Pityriasis rosea is a skin eruption commonly associated with pregnancy. The present work aims at presenting what obstetricians should know about pityriasis rosea (PR) diagnosis and treatment during pregnancy. PR displays as an acute exanthem disease, nonpruritic, usually located on the thorax and shoulders, with the appearance of erythematous plaques, in a “Christmas-tree pattern” distribution. Due to its etiology (human herpes viruses 6 and 7), the risk of spontaneous abortion can be analyzed. It resolves spontaneously within two months in 80% of cases. There is no evidence for vertical transmission. Pregnancy-associated PR has a favorable outcome and antiviral treatment is not helpful.
Pitiriazisul rozat în timpul sarcinii – ce ar trebui să ştie obstetricienii?
Pityriasis rosea during pregnancy – what obstetricians should know about it?
First published: 18 mai 2020
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/Gine.28.2.2020.3177
Abstract
Rezumat
Pitiriazisul rozat (PR) este o boală eruptivă cutanată care apare frecvent în sarcină. Lucrarea abordează diagnosticul şi tratamentul pe care ar trebui să le cunoască obstetricienii în privinţa acestei patologii. PR apare ca o erupţie eritemato-nepruriginoasă, de obicei localizată pe torace şi umeri, sub forma unor plăci eritemato-scuamoase cu dispoziţie caracteristică, în formă de „pom de Crăciun”. Se poate analiza asocierea cu creşterea riscului de avort spontan din cauza etiologiei – virusurile herpetice 6 şi 7. În 80% din cazuri, se remite spontan. Nu există dovezi ale transmiterii verticale. Sarcina asociată PR are o evoluţie favorabilă, tratamentul antiviral nefiind eficient.
Pregnancy represents a physiological condition associated with many adaptative changes caused especially by hormonal changes, including physiological skin changes. Pityriasis rosea of Gibert (PR) is a dermatological disease that affects young people and women of reproductive age(1). PR is diagnosed more frequently in pregnancy than in the general population(2). PR eruption is manifested during all trimester of pregnancy by acute exanthema, and may cause patients worry and stress. PR is not always diagnosed during pregnancy due to the lack of pruritus; the obstetrician does not notice the skin lesions subsequently(3). While associated with human herpes viruses 6 and 7 (HHV-6/7), PR may be a risk factor associated with spontaneous abortion, intrauterine death(4), or a possible explanation for fetal hydrops(5).
Based on our clinical cases, the aim of this paper is to present what obstetricians should know about pityriasis rosea during pregnancy. Undeniable, the diagnosis will be performed by dermatologist, a multidisciplinary approach being necessary in order to differentiate the pathological skin lesions from skin conditions during pregnancy.
The common physiological skin changes induced by pregnancy encompass three groups of normal cutaneous lesions: pigmentary changes, vascular changes and stretch marks, all of these presumed to occur from the effects of increased levels of melanocyte-stimulating hormone or estroprogestative high levels. Pigmentary changes are well known: hyperpigmentation of the linea alba, of the umbilicus and areolar skin, along with the macular pigmentation of the face and melasma. Dilatation and proliferation of the blood vessels determine vascular changes, including palmar erythema, spider angiomas and venous congestion, which regress spontaneously after delivery. These physiological skin changes may be differentiated from other pathological skin lesions occurred during pregnancy: pruritus gravidorum, pruritic urticarial papules and plaques of pregnancy (PUPPP), prurigo gestationis, pemphigoid gestationis (herpes gestations without any connection with a herpetic etiology), impetigo herpetiformis (pustular psoriasis).
