Introduction. Studies have shown that 60-80% of extremely low birth weight (ELBW) newborns develop hyperglycemia in the first two weeks of life. The cause is usually iatrogenic, through high rates of exogenous glucose infusion from total parenteral nutrition (TPN). Other mechanisms are represented by insulin resistance due to the immaturity of enzyme systems, increased hepatic production of glucose, the inability to inhibit gluconeogenesis, and by the effect of stressors through the action of catecholamines. Hyperglycemia generates multiple harmful effects, which can be reduced with the help of insulin therapy, optimizing growth and avoiding fluid overload. Materials and method. We present the case of three ELBW newborns with neonatal respiratory distress, who required surfactant and invasive mechanical ventilation, along with prolonged TPN. The newborns developed persistent hyperglycemia in the first weeks of life (plasma glucose above 180 mg/dl) despite the decrease in glucose concentration and infusion rate, requiring insulin infusion. Results. In all three cases, it was relatively difficult to control the plasma glucose levels. Weight gain was satisfactory, but the patients presented multiple severe complications. Conclusions. The management of metabolic complications of ELBW newborns is important due to the major implications in their prognosis and survival. A special interest has recently been given to the concept of cellular modulation in critical periods of development and the influence in the subsequent occurrence of some diseases. Extrauterine adaptation in ELBW newborns involves biochemical and metabolic adjustments, with immediate and long-term impact.
Dificultăţi în managementul hiperglicemiei la nou-născuţii cu greutate extrem de mică la naştere – prezentare de caz
Difficulties in managing hyperglycemia in extremely low birth weight infants – case report
First published: 30 noiembrie 2022
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/Gine.38.4.2022.7392
Abstract
Rezumat
Introducere. Studiile au arătat că 60-80% dintre nou-născuţii cu greutate extrem de mică la naştere (ELBW) dezvoltă hiperglicemie în primele două săptămâni de viaţă. Cauza este de obicei iatrogenă, prin rate mari de perfuzie exogenă de glucoză din nutriţia parenterală totală (NPT). Alte mecanisme sunt reprezentate de rezistenţa la insulină din cauza imaturităţii sistemelor enzimatice, creşterea producţiei hepatice de glucoză, incapacitatea de a inhiba gluconeogeneza şi de efectul factorilor de stres prin acţiunea catecolaminelor. Hiperglicemia generează multiple efecte nocive, care pot fi reduse cu ajutorul terapiei cu insulină, optimizând creşterea şi evitând supraîncărcarea cu lichide. Materiale şi metodă. Prezentăm cazul a trei nou-născuţi ELBW cu detresă respiratorie neonatală, care au necesitat surfactant şi ventilaţie mecanică invazivă, alături de NPT prelungită. Nou-născuţii au dezvoltat hiperglicemie persistentă în primele săptămâni de viaţă (glucoză plasmatică mai mare de 180 mg/dl), în ciuda scăderii concentraţiei de glucoză şi a ritmului de infuzie, necesitând administrarea de insulină. Rezultate. În toate cele trei cazuri, a fost relativ dificil să se controleze nivelurile plasmatice de glucoză. Creşterea în greutate a fost satisfăcătoare, dar pacienţii au prezentat complicaţii multiple şi severe. Concluzii. Managementul complicaţiilor metabolice ale nou-născuţilor ELBW este important, din cauza implicaţiilor majore în prognosticul şi supravieţuirea acestora. Un interes deosebit a fost acordat recent conceptului de modulare celulară în perioadele critice de dezvoltare şi influenţei în apariţia ulterioară a unor boli. Adaptarea extrauterină la nou-născuţii ELBW implică ajustări biochimice şi metabolice, cu impact imediat şi pe termen lung.
Introduction
Hyperglycemia can be a severe complication in the first weeks of life for 60-80% of extremely low birth weight (ELBW) infants, and it has been associated with higher risks involving the morbidity and mortality of these newborns(1). There is still an absence of agreement regarding the strict definition, diagnosis and treatment of hyperglycemia, as well as the long-term implications. Therefore, numerous studies consider as a threshold a value that varies between 126 and 239 mg/dl (7-13.3 mmol/l)(2). Although the European Society for Pediatric Gastroenterology, Hepatology and Nutrition (ESPHGAN) advise avoiding blood glucose above 145 mg/dl, the most widely used threshold for intervention is blood glucose above 180 mg/dl (10 mmol/l)(3,4). Not only the blood glucose level determines the neonatal adverse effects of hyperglycemia, but also the duration, gestation and the underlying illness of the newborn(5). However, the most frequent complications of hyperglycemia in ELBW infants are due to the osmolar changes, the existing evidence demonstrating that blood glucose level above 360 mg/dl (20 mmol/l) will generate important osmolar changes(6).
