Thrombophilia is a pathology introduced into the clinical practice, with risk of venous thromboembolism (VTE) in pregnancy. Objectives. The potential risks for the mother and fetus should be considered when using anticoagulants in pregnancy. Materials and method. In this study, we followed-up the evolution under prophylaxis with Fraxiparine® of 81 pregnant patients with inherited thrombophilia, of which 63 gave birth until the end of the study. Laboratory and adjacent tests were performed to determine the occurrence of complications related to thrombophilia, but also the adverse effects of the anticoagulant treatment. Results. Fraxiparine® was found not to be associated with dangerous adverse effects. Discussion. Procoagulant and fibrinolytic factors are involved in hemostasis. In addition, besides the coagulation cascade, the complement cascade, vascular factors, and quinine also play a role. Platelet aggregation can be diminished, being involved in pregnancy bleeding, especially at birth. The retractability of the clot may also be diminished by the occurrence of thrombasthenia, which should be monitored especially under anticoagulant treatment when a paradoxical thrombocytopenia syndrome might occur. Conclusions. We did not find any dangerous side effects, not even HIT syndrome (heparin-induced thrombocytopenia), which is common in other anticoagulant treatments.
Trombocitopenia la gravidele cu trombofilie tratate cu heparine cu greutate moleculară mică
Thrombocytopenia in pregnant women with thrombophilia treated with low-molecular-weight heparin
First published: 18 aprilie 2019
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/ObsGin.67.1.2019.2295
Abstract
Rezumat
Trombofilia este o patologie asociată cu risc de tromboembolism venos (TEV) în sarcină. Scopul lucrării. Utilizarea anticoagulantelor în sarcină necesită luarea în considerare a potenţialelor riscuri pentru mamă şi făt. Materiale şi metodă. În acest studiu am urmărit evoluţia profilaxiei în cazul a 81 de paciente gravide cu trombofilie moştenită tratate profilactic cu Fraxiparine®, din care 63 au născut până la finalul studiului. S-au efectuat teste paraclinice pentru a putea determina apariţia unor complicaţii legate de trombofilie, dar şi efectele adverse ale anticoagulantului. Rezultate. Nu s-au constatat efecte adverse în cazul utilizării Fraxiparinei®, nici măcar trombocitopenia asociată altor heparine cu greutate moleculară mică (LMWH). Discuţie. În hemostază sunt implicaţi factori procoagulanţi şi fibrinolitici. În plus, pe lângă cascada coagulării, joacă un rol şi cascada complementului, factorii vasculari şi chininele. Agregarea trombocitară poate fi diminuată, având un rol important în sângerările din sarcină şi implicit la naştere. Retractilitatea cheagului poate fi, de asemenea, diminuată prin apariţia trombasteniei, proces care ar trebui urmărit în special sub tratamentul anticoagulant, când poate apărea un sindrom paradoxal de trombocitopenie, fenomen studiat în cazul prezentat de noi. Concluzii. Acest sindrom paradoxal – denumit sindrom TIH (trombocitopenia indusă de heparină) – nu a apărut, potrivit studiului nostru.
Introduction
Thrombophilia is an important and highly debated medical issue, and the disease is often undiagnosed, with a series of severe comorbidities, including death(1-3). The management of thrombophilia involves anticoagulant therapy, but the use of these agents is not very well known anyway. The fact that there are a few studies in pregnancy and the recommendations are based mostly on investigations carried out on non-pregnant women(4-6).
Thrombophilia is associated with increased venous thromboembolism and placental damage, up to fetal loss and thrombocytopenic purpura in the newborn(1-5, 7-10).
The coagulation and fibrinolysis systems are two separate but linked enzyme cascades that regulate the formation and segmentation of the fibrinogen. The blood coagulation system, or the coagulation pathway, is a proteolytic cascade. Thrombophilia is a pathology recently introduced into the clinical practice.
The thrombophilias are classified as inherited and acquired. The inherited ones are those with inherited defects of the factors involved in the coagulation cascade. The acquired one is caused by the antiphospholipid antibodies developed in various pathologies. APLAs are antibodies directed towards phospholipid-binding proteins. These are lupus anticoagulant and anticardiolipin antibodies associated with autoimmune diseases(6,11).