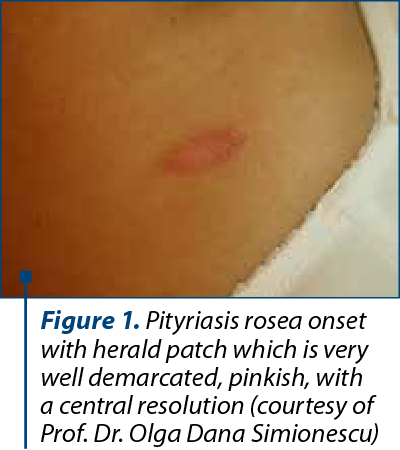
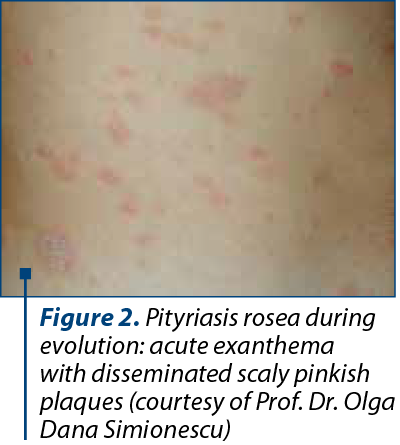
Pityriasis rosea is an acute exanthema, a common papulosquamous eruption. In the classic form, a single herald patch (“mother patch”), with 2-4 cm in diameter, appears on the trunk (Figure 1), enlarges and disseminates over several days. Within days to two weeks, in a second phase of eruption, much smaller lesions will appear, typically smaller and symmetrically oriented with their long axes along the cleavage lines of the trunk (“Christmas-tree pattern”), followed by a generalized eruption (Figures 2 and 3). They resolve spontaneously within two months in 80% of cases but in some cases can last for five months(6). Some reports suggested typical prodromal symptoms like gastrointestinal symptoms, nausea and respiratory symptoms, mild fever, when associated with herpes virus (HSV6/7) infection(7). Considered at the very beginning as a fungal infection and even a viral infection (ARN)(8), PR etiology is now considered a 6,7 herpesviridae-due. Drago et al. (1997) established the pathogenic role of reactivated human herpesvirus HHV-6 and HHV-7 in PR pathogenesis(9). No association was found with different types of herpesviruses: alpha herpesviruses (HHV-1 and HHV-2) from TORCH syndrome and Roseolovirus (HHV-6 and HHV-7), nor cytomegalovirus (CMV); and gamma herpesviruses, which include Epstein-Barr virus in the genus Lymphocryptovirus. Furthemore, others works confirmed that only systemic active infection with HHV-6, especially during the first weeks of gestation, can be connected with a major risk factor for complications during pregnancy. In Japan, M. Jasukawa suggested the reactivation of human herpesvirus 6 (HHV-6) in pityriasis rosea using polymerase chain reaction(PCR) for the virus genomes and virus isolation in peripheral blood mononuclear cells(10). HHV-6 isolated by PCR was found in association with cases with spontaneous abortion in pregnancy before 15 weeks(5).

During pregnancy, immunologic, endocrine, metabolic and vascular changes make the pregnant woman susceptible to aggravation of PR lesions. PR is not contagious. The association with HHV-6/7 makes it possible to question whether there is a vertical transmission of this infection. However, we consider that the 10-15% incidence of PR associated with spontaneous abortion overlaps with the spontaneous abortion rate in the general population(11,12). HHV-6 or 7 infections are considered a significant risk factor for abortion (such as history of a previous abortion or maternal age). Some authors recommended in case of an exanthema involving more than 50% of the body area the assessment of HHV-6 and HHV-7 viral DNA by calibrated quantitative polymerase chain reaction (CQ-PCR) assay in order to consider a high risk for pregnancy. Hall et al. have reported HHV-6 newborn’s cases with congenital infection while HHV-6 and HHV-7 DNA virus have been diagnosed in the vaginal tract of women(13). But HHV-6 or HHV-7 DNA was never detected in amniotic fluid(14). Virus shed in the saliva of asymptomatic contacts is presumed to be the most likely source of infection. Because during pregnancy the immune response is altered, possible viral HHV-6 reactivations are reported(15). The differential diagnosis of PR includes: secondary syphilis, seborrheic dermatosis, eczema or drug-induced PR (barbiturates, clonidine, D-penicillamnine etc.). In case of persistence of exanthema more than five months, supplementary laboratory tests are required.
