REVIEW
Tiolii albuminici - potențiali markeri ai statusului oxidativ în cancer
Albuminic thiols - potential markers of oxidative status in cancer
Abstract
Oxidative stress was associated for a long time with cancer physiopathology and the physiopatology of other diseases. The reactive species generated in excess increase genomic instability that causes damage to DNA and other cellular constituents and can cause mutations. The natural defense system of the organism against oxidative stress is composed by antioxidants that help restore the balance. Thiols, especially albumin, which is the main plasma low molecular antioxidant, are very important. Here, in this paper, we describe the antioxidant properties of albumin and the stages of redox reaction in which it is involved. We also evaluate its potential as a biomarker of oxidative stress mainly in cancer and in other oxidative stress related diseases.Keywords
oxidative stressalbumincancerRezumat
Stresul oxidativ a fost asociat, pentru o lungă perioadă de timp, cu fiziopatologia cancerului și a altor boli. Speciile reactive generate în exces duc la creșterea instabilității genomului care cauzează distrugeri ale ADN-ului și ale altor componente celulare și pot provoca mutații. Sistemul de apărare naturală a organismului împotriva stresului oxidativ este compus din antioxidanți care ajută la restabilirea echilibrului. Tiolii, în special albuminici, care sunt principalul antioxidant cu greutate moleculară mică, sunt foarte importanți. Aici, în această lucrare, vom descrie proprietățile antioxidante ale albuminei și etapele reacției redox în care este implicată. De asemenea, evaluăm potențialul său ca un biomarker al stresului oxidativ în principal în cancer și în alte boli legate de stresul oxidativ.Cuvinte Cheie
stres oxidativalbuminăcancerIntroduction
Oxidative stress is known as the shifting of balance between prooxidans and antioxidants in favor of prooxidants. This condition was observed in a various number of pathophysiological processes associated with human diseases.Antioxidants are the cells mechanism of defense against high concentrations of reactive species (in particular reactive oxygen species). These molecules prevent tumorigenesis and increase life’s duration. Evidence supports the fact that antioxidant supplementation during chemotherapy can reduce the dose-limiting toxicity, therefore they work as protective agents preventing cancer development(1). Among the most effective endogen antioxidants with enzymatic activity are superoxide dismutase, catalase and glutathione peroxidase. Nonenzymatic antioxidants are vitamin C, vitamin E, carotenoids, thiols (glutathione, thioredoxin, lipoic acid, albumin), natural flavonoids, melatonin and other compounds. Antioxidants act in different environments. Some of them are hydrophilic, some are hydrophobic and some are amphiphilic(2).