Pregnancy is physiologically a state of hypercoagulability. Thus, when thrombophilia overlaps it, we must carefully evaluate the balance of haemostasis(6,12-21).
Heparin-induced thrombocytopenia (HIT) represents the decrease in platelet count due to the anticoagulant treatment(20).
Recently, HIT is considered a paradoxical syndrome induced by the antithrombotic treatment, an important side effect for pregnant women.
Materials and method
In this study, the data was collected using Microsoft Excel. The statistical analysis was performed with SPSS v17. The use of these programs was possible due to the collaboration with the “Victor Babeş” University of Medicine and Pharmacy from Timişoara.
The study included 81 patients from Hunedoara county with various gestational ages, of whom 63 gave birth until its completion. It was a prospective study which took place in the Department of Obstetrics and Gynecology of the “Dr. A Simionescu” Municipal Hospital, Hunedoara. All of the pregnancies were correctly supervised over a six-year period.
The patients underwent various paraclinical tests to determine the severity of their condition and the adverse effects of the medication to which they had been subjected to.
We used the following exclusion criteria: infections, chronic HTA or renal disease, drug abuse, karyotype or congenital abnormalities, placental or umbilical abnormalities, diabetes, low progesterone.
The numerical variables were compared with the nonparametric Mann-Whitney test (we compared here 63 pregnant women with thrombophilia under Fraxiparine® treatment with 63 healthy pregnant women). The statistical significance was considered for p<0.05.
Results
In this study, we had a group of Caucasian women, aged 26 to 41 years old, in which we observed their responses to the use of Fraxiparine® during pregnancy. We calculated the central trend parameters and plotted the histogram of this variable (Table 1 and Figure 1). The volume of the sample in this case was 63, because only 63 of the 81 women reached the delivery date by the end of the study.

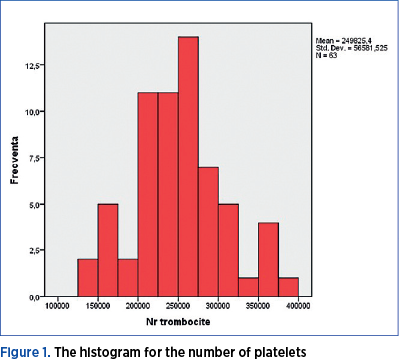
Fraxiparine® was found not to be accompanied by this dangerous adverse effect, as outlined in Table 1.
Only one patient developed thrombocytopenia during pregnancy (under 100,000 platelet/mm3), and this occurred in the third trimester of pregnancy. The treatment used was Fraxiparine® 0.4 IU/day in all cases.
We used a Mann-Whitney test, and we obtained a significantly lower p-value (p<0.001).
Heparin treatment has also side effects which we present in Table 2. We noticed that they were a few, rarely causing bleeding, osteoporosis, and allergic reactions such as hives or skin rash. In newborns – in which the physiology of the hemostasis has some particularities – the thrombocytopenic purpura described in the literature was not encountered.
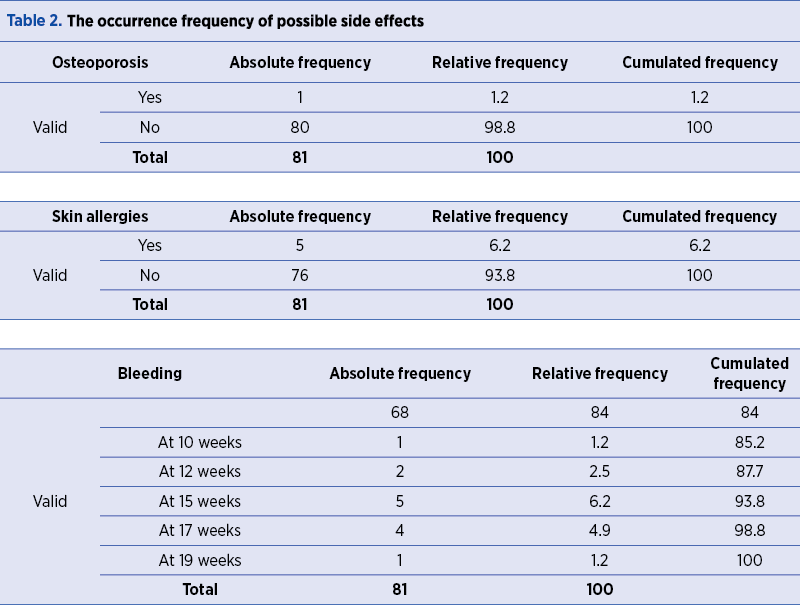
There is no specific adverse reaction for most low-molecular-weight heparin (LMWH) treatments, except for HIT syndrome. All pregnant women with thrombophilia used Fraxiparine® until delivery and six weeks after that, and all of them signed an informed consent regarding the study.
