Refacerea facilitată după chirurgie (ERAS) – recomandări preoperatorii şi postoperatorii pentru operaţia cezariană. Partea a II-a: Nou-născutul şi îngrijirea postoperatorie
Enhanced recovery after surgery (ERAS) – preoperative and intraoperative recommendations for caesarean delivery. Part 2: The neonate and the postoperative care
Abstract
Enhanced recovery after surgery (ERAS) is a standardized concept used to include all the measures taken pre-, intra- and postoperatively to reduce morbidity and facilitate early recovery. It is a paradigm shift that involves the multidisciplinary approach of the surgical patient and comprises a series of measures that should optimize recovery and shorten hospital stay, evolving from good practice advice to a standardized perioperative care program. ERAS guidelines have the aim of improving maternal and fetal outcome by increasing the safety of caesarean delivery (diminishing the rate of complications and readmission and the length of stay), enhancing recovery, improving the quality of health care, and reducing its cost.Keywords
caesarean deliveryenhanced recoveryRezumat
Refacerea facilitată după operaţia cezariană este un concept nou, standardizat, care include un complex de măsuri pre-, intra- şi postoperatorii, care au ca scop reducerea morbidităţii şi grăbirea recuperării după intervenţia chirurgicală. Este o abordare nouă, multidisciplinară, a pacientului chirurgical, bazată pe dovezi medicale şi care urmăreşte optimizarea recuperării postoperatorii şi reducerea spitalizării. Conceptul este reglementat de ghiduri care urmăresc ameliorarea rezultatelor terapeutice materne şi fetale după operaţia cezariană (scăzând rata complicaţiilor, a reinternării şi a duratei spitalizării), facilitând recuperarea, îmbunătăţind calitatea îngrijirilor medicale şi reducând costurile.Cuvinte Cheie
cezarianărefacere facilitatăThe neonate
It is widely accepted by professionals that the delivery of a baby is one of the most stressful life events. Major changes of the newborn physiology (cardiovascular and respiratory) are taking place. Therefore, all operating theatres where are performed caesarean deliveries (CD) must have everything that should be necessary (equipment, staff and training) for immediate neonatal resuscitation, if necessary(1). The status of the newborn will be evaluated by the traditional Apgar score, at 1, 5 and 10 minutes after delivery.
A succession of gestures must be followed after the delivery of a good-looking baby by caesarean section (CS). Immediate cord clamping is recommended in newborn babies with immediate need for resuscitation or in case of placental bleeding.
The delayed clamping of the umbilical cord
It was proven that the delay of clamping for at least 1 minute after delivery at term will decrease the incidence of anemia and will improve the child’s neurodevelopment(2-6). Practically, the newborn is placed on the legs of her mother until the cord is clamped. Cord milking is encouraged.
In preterm infants, delayed cord clamping is recommended for 30 seconds. Studies have shown that these infants have a better prognosis in terms of anemia, necrotizing enterocolitis and intraventricular hemorrhage compared with infants with immediate cord clamping(7).
Clinical trials have shown that delayed cord clamping could be associated with an increased risk for hyperbilirubinemia, thus the department of neonatology must poses all capabilities to monitor and treat the neonatal jaundice(2,3,6).
Prevention of hypothermia
Hypothermia is a cause of neonatal morbidity and even death. The optimal temperature in the operating theatre is 23°C.
This temperature will prevent maternal and neonatal hypothermia and all the detrimental consequences. Immediate covering of the fetal head and body will be the first gesture of the obstetrician or his/her assistants, in order to reduce the heat loss. Body temperature should be constantly monitored and kept between 36.5°C and 37.5°C(8).
Stimulation of the first breathing
Waiting for placental spontaneous detachment, the obstetrician or assistants will gently stimulate the back of the baby in order to initiate spontaneous respirations. Very few neonates will not respond, and neonatal resuscitation should be taken into consideration.
Routine suctioning of the upper airways or gastric aspiration will be strongly discouraged, even in case of meconium staining of the amniotic fluid(9,10). Aspiration will be performed only in case of secretions obstructing the airways.
Routine neonatal supplementation of the inspired air with oxygen is not recommended(11). In case of neonatal resuscitation, it is recommended to use the inspired air of the operating theatre. The early use of caffeine in order to stimulate breathing is still debated.
Skin-to-skin contact
Studies have reported benefits for the newborn and her mother after skin-to-skin contact, which were associated with increased rates and duration of breastfeeding and also with the decrease of the incidence of postnatal depression and maternal anxiety(12,14).