The antioxidants found in plasma have low molecular weight. Those are thiols, ascorbic acid etc. Thiols have a very low concentration (12-20 µM)(3). Despite the fact that there are more than 3000 of plasma proteins, quite of few of them have antioxidant potential. Among the most numerous 150 plasma proteins, only two of them can act as an antioxidant: human serum albumin and glutathione peroxidase 3(4).
Further, we focus on describing albumin’s antioxidant properties and its implication in cancer pathophysiology.
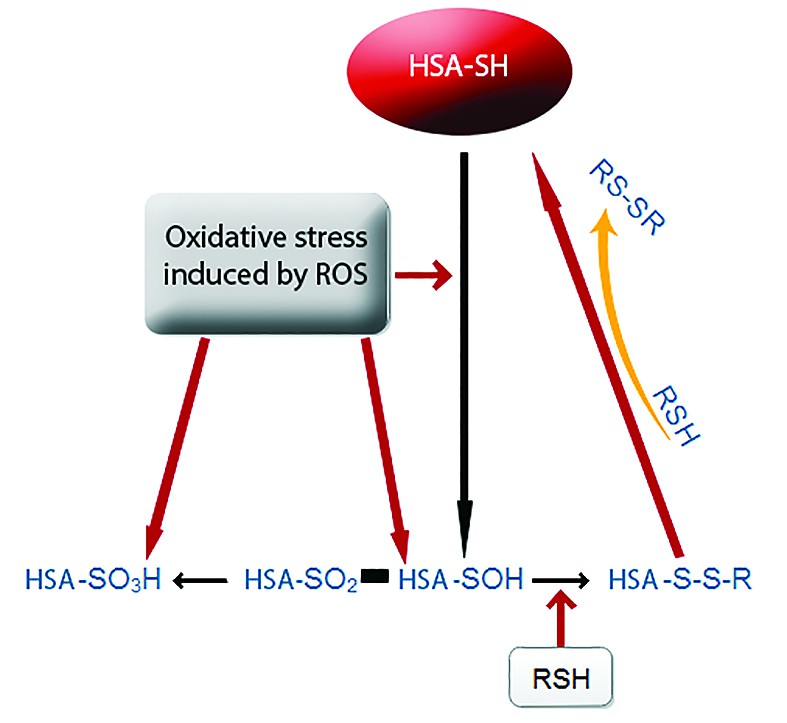
Abbreviations: ASU = human serum albumin, SRO = reactive oxygen species (Taverna et al., 2013)
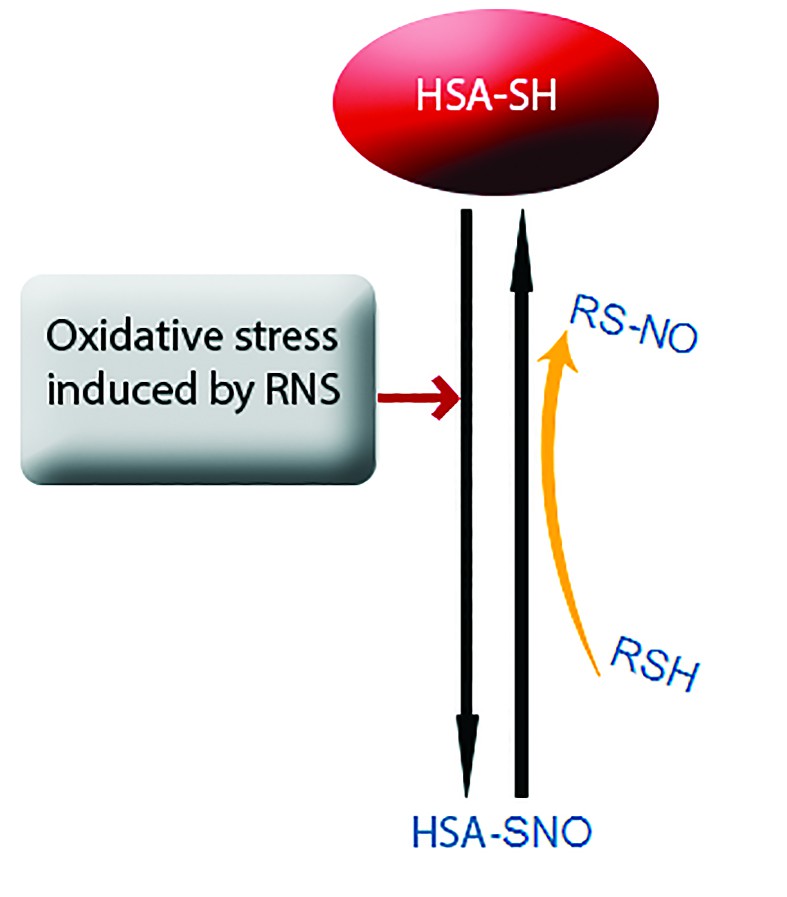
Abbreviations: HSA = human serum albumin, SRN = reactive nitrogen species (Taverna et al., 2013)
Antioxidant properties of thiol albumins
Human serum albumin exerts his antioxidant capacity in plasma. This compartment is meant for gas transport and is permanently exposed to oxidative stress. Serum albumin is the major extracellular source of thiol groups. The reduced sulfhydryl group from thiol albumin structure acts to remove reactive species. It seems that albumin acts as a donor of thiol groups and it regenerates other intravascular proteins by donating electrons. Redox status of the protein is important in covalent binding. For example, the more the methionine residues are exposed, the more it becomes more susceptible to oxidation and can be easily remove more reactive oxygen species (ROS)(5).Only the remaining free cysteine residue from mercaptalbumin’s structure reacts with the reactive species. In vitro studies showed that albumin can adjust certain portion from active pathways involved in mediating inflammatory reaction by binding and removing ROS from neutrophils. It was demonstrated that albumin protects against the oxidizing effect of carbon tetrachloride and uremic toxins, compounds involved in hepatic and renal function. In vivo studies on patients found that albumin has a favorable effect on antioxidant status of plasma thiols, thus reducing the oxidative damage of proteins in patients with acute lung diseases. Human serum albumin plays an important role in vivo, due to its ability to bind and transport substances such as bilirubin based antioxidant and nitric oxide, which are potent antioxidants lipid phase. Also, bilirubin can protect albumin from the oxidants mediated damage(6).