In ELBW infants, hyperglycemia can be determined by numerous factors, such as: inadequate insulin secretion, continuous glucose liver production, insulin resistance, increased stress hormones (epinephrine, norepinephrine), delayed enteral feedings, sepsis (high levels of proinflammatory cytokines), maternal and neonatal medications (dopamine, dobutamine, epinephrine, corticosteroids, caffeine, phenytoin), iatrogenic, due to high rates of exogenous glucose infusion from total parenteral nutrition (TPN). In terms of clinical findings, there are no specific signs associated to hyperglycemia, only related to the underlying illness, such as dehydration, increased urine output, fever or feeding difficulty. Persistent hyperglycemia can lead to osmotic diuresis, electrolyte imbalance, including hypernatremia, intraventricular hemorrhage (IVH), necrotizing enterocolitis (NEC), retinopathy of prematurity (ROP) and chronic lung disease (CLD)(7,8).
Hyperglycemia can damage the cells and determine an overexpression of GLUT-1, GLUT-2 and GLUT-3 (insulin independent glucose transporters), that will produce an increase in glucose uptake by endothelial, immune, hepatic and nerve cells(9).
Undoubtedly, the widely approach to the treatment of hyperglycemia is the restriction of glucose intake to a minimum of 4-6 mg/kg/min glucose infusion rate (GIR) and insulin therapy, along with treating the possible underlying cause. Recent studies suggest that, if necessary, the lowest GIR could be 3.7 mg/kg/min, which represents the necessary level to maintain the minimum metabolism of the infant. The amino acids that are part of the parenteral nutrition play an important role, because it has been demonstrated that they stimulate insulin secretion; moreover, the low level of plasma arginine was correlated to hyperglycemia(13).
Insulin is an important factor in glucose homeostasis and promotes glucose uptake, leading to an increased transfer of glucose, amino acid and potassium into insulin sensitive cells(12). When considering insulin therapy, we need to assess the benefits as well as the risks associated, mainly hypoglycemia and hypokalemia, but also the anabolic effect, therefore insulin infusion warrants close surveillance. One of the advantages that insulin therapy may provide is to regulate the levels of insulin-like growth factor-1 (IGF-1), which is responsible for increasing glycogen synthesis and for the use of peripheral glucose that can decrease hepatic glucose production. It is essential to avoid excess glucose delivery, because surpassing maximum glucose oxidation rates can lead to a higher production of carbon dioxide, lipogenesis and fat deposition, even to liver steatosis(14).
Retinopathy of prematurity, a proliferative vascular disorder of the immature retina, has been associated with low levels of IGF-1. These levels are reduced after preterm birth and in several other situations, such as relative insulin deficiency, starvation or acute illness. Subsequent publications have recently underlined the association between hyperglycemia and retinopathy of prematurity, describing hyperglycemia as a new risk factor for ROP in ELBW infants. Garg et al. pointed out that, “for every 10 mg/dl increase of serum glucose, there was a 2.7-fold increase in the risk of developing ROP”, and each supplementary day of hyperglycemia increases the risk to develop ROP by 7%(7,11).
It has not been established so far with certainty whether the prolonged hyperglycemia has implications on the long-term neurocognitive outcome in case of ELBW infants, but the existing evidence has demonstrated the association between the duration of hyperglycemia and a poor neonatal prognosis.
Clinical presentation
We present the case of three ELBW infants with an average gestational age of 24 weeks of gestation, who presented persistent hyperglycemia, which developed in the first week of life, despite lowering the GIR and starting insulin therapy. The newborns were hospitalized in the Neonatal Intensive Care Unit (NICU) of the Emergency University Hospital Bucharest, between 5 January 2022 and 15 July 2022, where they required thermal comfort and appropriate humidity, invasive mechanical ventilation, broad-spectrum intravenous antibiotic therapy, inotrope and vasopressor support, caffeine, along with TPN.
