The most common etiology of glottal insufficiency is vocal fold paralysis usually from damage to the nerve supplying the laryngeal muscles. Glottal closure is an important part of the mechanism that protects the airway during the normal swallow. Vocal fold paralysis or atrophy disrupts the normal closure of glottis during swallowing, thus placing patient at increased risk for aspiration. Treatment of glottal insufficiency can be classified as surgical, provided by the otolaryngologist, or non-surgical. Non-surgical treatment includes voice therapy which may incorporate resonant voice therapy, counseling and massaging techniques. Swallowing rehabilitation, another form of non-surgical intervention, consists of direct therapy (swallowing maneuvers, head postures, or changes in diet consistencies) or indirect therapy (swallowing exercises), or combined.
Insuficiența glotică și riscurile asociate cu disfagia
Glottal insufficiency and the associated risk of dysphagia
First published: 27 noiembrie 2016
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/Orl.33.4.2016.157
Abstract
Rezumat
Cea mea frecventă etiologie a insuficienței glotale este paralizia corzilor vocale, cauzată de lezarea nervului care suplinește mușchii laringeali. Închiderea glotică este o parte importantă a mecanismului care protejează căile respiratorii superioare în timpul procesului de deglutiție. Atrofia sau paralizia corzilor vocale afectează închiderea glotică corectă în timpul procesului de deglutiție, ceea ce expune pacientul la un risc ridicat de aspirație. Tratamentul insuficienței glotice poate fi chirurgical, efectuat de otorinolaringog, sau non-chirurgical. Tratamentul non-chirurgical include terapia vocii, respectiv terapia vocii de rezonanță, diverse tehnici de consiliere și masaj. O altă formă de intervenție non-invazivă este reabilitarea procesului de deglutiție în cazul disfagiei. Aceasta constă în terapia directă (manevre de deglutiție, posturi ale capului ori schimbări în consistența dietei), terapie indirectă (exerciții de deglutiție) sau acestea combinate.
The most essential function of the larynx is as a primary protector of the airway, preventing aspiration of one’s own secretions, gastric juices, and with intake of food and liquids. Secondarily, the voice also allows for the production of sound. In this article we will briefly discuss laryngeal dysfunction as it pertains of glottal insufficiency and its implications in developing swallowing disorders, in addition to voice problems. We may broadly describe the vocal mechanism as providing both airway safety and phonation with use of vocal fold closure. The correct function of the larynx can be largely affected by neurogenic voice pathologies or other anatomical abnormalities. In the scope of this paper, we will focus specifically on neurological dysfunction, such as paralysis, paresis, and vocal fold atrophy.
Normal swallow
A safe and efficient swallow contains in a series of events that begin in the oral cavity and ends with the closure of the glottis and laryngeal vestibule, and opening of upper esophageal sphincter. Thus, swallowing has been described as having three discrete phases that interact with each other: oral stage, pharyngeal stage, and esophageal stage (Logemann, Kahrilas)(1). During the oral stage, a voluntary act, the food or liquid bolus is formed into a cohesive ball and transported posteriorly for swallowing. The pharyngeal stage of swallowing begins as the head of the bolus reaches the anterior tonsillar arches. At this point, the swallow becomes involuntary, and a very complex sequence of coordinated events occur resulting in both passage of the bolus from mouth to stomach and airway protection.
Glottal closure is a vital step in the pharyngeal phase of swallowing, and timing is the key for airway protection. Using radiographic examination, several studies have observed a sequence of laryngeal activity for airway protection: closure of the laryngeal vestibule, glottic closure, and inversion of the epiglottis. A closer look via computed tomography by Inamoto and colleagues(2) depicted the following sequence: (1) The hyoid elevation and forward movement was the first event; (2) followed by pharyngoesophageal (PES) opening. Then (3) true vocal cord closure occurs simultaneously with closure of the laryngeal vestibule at the arytenoid to epiglottis base, and lastly (4) epiglottic inversion occurs after the hyoid is maximally displaced. Based on this study, temporal variations were noticed between normal individuals. According to Inamoto, glottal closure takes approximately 0.4 seconds. Thus, there is little time for errors.