Recent studies have clearly demonstrated that the redox potential of human serum albumin changes when it is oxidated by various oxidants, thus becoming a key biomarker of this proces. After its oxidation, albumin properties are altered: the proteins become more sensitive to trypsin digestion and are degraded faster(7). The antioxidant propriety of albumin results from its ability to bind bilirubin, serum homocysteine and lipids, but more important is the antioxidant role of metal ions from its structure. Human serum albumin binds to bilirubin to a single site (Lys240) and after that it functions as an inhibitor of lipid peroxidation. Homocysteine is a sulfur containing amino acid resulting from the catabolism of methionine residues. Some studies have suggested that the regulation of albumin polyunsaturated fatty acids and sterols may contribute to its antioxidant proprieties, preventing lipid peroxidation(8).
The free Cys34 residue from mercaptabumin structure is capable of removing various ROS and RNS such as H2O2, ONOO-, superoxide or HOCl. The thiol group is exposed to changes in conformation and is oxidized to form a -SOH group as a result of oxidative stress by peroxynitrite of hydrogen peroxide. Oxidized albumin is the central intermediate in redox modulation of reactive species. The end result of the oxidative process depends if the sulfenic acid is further oxidized or if is reduced by obtaining the original thiol albumin. Sulfenic acid can be oxidized to sulfuric acid or sulfonic acid. This process is irreversible. Sulfenic acid can be converted to disulfides by reactions with low molecular weight thiols allowing to return to the intial albumin structure. The involvment of serum albumin in formation of disulfide groups demonstrate its function in regulating the extracelular redox status (Figure 1). Human serum albumin can protect cells againts oxidative stress by modeling cellular levels of reduced glutathione. Indeed, the human serum albumin catabolism may be a source of sulfur amino acids for the cells contributing to the synthesis of other molecules with thiol groups, such as glutathione(8,9).
Following oxidative stress induced by nitric oxide or other nitrozilation agents, mercaptalbumin can be converted into nitrosoalbumin. Albumin can transfer NO cation to thiols with low molecular weight. This reaction allows albumin to return to its original form and makes the protein to be a rich source of NO (Figure 2)(9).
Thiol albumins and pathophysiology of cancer
Oxidative modification of human serum albumin is a potential biomarker of oxidative stress involvement in pathophysiological processes and antioxidant role of mercaptalbumine(3).The determination of concentration of human serum albumin from blood is one of the methods for the evaluation of the nutritional status for patients with cancer, for assessing the severity of the disease and the prognosis. Human serum albumin has been described as an independent marker of prognosis and evolution of therapy in different forms of tumors such as lung cancer, pancreatic, gastric, colorectal, and breast cancer(10).
Malnutrition plays a significant role in the survival of cancer patients. In patients with cancer, there is an inverse correlation between body weight index and synthesis of human serum albumin which supports the existence of a possible compensatory albumin synthesis. After several studies with human serum albumin radiolabelled or conjugated with dye, it was revealed that between 2% and 25% of the applied dose was found in tumor(11).
Therefore, protein oxidation and antioxidant function loss may lead to low concentration of albumin in cancer.
There are advantages and disadvantages of using human serum albumin levels as an indicator of nutritional status of cancer patients. One of the advantages is that the concentration of albumin can be made with low cost, with strong and reproductible methods. Also, albumin can be used as an independent indicator for the need of aggressive nutritional intervention, because its low concentration is associated with a poor prognosis in cancer patients.
Among the major disadvantages, interpretation of albumin level is often difficult due to non-nutritional factors, such as the degree of hydration and the progress of the disease. They can mask the effects of current nutritional deprivation. In addition, the human serum albumin has a long half-life, so the evaluation of nutritional status changes for a short period of time is difficult(12).
Conclusions
Thiol albumins are the main antioxidants from plasma. The Cys34 residue can remove reactive species converting albumin to its oxidized form. Therefore, the oxidative changes in albumin structure can represent a potential biomarker for oxidative stress.As it is well know, oxidative stress is involved in the pathophysiology of cancer and other diseases and is correlated with the severity of the disease and prognostic especially in the early stages. Consequently, by determination of thiol albumins concentration from blood and other biological materials it is possible to evaluate the disease progression and the treatment effectiveness in reducing the oxidative stress. Further studies are necessary to establish if thiol albumins are suitable biomarkers, not only in cancer, but as well in other diseases that have high oxidative stress levels such as rheumatoid arthritis, ischaemia etc. n
Bibliografie
1. Gupta SC, Pandey MK, Tyagi AK, Deb L, Prasad S, Deb L. Oxidative Stress and Cancer: Advances and Challenges. 2016.
2. Valko M, Rhodes CJ, Moncol J, Izakovic M, Mazur M. Free radicals, metals and antioxidants in oxidative stress-induced cancer. Chem Biol Interact. 2006; 160(1):1–40.