All newborns were investigated for pathologies associated with hyperglycemia and insulin therapy: early onset and late onset neonatal sepsis, respiratory distress syndrome (RDS), CLD, IVH, NEC, periventricular leukomalacia (PVL) and ROP. The patients were discharged home after prolonged NICU hospitalization, and they were included in the follow-up program requiring long time surveillance.
Case 1
A 29-year-old woman was referred to our unit at 23 weeks and 1 day of gestation with ruptured membranes for more than 24 hours. Subsequently, an emergency caesarean section was performed. A female infant (birth weight 600 g) required resuscitation maneuvers in the delivery room (Apgar score 2/3/7), intubation one minute after birth and the administration of natural surfactant for RDS due to surfactant deficiency. After admission to NICU, the infant was stabilized, requiring invasive mechanical ventilation for 40 days, later non-invasively for 30 days, SaO2 between 90-95%, intravenous antibiotic therapy, TPN, blood pressure maintained within normal limits with the help of inotropic and vasopressor treatment with dopamine and dobutamine. To maintain blood glucose (BG) levels, intravenous glucose infusion was started with 6 mg/kg/min of GIR.
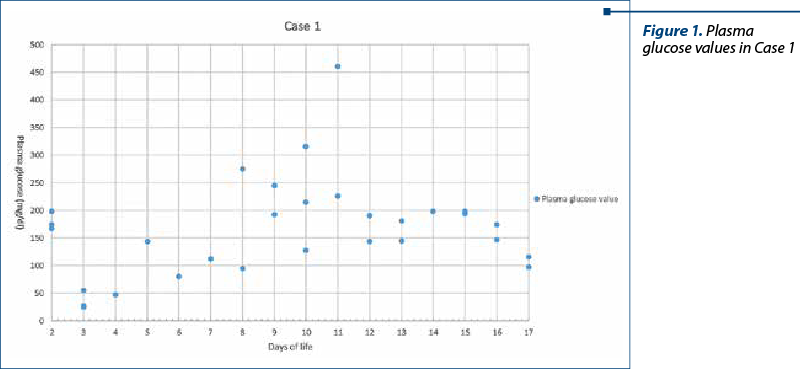
From the second day of life, the newborn presented persistent hyperglycemia, after developing severe pneumothorax, which required thoracentesis, with values above 180 mg/dl, with a maximum value of 460 mg/dl, associated with glycosuria (+1/+2); initially, we lowered the GIR to 4 mg/kg/min in association with the reduction of lipid infusion to 0.5-1 g/kg/day, but with no success. Afterwards, bolus insulin therapy was initiated in a minimal dose of 0.05 IU/kg/dose, for a short period of time, until it was clear that hyperglycemia was persistent. Since we did not achieve stable glycemic control and we encountered an alternation between hypoglycemia and hyperglycemia episodes, we decided to initiate continuous insulin infusion accompanied by increasing the GIR slowly by 0.5-1 mg/kg/min/day, obtaining good glycemic control. When attempting to stop the continuous insulin infusion, hyperglycemia recurred, therefore insulin infusion was reintroduced at BG levels above 180 mg/dl, with a favorable evolution of the patient, tolerance of enteral feeding from the 12th day of life, ascending weight curve, with the possibility of stopping insulin therapy after 11 days of treatment.
The associated pathologies in the first case were RDS, CLD, grade III IVH, without PVL lesions, early- and late-onset neonatal sepsis and grade II ROP, which required Avastin® therapy.
Case 2
A 25-year-old woman was referred to our unit at 25 weeks of gestation, with a twin pregnancy, obtained by artificial insemination, threat of abortion from 20 weeks, with ruptured membranes more than 72 hours before. An emergency caesarean section was performed and she gave birth to a female infant, who died approximately 5 hours postnatally, and to a male infant. The male infant, twin II (birth weight 700 g), required resuscitation maneuvers in the delivery room (Apgar score 4/5/7), being intubated 3 minutes after birth, with the administration of natural surfactant for RDS due to surfactant deficiency. In NICU, the infant required invasive mechanical ventilation for 52 days, later noninvasive for 60 days, SaO2 maintaining between 85% and 95%, intravenous antibiotic therapy, TPN, inotropic and vasopressor support for arterial hypotension from the first day of life. In order to maintain the BG levels, intravenous glucose infusion was started with 4 mg/kg/min of GIR.