Discussion
The risk of thromboembolism is six times higher in pregnant women than in non-pregnant women, according to literature data. Also, the women with a history of venous thromboembolism (VTE) have a 3-4 times higher risk of recurrence. Studies up until now suggest that the pharmacological thromboprophylaxis is underused in at least 30% of patients at high and very high risk of VTE and in over 50% of those at medium risk. The causes are multiple. However, associating thrombophilia with pregnancy complications presents methodological limitations which make it difficult to obtain accurate data. A significant proportion of the population has a detectable abnormalities, but most of them develop thrombosis in the presence of an additional risk factor(8-10).
Conclusions
The platelet count is very important for the pregnant woman, especially near the date of delivery, as giving birth is a process associated with an important loss of blood. This is why we considered it necessary to do also a descriptive statistic on the numeric variable platelet count.
Until now, low-molecular-weight heparin has been found to be able to induce long-term thrombocytopenia, and should be monitored in pregnancy. But in our study we obtained good results with Fraxiparine®, which is the only LMWH not accompanied by thrombocytopenia and bleeding risk at delivery.
However, the treatment in pregnancy should be judiciously evaluated, taking also into account the physiological changes in pregnancy, especially when approaching the delivery date, which is a process accompanied by major bleeding risk(21,22).
Conflict of interests: The authors declare no conflict of interests.
Bibliografie
2. Mitranovici MI, Puşcaşiu L, Craina M, Iacob D, Chiriac VD, Ioniţă I, Moleriu RD, Furau G, Sisu A, Petre I. The Role of Low Molecular Weight Heparin in Pregnancies of Patients with Inherited Thrombophilia that Have Presented (and) Thrombotic Complications During Previous Pregnancies. Revista de Chimie. 2017; Bucureşti, vol 68, nr. 12, pag. 2970–2973.
3. Iordache O, Anastasiu D, Craina M, Moleriu LC, Ioniţă I, Furau C, Iacob Daniela, Bacean-Miloicov OC, Petre I. Hereditary Thrombophilias and their Involvement in Spontaneous Abortion. The 13th Conference of the Romanian-German Society of Obstetrics and Gynecology,Timişoara, Romania, 14-16 September, pag 166–170, Ed. Filodiritto, 2017.
4. Nicolaides AN, Breddin HK, Cervantier P, et al. Thrombophilia and venous thromboembolism. International consensus statement. Guidelines according to scientific evidence. Int Angiol. 2005; 24, 1-26.
5. Crowther MA, Kelton JC. Congenital thrombophilic states associated with venous thrombosis: a qualitative overview and proposed classification system. Ann Intern Med. 2003; 138, 128-134.
6. Middeldorp S, Libanred EJ, Hamulyak K, et al. The risk of pregnancy-related venous thromboembolism in women who are homozygous for factor V Leiden. Br J Haematol. 2001; 113, 550–555.
7. Iordache O, Petre I, Craina M, Moleriu RD, Boglut A, Iacob D, Miloicov-Bacean OC, Ioniţă I. Thrombophilia and Pregnancy. The 5th Congress of the Romanian Society of Ultrasound in Obstetrics and Gynecology, Filodiritto Editore Proceeding, Târgu-Mureş, Romania, 20-22 April. 2017; 497-500.