During this succession of events, a series of pressure gradients are created. Jeri Logeman(3) described the swallowing mechanism as a series of valves and pipes that widen and close to create positive pressure for safe and adequate bolus transfer into the stomach. The valves within the swallowing mechanism are the lips, soft palate, glottis, upper and lower esophageal sphincters. The fine interplay between these structures leads to a successful swallow. A mechanical break/change in any of these valves or pipes can lead to a change in pressure which may translate into swallowing impairments.
The purpose of this article is to focus on the glottic valve. Glottal insufficiency refers to incomplete closure of the vocal folds during speech, swallow or cough (functional or biological functions). When there is glottal insufficiency, pressure may be lost, which may lead to dysphagia with or without penetration or aspiration.
Normal phonation
The voice is produced as a byproduct of airflow (power), vocal fold vibration (source), and subsequent resonance of the resultant acoustic energy by the vocal tract (filter). The respiratory system is responsible for generating enough airflow to engage vocal fold vibration, and must be sustained over the course of a vocalization. Vocal fold vibration is a complex interplay between the pressure differential above and below the vocal folds, the biodynamic quality of the vocal folds, and finally the level of closure that is achieved(4). The flexible, myoelastic nature of the tissue and muscle allows for sequential, biphasic opening and closing of the vocal folds. This generates a differential between positive subglottal pressure and negative supraglottal pressure that is crucial to the propagation and translation of aerodynamic flow into acoustic energy. Pitch is governed by the number of cycles per second that the vocal folds vibrate. This is controlled by the length, tension, and stiffness of the thyroarytenoid muscles. Volume is controlled both by subglottic pressure and by the level of vocal fold closure (further defined by the degree and speed of closure). The length and shape of the spaces above the vocal folds, which includes the pharynx, nasopharynx and oral cavity, define the resultant resonance of the voice. The vocal tract is often described as a “filter” to the sound source as it functions to enhance certain frequencies, while dampening others. The beauty of the human voice is that we can change the resonance of voice; engaging a muffled “dark” sound or a bright sharp sound simply by changing the shape of the vocal tract. Thus, the three subsystems of phonation, also referred to as the “power source filter”, must be in exquisite balance with one another in a dance of aerodynamic flow, myoelastic vibration of the vocal folds, and enhancement of the acoustic signal.
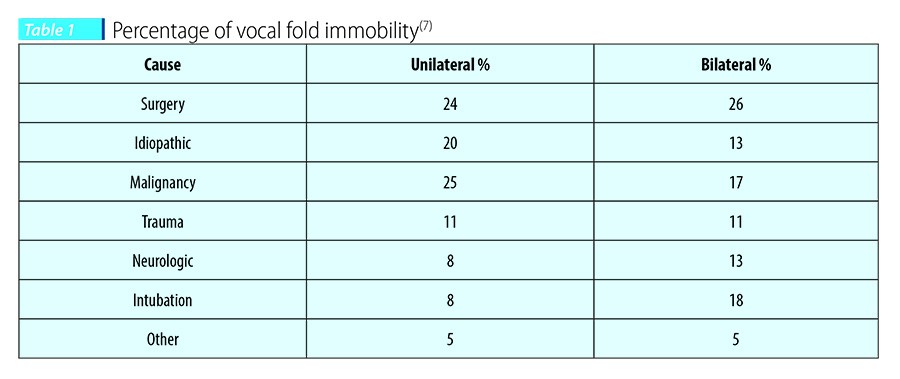
Etiology and incidence of dysphagia due to glottal insufficiency
Vocal fold immobility or hypomobility is by far the most common etiology and can affect one or both vocal folds. The overall severity is exacerbated if there is more proximal involvement of the vagus nerve, affecting muscles innervated by both the recurrent and superior laryngeal nerves.
Paralysis of the vocal cord(s) may be due to traumatic injury, nerve compression as a complication of surgery (e.g., anterior cervical spine fusion, thyroidectomy, cardiac surgery), and compression of the vagus nerve secondary to tumor formation. Idiopathic cases represent approximately 30-35% of the cases(5,6), although more recent estimates are closer to 15%, likely associated with improved diagnostics. Patients with idiopathic onset of vocal paralysis often report symptoms occurring after a viral infection.
Paralysis thus can be iatrogenic (thyroidectomy, mediastinal surgery, cervical), idiopathic (viral illness), due to neoplasia (skull-base tumors, vagal schwannomas), or trauma (arytenoid subluxation, neck trauma). In addition, more systemic neurodegenerative disorders such as amyotrophic lateral sclerosis, neuronitis, Parkinson’s disease, or an autoimmune condition (e.g., systemic lupus erythematosus) may also influence muscle tone and function, resulting in poor glottic closure.