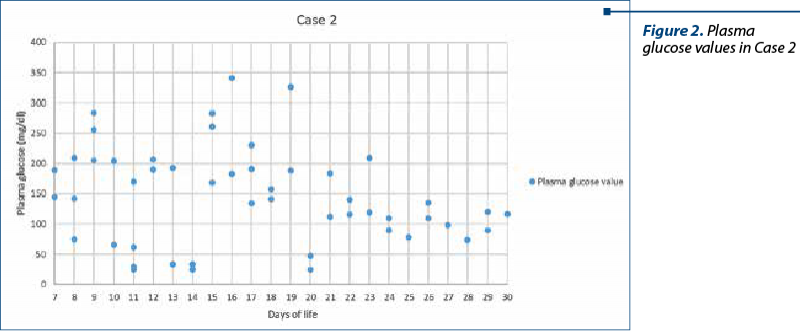
From the seventh day of life, the infant presented persistent hyperglycemia, with values above 180 mg/dl, associated with glycosuria (+2). At first, we lowered the GIR to a minimum of 4 mg/kg/min, at the same time with the reduction of lipid infusion to 0.5-1 g/kg/day, with no significant improvement, therefore we decided to initiate continuous insulin infusion, accompanied by increasing the GIR slowly by 0.5-1 mg/kg/min/day. Unfortunately, due to the underlying critical illness, it was very difficult to maintain a stable level of blood glycemia, and we recorded several episodes of severe hypoglycemia and also hyperglycemia. The lowest and the highest value recorded were 25 mg/dl, respectively 326 mg/dl. After the stabilization of the BG values, an attempt was made to stop the continuous infusion with insulin and bolus insulin therapy was initiated in doses of 0.05 IU/kg/dose at a BG level above 180 mg/dl for a limited period of time. We obtained poor glycemic control, so continuous infusion with insulin was reinitiated, with slowly favorable evolution of the patient, tolerance of enteral nutrition from the 22nd day of life, upward weight curve, with the possibility of stopping insulin therapy after 21 days of treatment, concurrently with the stabilization of the patient’s clinical condition.
The associated pathologies in the second case were RDS, CLD, NEC, grade III IVH with right paraventricular infarction, without PVL lesions, early- and late-onset neonatal sepsis and grade II ROP.
Case 3
A 26-year-old woman was referred to a level II maternity hospital at 24 weeks of gestation, with infectious risk (mother with hepatitis C virus infection), and an emergency caesarean section was performed. A female infant (birth weight 690 g), who required resuscitation maneuvers with noninvasive ventilation in the delivery room and subsequent administration of surfactant for RDS due to surfactant deficiency, arrived by transfer at 5 hours of life at our unit. When admitted in NICU, initially non-invasive mechanical ventilation was ensured (22 days), later invasive (25 days), broad-spectrum intravenous antibiotic therapy for early neonatal sepsis, TPN, inotropic and vasopressor support for arterial hypotension that occurred in the first days of life. In order to maintain the BG levels, intravenous glucose infusion was started with 6 mg/kg/min of GIR.
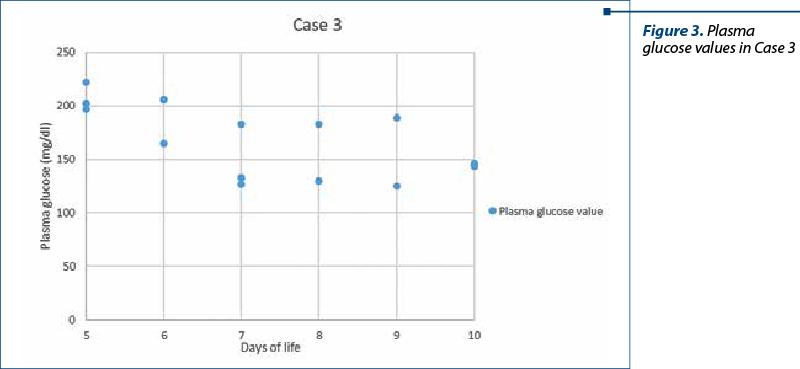
From the fifth day of life, the infant presented persistent hyperglycemia, with values above 180 mg/dl, associated with glycosuria (+1). In the first stage, the GIR and glucose concentration were recalculated, being reduced to 4 mg/kg/min, associated with the reduction of lipid infusion to 0.5-1 g/kg/day, but with the persistence of hyperglycemia. Later, insulin infusion was initiated, obtaining good glycemic control, with a maximum glucose value of 222 mg/dl, tolerance of enteral nutrition from the eighth day of life, with upward weight curve. Insulin therapy was stopped in this case after five days of treatment.