8. Ioniţă I, Grigoriţa L, Miloicov-Bacean OC, Petre I, Bernad E, Craina M, Diaconu M, Citu C, Felix Radu, Oros D, Boglut A, Furau G, Enătescu V. The Role of Thrombophilia in Pregnancy. Revista de Chimie. 2016; vol 67, no 12, pag. 2643-2647.
9. Moza A, Petre I, Pantea S. The Management of Pregnancy with Hereditary Angioedema. The 13th Conference of the Romanian-German Society of Obstetrics and Gynecology, Timişoara, Romania. Ed. Filodiritto Editore Proceeding. 2017; pag. 184–188.
10. Bonte DC, Olivera I, Petre I, Craina M, Chiriac VD, Stoian D, Moleriu LC, Sisu A. Macroscopic Examination of Placental Vascularization with a Corrosive Agent in Pregnant Women Diagnosed with Thrombophilia. Revista Materiale Plastice. 2017; 54, no. 4, pag 678–681.
11. Legnani C, Palareti G, Gnazzaloca G, et al. Venous thromboembolism in young women: role of thrombophilic mutations and oral contraceptive use. Eur Heart J. 2000. 23, 984–990.
12. Toth G, Anastasiu D, Craina M, Cîrlogea A, Moleriu RD, Citu I, Citu C, Ioniţă I, Petre I. First Trimester Screening for Preeclampsia. The 13th Conference of the Romanian-German Society of Obstetrics and Gynecology, Timişoara, Romania. Ed. Filodiritto Editore Proceeding. 2017; pag. 304–309.
13. Toth G, Petre I, Craina M, Moleriu RD, Boglut A, Iacob D, Ioniţă I, Miloicov-Bacean OC. Paraclinical Correlations and the Macroscopic Aspect of Placenta in Cases of HTAIS Diagnosis. The 5th Congress of The Romanian Society of Ultrasound in Obstetrics and Gynecology. Filodiritto Editore Proceeding, Târgu-Mureş, Romania, 2017; pag. 482–486.
14. Petre I, Folescu R, Băcean O, Bordianu A, Luca IM, Bernad E, Craina ML. The macroscopic examination of the placental vasculature with a corrosive agent. Romanian Journal of Morphology and Embriology. 2014; vol. 55, no. 2 Suppl., pag. 613-617.
15. Rusu MC, Pop E. Fenestrated vertebral artery. Anat Sci Int. 2013 Sep; 88(4):249-53.
16. Nicolov M, Heghes A, Petre I, Mederle OA. Theoretical and Experimental Analysis of SiO2 – a Proof of Hartman Perdok Method. Revista de Chimie. 2018; Bucharest, vol. 69, no. 4, pag. 949–951.
17. Ivan MV, Petre I, Vlaicu B, Apostol A, Tesloianu D, Munteanu M, Costăchescu R, Moleriu LC, Lazăr F. The Use of Pulse Wave Velocity in Predicting Pre-Eclampsia in High-Risk Women. Revista de Chimie. 2018; Bucharest, vol. 69, no. 5, pag. 1260–1263.
18. Manea A, Boia M, Iacob D, Dima M, Iacob RE. Benefits of early enteral nutrition in extremely low birth weight infants. Singapore Med J. 2016 Nov; 57(11):616-618.
19. Iordache O, Anastasiu D, Craina M, Moleriu LC, Boglut A, Cîrlogea A, Petre I. Natural Delivery Versus Caesarean Section – Statistical Data – Advantages and Disadvantages. The 13th Conference of the Romanian-German Society of Obstetrics and Gynecology, Timişoara, Romania, 14-16 September 2017, Ed. Filodiritto Editore Proceeding, pag. 171–176.
20. Warkentin TE, Sheppard JAI, Horsewood P, et al. Impact of the patient population on the risk for heparin-induced thrombocytopenia. Blood. 2000; 96:1703-8.
21. Alving BM. How I treat heparin-induced thrombocytopenia and thrombosis. Blood. 2003; 101: 31-7.
22. Hui C, Lili M, Libin C, Rui Z, Fang G, Ling G, Jianping Z. Changes in coagulation and hemodynamics during pregnancy: a prospective longitudinal study of 58 cases. Arch Gynecol Obstet. 2012; 285(5):1231-6.