Distinct mechanism for antidepressant activity by blockade of central substance P receptors. Science. 1998;281:1640–1645.
-
Stout SC, Owens MJ, Nemeroff CB. Neurokinin (1) receptor antagonists as potential antidepressants. Annu Rev Pharmacol Toxicol. 2001;41:877–906.
-
Griebel G, Holsboer F. Neuropeptide receptor ligands as drugs for psychiatric diseases: The end of the beginning? Nat Rev Drug Discov. 2012;11:46278.
-
Sarkhel S. Kaplan and Sadock’s synopsis of psychiatry: Behavioral sciences/clinical psychiatry, 10th edition. Indian J Psychiatry. 2009;51(4):331.
-
Abid MS, Mousavi S, Checco JW. Identifying receptors for neuropeptides and peptide hormones: Challenges and recent progress. ACS Chem Biol. 2021;16:25163.
-
Van den Pol AN. Neuropeptide transmission in brain circuits. Neuron. 2012;76:98–115.
-
Russo AF. Calcitonin gene-related peptide (CGRP): A new target for migraine. Ann Rev Pharmacol Toxicol. 2015;55:533–552.
-
Martin JB. Molecular basis of the neurodegenerative disorders. N Engl J Med. 1999;340:1970-80.
-
V Euler US, Gaddum JH. An unidentified depressor substance in certain tissue extracts. J Physiol. 1931;72(1):74-87.
-
O’Connor TM, O’Connell J, O’Brien DI, Goode T, Bredin CP, Shanahan F. The role of substance P in inflammatory disease. J Cell Physiol. 2004;201:167-80.
-
Emson P, Arregui A, Clement-Jones V, Sandberg B, Rossor M. Regional distribution of methionine-enkephalin and substance P-like immunoreactivity in normal human brain and in Huntington’s disease. Brain Res. 1980;199:147-60.
-
Kubota Y, Inagaki S, Kito S. Innervation of substance P neurons by catecholaminergic terminals in the neostriatum. Brain Res. 1986;375:163-7.
-
Dietl M, Sanchez M, Probst A, Palacios J. Substance P receptors in the human spinal cord: decrease in amyotrophic lateral sclerosis. Brain Res. 1989;483:39-49.
-
Thornton E, Vink R. Substance P and its tachykinin NK1 receptor: a novel neuroprotective target for Parkinson’s disease. Neural Regen Res. 2015;10:1403-5.
-
Tenovuo O, Rinne U, Viljanen M. Substance P immunoreactivity in the postmortem parkinsonian brain. Brain Res. 1984;303:113-6.
-
Barker R. Substance P and Parkinson’s disease: a causal relationship? J Theor Biol. 1986;120:353-62.
-
Carpenter LL, Owens MJ, Baker DG, Ekhator NN, Horn PS, et al. Elevated cerebrospinal fluid substance P concentrations in posttraumatic stress disorder and major depression. Am J Psychiatry. 2006;163:63743.
-
Ebner K, Sartori SB, Singewald N. Tachykinin receptors as therapeutic targets in stressrelated disorders. Curr Pharm Des. 2009;15:164774.
-
Tandon R, Keshavan MS, Nasrallah HA. Schizophrenia, ‘just the facts’ what we know in 2008. 2. Epidemiology and etiology. Schizophr Res. 2008;102:1–18.
-
Insel TR. Rethinking schizophrenia. Nature. 2010;468:187–193.
-
Roussos P, Haroutunian V. Schizophrenia: susceptibility genes and oligodendroglial and myelin related abnormalities. Front Cell Neurosci. 2014;8:5.
-
Kleinman JE, Hong J, Iadarola M, Govoni S, Gillin CJ . Neuropeptides in human brain – postmortem studies. Prog Neuropsychopharmacol Biol Psychiatry. 1985;9:91–95.
-
Zech M, Bogerts B. Methionine-enkephalin and substance P in the basal ganglia of normals, Parkinson patients, Huntington patients, and schizophrenics. A qualitative immunohistochemical study. Acta Neuropathol. 1985;68:32–38.
-
Barson JR, Ho HT, Leibowitz SF. Anterior thalamic paraventricular nucleus is involved in intermittent access ethanol drinking: Role of orexin receptor 2. Addict Biol. 2015;20:46981.
-
Ebner K, Muigg P, Singewald G, Singewald N. The role of substance P in stress and anxiety responses. Amino Acids. 2006;31(3):251-72.
-
Russo AF. Overview of neuropeptides: awakening the senses? Headache. 2017;57(Suppl 2):37–46. doi:10.1111/head.13084.