The associated pathologies in the third case were RDS, CLD, grade II-III IVH, without PVL lesions, early- and late-onset neonatal sepsis and grade I ROP.
Discussion
The findings in this report indicate that hyperglycemia in our three cases of ELBW occurred in the first days of life and, unfortunately, in two of the three cases lasted more than 14 days.
As guidelines recommend, in all three cases, we started the management of hyperglycemia by reducing the GIR, with no improvement in the BG values, a situation that required further administration of insulin therapy. Contrary to our expectations, it was noticed that there were oscillations of hypoglycemia and hyperglycemia during the administration of insulin therapy both in continuous and bolus infusion, leading to a very slow and challenging process of stabilization of the glucose values. Nevertheless, it was no surprise, considering the complexity of the associated pathologies and the extreme prematurity in two of the cases (Case 1 and Case 3).
Regarding Case 2, after the relative stabilization of blood glucose after 19 days of life, upon the attempt to stop the continuous infusion with insulin, hyperglycemia reappeared, so we decided to increase the GIR in accordance with continuous insulin infusion, with slowly favorable results, with the possibility of decreasing the rate of continuous insulin infusion and stopping it 21 days later.
It is also important to mention that, in all three cases, during the first days of life we could not initiate oral feeding, but continuing the amino-acid enriched TPN at the same time as the insulin infusion, we recorded a considerable improvement of the digestive tolerance of infants, leading to a significant increase in calorie intake and to a weight gain of 15-20 g/kg/day, but even so, the birth weight was very slowly regained.
Intensive care interventions, such as inotropic support, mechanical ventilation and caffeine administration, metabolic imbalances and the stress associated with the invasive maneuvers to which the newborns were subjected were followed by high levels of BG which suggests that all the aforementioned situations had a significant effect on the BG values. After the inotropic support was decreased and finally stopped and the mechanical ventilation was switched to noninvasive, a stabilization of the BG values was observed in all three cases, suggesting that the management of the underlying illness is essential, rather than just trying to achieve a normal BG level.
We found it very difficult to stop the administration of vasopressor drugs during the critical phase of illness of the two ELBW infants (Case 1 and Case 2) and we noticed a strong correlation between the use of dopamine and the peaks of hyperglycemia; moreover, the addition of norepinephrine or hydrocortisone made us increase the insulin dose to maintain a relative normal and stable level of BG level.
As multiple studies suggest, in our cases, persistent hyperglycemia was also associated with NEC, IVH and infection, and the severity of these complications was strictly correlated with the duration of persistent hyperglycemia and insulin infusion, although the effect of extreme prematurity could have had a strong impact also.
Retinopathy of prematurity, a multifactorial disorder, is known to be related to oxygen therapy and the associated oxidative stress, nutritional status and growth, all the above factors being found in the evolution of our three ELBW infants. Unfortunately, the existing evidence has demonstrated that hyperglycemia has recently been acknowledged as an independent risk factor for the occurrence of ROP. We hereby consider that, in our cases, there could be a correlation between the severity of the ROP with the duration of hyperglycemia and the prolonged insulin therapy, Cases 1 and 2 having the most severe ocular damage compared to Case 3. However, the infants in the first two cases had the most critical condition, with the underlying illness that imposed aggressive therapy, prolonged respiratory and inotropic support, along with prolonged TPN, all leading to important glucose imbalance, difficult to handle considering their prematurity associated pathologies.
Conclusions
In ELBW infants, the first weeks of life are critical to their development, and prolonged hyperglycemia is one of the factors that can be damaging, with short-term and long-term clinical implications. Furthermore, it has not been established so far if the adverse effects of hyperglycemia are determined by the immediate results or by the consequences of the relative insulin deficiency, that can lead to reduced IGF-I generation, therefore influencing metabolism and growth(10).