Postoperative care
The checklist of the postoperative caesarean delivery will comprise the following items.
Preventing postoperative nausea and vomiting
Postoperative nausea and vomiting (PONV) can have a negative impact on the recovery after CD. First of all, it could delay the early oral intake of food and drugs. It is more frequent in gravidas receiving neuraxial opioids analgesia for CD(15).
Preventing PONV is a key point of ERAS protocol for CD. For a highly successful regimen, it is recommended a combination of two antiemetics instead of one.
One of the most recommended combinations is odansetron and dexamethasone. Droperidol, an antidopaminergic agent, could be equally used for PONV prevention(16,17).
Early oral intake
Traditionally, the oral intake was allowed when there appeared the first clinical signs of gas sounds or even after the first stool. Recent studies have shown that early oral intake stimulates the return of the bowel function and so does the early ambulation.
Solid diet (low fat/fiber should start late in the afternoon of the same day of CD)(18). The chewing of gum is recommended three times per day, for 30 minutes after meals, to stimulate early bowel function return(19). In the first day post-CD, the patient should receive a normal diet.
Glycemia should be maintained <180 mg/dL and insulin should be used only above this limit(20).
Treatment of pain
Offering an adequate post-CD analgesia is an important part of the ERAS protocols. A suboptimal analgesia will impede early ambulation, increasing the risk of thromboembolic events, will delay the early functional recovery, leading to a poor maternal interaction with her baby, with delay in breastfeeding and increased risk of postpartum depression. All ERAS protocols recommend a multimodal regimen for pain control with the aim to offer the best result with minimal side effects, avoiding, whenever is possible, the opioids.
Acetaminophen orally, 1000 mg, on request, not exceeding 4000 mg per day, is a good option.
Nonsteroidal anti-inflammatory drugs (NSAID) orally (ibuprofen 400-800 mg, every 6 hours), are another option.
The two types of analgesic drugs can be associated routinely, if necessary, and unless contraindicated, they have an additive analgesic effect.
Transversus abdominis block and wound infiltration with long-acting liposomal bupivacaine or ropivacaine are alternative techniques to improve post-CD analgesia(21,22).
Early ambulation
Together with optimal analgesia, the early ambulation improves pulmonary function and tissue oxygenation, reduces the risk of thromboembolic events, reduces the insulin resistance, and shortens the hospital stay.
Thromboprophylaxis
Obstetric venous thromboembolism is a leading cause of severe maternal morbidity and mortality. Maternal death due to thromboembolism can be prevented by thromboprophylaxis
Every gravida prepared for a CD should be carefully evaluated for a potential risk of thrombotic event. Pharmacologic thromboprophylaxis should not be offered routinely(23).
For CD, the American College of Obstetricians and Gynecologists (ACOG) and the American College of Chest Physicians (ACCP) recommend universal perioperative use of pneumatic compression devices in all women who are not already receiving pharmacologic thromboprophylaxis(24,25).
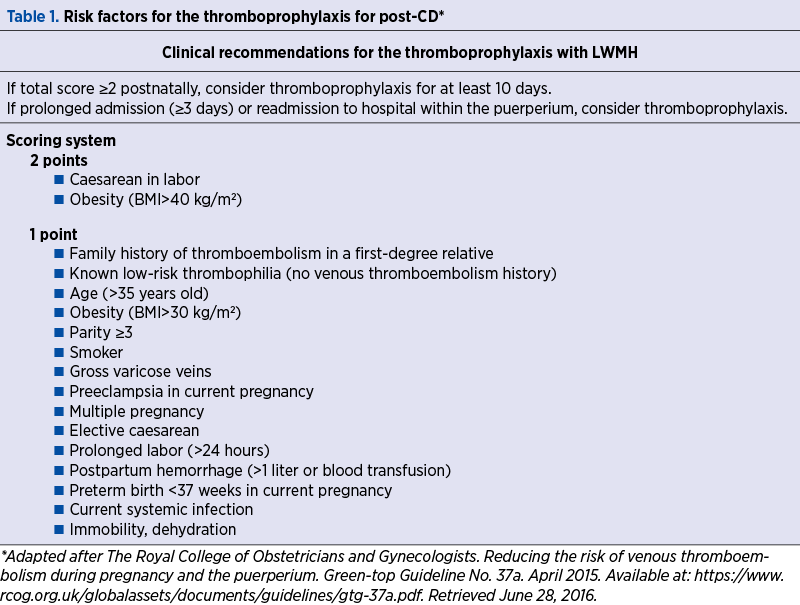
The recommendations of the Royal College of Obstetricians and Gynecologists (RCOG) are more aggressive, considering that in UK thromboembolism is responsible for 31.1% of all maternal deaths(26). The RCOG guidelines published in 2015 recommend prenatal and postnatal thromboprophylaxis with LWMH in all pregnant patients with risk factors (obesity with BMI>30, multiple gestation, age over 35 years old, pregnancy following assisted reproduction etc.).