A series of studies showed a high incidence of dysphagia in patients with glottal insufficiency. A retrospective study by Tabaee et al.(8) on 81 patients with unilateral vocal cord immobility showed an aspiration rate of 35% with thin liquids and 18% rate with purée. Their study shows that besides glottal insufficiency, a majority of patients showed laryngeal edema (90%), decreased laryngeal sensation (83%) and difficulty managing secretions (60%), which further may have added to the degree of dysphagia. In two consecutive studies of patients with dysphagia in a large hospital (a total of 4,102 dysphagic patients), Leder et al.(9,10) looked at the incidence of vocal fold immobility (VFI) and its relationship to aspiration and pharyngeal dysphagia. The results showed that the incidence of VFI in all patients with dysphagia in this large sample was 4.7%. Aspiration occurred in 24.5% of the patients with VFI. Thus, a patient with VFI had 2.5 times the odds of aspirating as someone without VFI. Additionally, the study showed that those patients with VFI aspirated liquid more often than purée.
Symptoms
The severity of symptoms experienced by the patient is directly related to the degree of glottal insufficiency, governed by the position of the paralyzed vocal fold or the level of vocal fold atrophy. The most commonly reported symptoms include:
- Weak and breathy voice.
- Decreased volume or projection.
- Vocal fatigue (especially late in the day).
- Strained voice (overcompensation for poor vocal closure).
- Shortness of breath (sensation of breathlessness with speech).
- Coughing while eating (suggesting a possible aspiration of liquids).
- “Wet” vocal quality.
- Odynophagia (pain with swallowing especially with prolong use).
- Odynophonia (pain after speaking for long periods of time).
When a patient complains of coughing while eating or drinking, this raises the suspicion of possible aspiration. The reflexive cough is a defense mechanism and in normal people it is immediately triggered by an aspirant, thus protecting the lungs. Coughing while eating or drinking is probably the most common symptom of aspiration. A “wet” vocal quality, particularly during meals, may also indicate reduced airway protection. This symptom suggests that, during deglutition, food or liquid is entering the laryngeal vestibule and possibly resting on the vocal cords. Therefore, a patient might exhibit a “wet” or “gurgly” vocal quality during phonation. Fatigue with meals and a weak or breathy voice may also alert the swallowing specialist on the potential of reduced upper airway protection.
As mentioned before, an atrophic, breathy, and high-pitched voice, with intermittent diplophonia, are the hallmarks of glottal insufficiency. Beware, however, that sound quality and strength can be influenced by muscle strain to compensate the poor vocal fold closure, and may not be a true representation of the underlying glottal incompetence. The patient may also complain of sensations of shortness of breath while speaking, vocal fatigue with prolonged conversation, muscle strain or throat pain, and difficulty being heard, especially over background noise.
Voice assessment
The need for a voice assessment typically depends on the patient’s level of vocal function regarding control of volume, pitch, and/or vocal stamina, and his or her resultant decrease in quality of life. Assessment tools available to the clinician include visualization of the vocal folds, acoustic and aerodynamic measures, and physiological measures. The gold standard for visualization of the vocal fold closure is stroboscopic videolaryngoscopy. Although a basic laryngoscopic examination can reveal gross vocal fold movement, the relevant degree of vocal fold closure during phonation is lost with the imaging under stroboscopic lighting. The flashing light of the strobe is timed to be just off the fundamental frequency of the voice. This superficially slows down vocal fold vibration by capturing sequential images at different points of the vibratory phase. Specific laryngoscopic features to assess include: vocal fold bowing, reduced vocal fold movement, asymmetric mucosal waveforms, and supraglottic constriction(11,12). Various acoustic and aerodynamic parameters are supported in the literature as well and can help the clinician to document the level of severity. Spectral analysis, a graphical representation of vocal frequency and strength as a function of time, as well as cepstral analysis are considered the most reliable in acoustic measures(13). Aerodynamic measures of subglottic pressure (measured as Psub) and glottal flow (measured as mL/sec) are also helpful in quantifying the degree of glottic function. Finally, physiologic measures of frequency range (Hz), intensity range (dB), and maximal phonation time may reveal limitations consistent with poor vocal strength. In addition to objective measures, a subjective evaluation including a perceptual evaluation and patient self-assessment are helpful in documenting the severity of the voice problem as it relates to the patient’s quality of life. Hirano’s(14) standardized approach to perceptual evaluation of the voice breaks down dysphonia into overall grade, roughness, breathiness, asthenia and strain. Self-assessment surveys are used to gauge the level of perceived handicap. Many such measures include functional, social, and emotional aspects of voicing that can impact a person’s quality of life. Interestingly enough, many times the level of perceived difficulty is not a reflection of the actual level of vocal function. Nevertheless, the “big picture” gained from visual analysis, acoustic and aerodynamic measures, as well as self-assessment and perceptual evaluations can be critical in giving the clinician or physician a clearer understanding of overall vocal function and subsequent medical and behavioral management.