Unlike in non-pregnant women, PR during pregnancy is not associated with pruritus or with significant clinical symptoms. This may be explained by the increased vascularity during pregnancy, increased activity of sweat glands combined with the patient use of creams and oils for itching and keeping the skin soft persistent. If persistent, severe pruritus should be further evaluated. There are some reports indicating oral antiviral drugs such as acyclovir or valacyclovir (800 mg thrice daily for 7 days), but this medication did not shorten the duration of eruption(9). Antiviral drugs activity against HHS-6/7 is poor because antivirals do not replicate intracellularly and do not inhibit viral thymidine kinase, unlike herspes simplex 1,2 or varicella-zoster virus infection.
In conclusion, an acute exanthema starting with a single herald patch followed by an erythematous exanthema is typical for PR. Pregnancy-associated PR has a favorable outcome and antiviral treatment is not necessary.
Conflict of interests: The author declares no conflict of interests.
Bibliografie
-
Bolognia JL, Schaffer JV, Cerroni L. Pityriasis rosea in Fourth Edition Dermatology, Ed Elsevier, 2018.
-
Corson EF, Luscombe HA. Coincidence of pityriasis rosea with pregnancy. AMA Arch Derm Syphilol. 1950; 62(4):562-4.
-
Eisman S, Sinclair R. Pityriasis rosea. BMJ. 2015; 29, 351:h5233, doi: 10.1136/bmj.h5233.
-
Drago F, Broccolo F, Zaccaria E, Malnati M, Cocuzza C, Lusso P, Rebora A. Pregnancy outcome in patients with pityriasis rosea. J Am Acad Dermatol. 2008; 58(5 Suppl 1):S78-83.
-
Al-Buhtori M, Moore L, Benbow EW, Cooper RJ. Viral detection in hydrops fetalis, spontaneous abortion, and unexplained fetal death in utero. J Med Virol. 2011 Apr; 83(4):679-84.
-
Drago F, Broccolo F, Rebolla A. Pithyriasis rosea: an update with critical appraisal of its possible herpesviral etiology. J Am Acad Dermatol. 2009; 61:303-318.
-
Sharma PK, Yadav TP, Gautam RK, Taneja N, Satyanarayana L. Erythromycin in pityriasis rosea: a double-blind, placebo-controlled clinical trial. J Am Acad Dermatol. 2000; 42:241-244.
-
Machet L, Vaillant L. Dermatologie en gynécologie obstetrique. Ed Masson, 2001.
-
Drago F, Ciccarese G, Herzum A, Rebora A, Parodi A. Pityriasis Rosea during Pregnancy: Major and Minor Alarming Signs. Dermatology. 2018; 234(1-2):31-36.
-
Yasukawa M, Sada E, MacHino H, Fujita S. Reactivation of human herpesvirus 6 in pityriasis rosea. Br J Dermatol. 1999; 140(1):169-70.
-
Ellish NJ, Saboda K, O’Connor JO, Nasca PC, Stanek EJ, Boyle C. A prospective study of early pregnancy loss. Hum Reprod. 1996; 11:406-412.
-
Andersen AM, Wohlfahrt J, Christens P, Olsen J, Melbye M. Maternal age and fetal loss: population based register linkage study. Br Med J. 2000; 320: 1708-1712.
-
Hall CB, Caserta MT, Schnabel KC, Boettrich C, McDermott MP, Lofthus GK, Carnahan JA, Dewhurst S. Congenital infections with human herpesvirus 6 (HHV6) and human herpesvirus7 (HHV7). J Pediatr. 2004; 145(4):472-477.
-
Boutolleau D, Cointe D, Gautheret-Dejean A, Macé M, Agut H, Grangeot-Keros L, Ingrand D. No evidence for a major risk of roseolovirus vertical transmission during pregnancy. Clin Infect Dis. 2003; 15, 36(12):1634-1635.
-
Drago F, Broccollo F, Javor S, Drago F, Rebora A, Parodi A. Evidence of human herpesvirus-6 and-7 reactivation in miscarrying women with pityriasis rosea.J Am Acad Dermatol. 2014; 71(1):198-199.
Articole din ediţiile anterioare
Tratamentul leziunilor dentare cervicale în sarcină
În practica de medicină dentară ne confruntăm frecvent cu necesitatea tratamentelor stomatologice la femeia însărcinată, fie că este vorba despre m...