The National Partnership for Maternal Safety, Consensus Bundle on Venous Thromboembolism (NPMS) makes recommendations after analyzing RCOG, ACOG and ACCP guidelines. The NPMS working group’s approach is supporting the RCOG recommendations for routine administration of LWMH or unfractionated heparin (UFH) in all women identified with risk factors, undergoing CD, unless specific contraindications (Table 1)(27).
Early removal of Foley catheter
ERAS protocols recommend that the bladder catheter should be removed late in the afternoon of the day of CD. In a recent study, the removal of the Foley catheter 7 hours after CD led to no complications(28).
Plan and organize early discharge
The patient who was discharged from the maternity together with her baby must receive written information about a reliable means of communication with the labor and delivery unit and with the neonatology unit (a phone number for calling, a person to contact) in case there are concerns.
ERAS audit
Audit is an important part of the ERAS program. It allows the estimation of compliance to the individual recommendations of the ERAS guidelines. The most analyzed variables are the length of stay in the hospital, surgical site infection, readmission rate and the complications registered within the 30 days post-discharge. Audit has the potential to improve these parameters, increasing the quality of health services and decreasing the costs(29).
ERAS team development
A successful ERAS program must develop as a multidisciplinary team program, evolving and having as final goal the improvement of perioperative outcomes. This team comprises obstetricians, midwives, anesthetists and nurse anesthetists, neonatologists and nurse neonatologists, residents and fellows, outpatient and inpatient nursing team, hospital pharmacist, dietitian, research data coordinator, data manager and statistician. This team should meet at least twice a month in order to communicate and analyze the implementation and compliance of individual aspects of the ERAS program. The team members have to be leaders in their units, supporting and promoting the program(29).
Conclusions
The recommendations of ERAS protocols are based on the most recent and best existing evidence. ERAS is a team project and measuring its support and compliance will be the best required tool for its success and sustainability.
Implementing the ERAS protocols will improve the quality of health care and will diminish the cost of health services.
Conflict of interests: The authors declare no conflict of interests.
Bibliografie
1. Perlman JM, Willie J, Kattwinkel J, et al. Part 7: Neonatal resuscitation; 2015 International Consensus on cardiopulmonary resuscitation and emergency cardiovascular care science with treatment recommendations. Circulation. 2015; 132 (suppl);S204-241.
2. McDonald SJ, Middleton P, Dowswell T, et al. Effect of timing of umbilical cord clamping of term infants on maternal and neonatal outcomes. Cochrane Database Syst Rev. 2013; 7:CD004074.
3. Ashish KC, Rana N, Malqvist M, et al. Effect of umbilical cord clamping vs. early clamping on anemia in infants at 8 and 12 months: a randomized clinical trial. JAMA Pediatr. 2017; 171:264-70.
4. Committee Opinion No. 684 Summary: delayed umbilical cord clamping after birth. Obstet Gynecol. 2017; 129:232-3.
5. Delayed umbilical cord clamping after birth. American Academy of Pediatrics. 2017; 139:e20170957.
6. Bayer K. Delayed umbilical cord clamping in the 21st century: indicators for practice. Adv Neonat Care. 2016; 16:68-73.
7. Tarnow-Mordi W, Morris J, Kirby A, et al. Delayed vs. immediate cord clamping in preterm infants. N Engl J Med. 2017; 377:2445-55.
8. Duryea EL, Nelson DB, Wykhoff MH, et al. The impact of ambient operating room temperature on neonatal and maternal hypothermia and associated morbidities: a randomized controlled trial. Am J Obstet Gynecol. 2016; 214:505.
9. Committee opinion No. 689. Summary: delivery of a newborn with meconium-stained amniotic fluid. Obstet Gynecol. 2017; 129:593-4.