3. Turell L, Radi R, Alvarez B. The thiol pool in human plasma: the central contribution of albumin to redox processes. Free Radic Biol Med [Internet]. 2013 Dec [cited 2016 Apr 7]; 65:244–53. Available from: http://www.sciencedirect.com/science/article/pii/S0891584913002815
4. Hortin GL, Sviridov D, Anderson NL. High-abundance polypeptides of the human plasma proteome comprising the top 4 logs of polypeptide abundance. Clin Chem [Internet]. 2008 Oct [cited 2016 Mar 10]; 54(10):1608–16. Available from: http://www.ncbi.nlm.nih.gov/pubmed/18687737
5. Rondeau P, Bourdon E. The glycation of albumin: structural and functional impacts. Biochimie [Internet]. 2011 Apr [cited 2016 May 23];93(4):645–58. Available from: http://www.ncbi.nlm.nih.gov/pubmed/21167901
6. Garcovich M, Zocco MA, Gasbarrini A. Clinical use of albumin in hepatology. Blood Transfus. 2009; 7(4):268–77.
7. Candiano G, Petretto A, Bruschi M, Santucci L, Dimuccio V, Prunotto M, et al. The oxido-redox potential of albumin methodological approach and relevance to human diseases. J Proteomics [Internet]. 2009 Dec 1 [cited 2016 May 23]; 73(2):188–95. Available from: http://www.sciencedirect.com/science/article/pii/S1874391909001730
8. Taverna M, Marie A-L, Mira J-P, Guidet B. Specific antioxidant properties of human serum albumin. Ann Intensive Care [Internet]. 2013;3(1):4. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3577569&tool=pmcentrez&rendertype=abstract
9. Quinlan GJ, Martin GS, Evans TW. Albumin: biochemical properties and therapeutic potential. Hepatology [Internet]. 2005 Jun [cited 2016 May 23]; 41(6):1211–9. Available from: http://www.ncbi.nlm.nih.gov/pubmed/15915465
10. Fanali G, Di Masi A, Trezza V, Marino M, Fasano M, Ascenzi P. Human serum albumin: From bench to bedside. Mol Aspects Med [Internet]. Elsevier Ltd; 2012; 33(3):209–90. Available from: http://dx.doi.org/10.1016/j.mam.2011.12.002
11. Kratz F. Albumin as a drug carrier: design of prodrugs, drug conjugates and nanoparticles. J Control Release [Internet]. 2008 Dec 18 [cited 2016 Feb 22];132(3):171–83. Available from: http://www.ncbi.nlm.nih.gov/pubmed/18582981
12. Gupta D, Lis CG. Pretreatment serum albumin as a predictor of cancer survival: a systematic review of the epidemiological literature. Nutr J [Internet]. 2010 Jan [cited 2016 May 23]; 9:69. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3019132&tool=pmcentrez&rendertype=abstract
Articole din ediția curentă
REVIEW
Radioterapia stereotactică - principii şi aspecte practice
Ciprian Enăchescu, Sena Yossi
Radioterapia stereotactică este o formă de iradiere externă care distribuie într-o şedinţă unică sau în câteva şedinţe doze mari de iradiere în volume-ţintă mici, tratament ce necesită o distribuţie a dozei extrem de precisă, astfel încât doza maximă să fie distribuită în volumul tumoral şi să scadă rapid per...
REVIEW
Prevenirea cancerului prin intermediul unor programe de screening
Dan Ilie-Damboiu
Prevenţia cancerului prin screening este o resursă importantă care ar trebui folosită judicios, în special prin programe de sănătate la nivel naţional şi mai ales la populaţia la risc. Beneficiile sunt evidente în anumite cazuri: prelungirea supravieţuieii la cei cu boală curabilă, scăderea morbidităţii,...Articole din edițiile anterioare
REVIEW
Radioterapia stereotactică - principii şi aspecte practice
Ciprian Enăchescu, Sena Yossi
Radioterapia stereotactică este o formă de iradiere externă care distribuie într-o şedinţă unică sau în câteva şedinţe doze mari de iradiere în volume-ţintă mici, tratament ce necesită o distribuţie a dozei extrem de precisă, astfel încât doza maximă să fie distribuită în volumul tumoral şi să scadă rapid per...
REVIEW
Prevenirea cancerului prin intermediul unor programe de screening
Dan Ilie-Damboiu
Prevenţia cancerului prin screening este o resursă importantă care ar trebui folosită judicios, în special prin programe de sănătate la nivel naţional şi mai ales la populaţia la risc. Beneficiile sunt evidente în anumite cazuri: prelungirea supravieţuieii la cei cu boală curabilă, scăderea morbidităţii,...
REVIEW