There are still controversies regarding the optimal clinical targets, thresholds for intervention and treatment approaches. Thereby, in the light of the necessity of high GIR, the low glucose uptake rate, the inadequate glucoregulatory hormone control and, ultimately, the improvement in survival of the ELBW infants, hyperglycemia remains to be expected in the NICU incredible small patients.
Financial support: none declared.
Conflict of interests: The authors declare no conflict of interests.
Bibliografie
-
Kao LS, Morris BH, Lally KP, Stewart CD, Huseby V, Kennedy KA. Hyperglycemia and morbidity and mortality in extremely low birth weight infants. J Perinatol. 2006;26(12):730-6.
-
Morgan C. The potential risks and benefits of insulin treatment in hyperglycaemic preterm neonates. Early Hum Dev. 2015;91(11):655-9.
-
Alsweiler JM, Kuschel CA, Bloomfield FH. Survey of the management of neonatal hyperglycaemia in Australasia. J Paediatr Child Health. 2007;43:632-5.
-
Mesotten D, Joosten K,van Kempen A, Verbruggen S, ESPGHAN/ESPEN/ESPR/CSPEN working group on pediatric parenteral nutrition. ESPGHAN/ESPEN/ESPR/CSPEN guidelines on pediatric parenteral nutrition: carbohydrates. Clin Nutr. 2018;37(6 Pt B):2337-43.
-
Coulthard MG, Hey EN. Renal processing of glucose in well and sick neonates. Arch Dis Child Fetal Neonatal Ed. 1999;81:F92-F98.
-
Hawdon JM. Disorders of blood glucose homoestasis in the neonate. In: Rennie JM, Roberton NRC, ed Textbook of Neonatology, 4th ed. London: Elsevier Churchill Livingstone. 2005:862-5.
-
Garg R, Agthe AG, Donohue PK, Lehmann CY. Hyperglycemia and retinopathy of prematurity in very low birth weight infants. J Perinatol. 2003;23:186-94.
-
Hays SP, Smith EO, Sunehag AL. Hyperglycemia is a risk factor for early death and morbidity in extremely low birth-weight infants. Pediatrics. 2006;118(5):1811-8.
-
Van den Berghe G. How does blood glucose control with insulin save lives in intensive care? J Clin Invest. 2004;114(9):1187-95.
-
Gardelis JG, Hatzis TD, Stamogiannou LN, Donna AA, Fotinou AD, Brestas PS,
-
et al. Activity of the growth hormone/insuline-like growth factor-I axis in critically ill children. J Pediatr Endocrinol Metab. 2005;18(4):363-72.
-
Mohamed S, Murray JC, Dagle JM, Colaizy T. Hyperglycemia as a risk factor for the development of retinopathy of prematurity. BMC Pediatrics. 2013;13:78.
-
Ditzenberger GR, Collins SD, Binder N. Continuous insulin intravenous infusion therapy for VLBW infants. J Perinat Neonatal Nurs. 1999;13(3):70-82.
-
Burgess L, Morgan C, Mayes K, Tan M. Plasma arginine levels and blood glucose control in very preterm infants receiving 2 different parenteral nutrition regimens. JPEN J Parenter Enteral Nutr. 2014;38(2):243-53.
-
Farrag HM, Cowett RM. Glucose homeostasis in the micropremie. Clin Perinatal. 2000;27(1):1-22.
Articole din ediţiile anterioare
Depresia perinatală
Depresia postpartum reprezintă un subiect important în obstetrică, afectând 13% dintre femei în timpul sarcinii sau în primul an post-partum, du...
Complicaţiile perinatale şi neonatale la pacientele cu răspuns ovarian scăzut în sarcinile obţinute prin proceduri de reproducere umană asistată
In vitro fertilization (IVF) technologies with a controlled ovarian hyperstimulation approach have classified patients into three different groups ...
Diabetul gestaţional
Diabetul gestaţional afectează 3-9% din sarcini, implicaţiile acestuia asupra mamei şi copilului fiind uriaşe, întrucât creşte riscul complicaţiilo...
Complicaţiile hipertensiunii arteriale induse de sarcină – management terapeutic şi prognostic
Managementul sarcinii asociate cu hipertensiune gestaţională se realizează în funcţie de severitatea hipertensiunii arteriale, de vârsta ges...