10. Foster JP, Dawson JA, Davis PG, et al. Routine oro/nasopharyngeal suction versus no suction at birth. Cochrane Database Syst Rev. 2017; 4;CD010332.
11. Tan A, Schultze A, O’Donnell CP, et al. Air versus oxygen for resuscitation of infants at birth. Cochrane Database Syst Rev. 2005; 2; CD002273.
12. Bramson L, Lee JW, Moore E, et al. Effect of early skin-to-skin mother-infant contact during the first 3 hours following birth on exclusive breastfeeding during the maternity hospital stay. J Hum Lact. 2010; 26(2):130–7.
13. Bigelow A, Power M, MacLellan-Peters J, et al. Effect of mother/infant skin-to-skin contact on postpartum depressive symptoms and maternal physiological stress. J Obstet Gynecol Neonatal Nurs. 2012; 41(3):369–82.
14. Moore ER, Anderson GC, Bergman N, et al. Early skin-to-skin contact for mothers and their healthy newborn infants. Cochrane Database Syst Rev. 2012; (5):CD003519.
15. Balki M, Carvalho JC. Intraoperative nausea and vomiting during cesarean section under regional anesthesia. Int J Obstet Anesth. 2005; 14(3):230–41.
16. Habib AS, George RB, McKeen DM, et al. Antiemetics added to phenylephrine infusion during cesarean delivery: a randomized controlled trial. Obstet Gynecol. 2013; 121(3):615–23.
17. Wu JI, Lo Y, Chia YY, et al. Prevention of postoperative nausea and vomiting after intrathecal morphine for caesarean section: a randomized comparison of dexamethasone, droperidol, and a combination. Int J Obstet Anesth. 2007; 16(2):122–7.
18. Charoenkwan K, Phillipson G, Vutyavanich T. Early versus delayed (traditional) oral fluids and food for reducing complications after major abdominal gynaecologic surgery. Cochrane Database Syst Rev. 2007; CD004508.
19. Ertas IE, Gungorduk K, Ozdemir A, et al. Influence of gum chewing on postoperative bowel activity after complete staging surgery for gynecological malignancies: a randomized controlled trial. Gynecol Oncol. 2013; 131(1):118–122.
20. Berríos-Torres SI, Umscheid CA, Bratzler DW, et al. Centers for Disease Control and Prevention Guideline for the Prevention of Surgical Site Infection. JAMA Surg. 2017 May; https://doi.org/10.1001/jamasurg 2017.0904
21. Abdallah FW, Halpern SH, Margarido CB. Transversus abdominis plane block for postoperative analgesia after Caesarean delivery performed under spinal anaesthesia? A systematic review and meta-analysis. Br J Anaesth. 2012; 109(5):679–87.
22. Adesope O, Ituk U, Habib AS. Local anaesthetic wound infiltration for postcaesarean section analgesia: A systematic review and meta-analysis. Eur J Anaesthesiol. 2016; 33(10):731–42.
23. D’Alton ME, Friedman AM, Smiley RM, et al. National Partnership for Maternal Safety: Consensus Bundle on Venous Thromboembolism. Anesth Analg. 2016; 123(4): 942–9.
24. Inherited thrombophilias in pregnancy. Practice Bulletin No. 138. American College of Obstetricians and Gynecologists. Obstet Gynecol. 2013; 122:706-717.
25. Thromboembolism in pregnancy. Practice Bulletin No. 123. American College of Obstetricians and Gynecologists. Obstet Gynecol. 2011; 118:718-729.
26. Cantwell R, Clutton-Brock T, Cooper G, et al. Saving mothers’ lives: reviewing maternal deaths to make motherhood safer: 2006–2008. The Eighth Report of the Confidential Enquiries into Maternal Deaths in the United Kingdom. BJOG. 2011; 118(suppl 1):1–203.
27. The Royal College of Obstetricians and Gynaecologists. Reducing the risk of venous thromboembolism during pregnancy and the puerperium. Green-top Guideline No. 37a. April 2015.
28. Deniau B, Bouhadjari N, Faitot V, et al. Evaluation of a continuous improvement programme of enhanced recovery after caesarean delivery under neuraxial anaesthesia. Anaesth Crit Care Pain Med. 2016; 35(6):395–9.
29. Nelson G, Dowdy SC, Lasala J, et al. Enhanced recovery after surgery (ERAS®) in gynecologic oncology – Practical considerations for program development. Gynecol Oncol. 2017; 147:617-620.