Voice management
The primary goal of voice management is to allow for the best possible voice in the presence of motion impairment of the voice. Factors to be considered when referring a patient for voice therapy include the size of the glottal gap and the level of compensatory strain. Many cases of unilateral paresis/paralysis or bilateral vocal atrophy can be well managed with vocal fold augmentation via injection medialization procedures, thyroplasty, or autologous fat augmentation. Yung et al. (2011)(15) found that patients undergoing temporary injection medialization procedures were statistically significantly less likely to undergo permanent laryngoplasty procedures.
Behavioral management (voice therapy) can be helpful either as the primary form of treatment, or as an adjunct to surgical management. If voice therapy is used as the sole treatment, the vocal folds must have at least touch closure in order for the patient to benefit(16). The focus of treatment is to decrease unneeded muscle tension and improve the patient’s coordination of respiration, phonation, and resonance. In addition, the patient will also benefit from a basic understanding of anatomy & physiology, and the way he breathes, voice quality, and swallowing can change with the diagnosis. Although there is no direct indication that voice therapy directly influences the strength of vocal fold closure, it is believed that patients make gains by maximizing the overall function of the vocal mechanism. Specific vocal exercises are geared towards maximizing vocal output while minimizing effort, all the while potentially building vocal strength, balance, and stamina(17). More specific activities and tasks in treatment might include counseling and training the patient to accept a weaker voice by using less volume in speaking while reducing utterance length and/or increasing pause time. In addition, the patient can be taught to use massage of the extrinsic laryngeal musculature, which can also be empowering when patients complain of physical fatigue and pain. Resonant voice training is also helpful in enhancing the patient’s sense of vocal strength and improving efficiency of airflow. The theoretical concept of resonant voice training is improving upon the function of the source of vocalizations (e.g., the vocal fold closure patterns) by focusing on vibratory sensations produced by the resonant system. This can most easily be demonstrated by feeling the sensation of vibration during the production of a hum. The Lessac-Madsen Resonant Voice training protocol developed by Katherine Verdolini(18) also allows for a systematic building of kinesthetic awareness as well as auditory feedback. Vocal function exercises, an organized system of exercises designed to “bring the vocal folds to the gym”, has also been described in literature as having significant effects on neurogenic voice disorders. Based on a series of maximally sustained tones and maximal pitch glides, the patient learns to train stamina for greater efficiency of breath with reduced laryngeal strain. Pushing and pulling exercises(19,20) or those involving hard glottal attacks should be avoided unless the practitioner is vigilant about avoiding excessive vocal strain or hyperfunction.
Swallow assessment
Assessment of swallowing impairments associated with voice disorders includes: (1) subjective reports of dysphagia either in clinical interviews, or by completing dysphagia questionnaires, (2) clinical evaluation of swallowing by physician or swallowing specialist in office, and (3) objective evaluation of swallowing via instrumental means.
A series of questionnaires have been developed to address dysphagia. Most used in the United States are EAT-10(21), SWAL-QOL(22), and the M.D. Anderson Dysphagia Inventory(23). EAT-10, developed by dr. Peter Belfasky, contains 10 simple questions easily administered. A score higher than 3 indicates a degree of swallowing impairment. These outcomes will prompt an evaluation by a dysphagia specialist (speech-language pathologist or physician). SWAL-QOL is an outcomes tool for measuring the impact of dysphagia on quality of life from patient’s perspective. The tool has documented validity and reliability. It has 44 items that address desire for eating, dysphagia symptom frequency, mental health, social concerns related to swallowing problems, food selection, fear related to eating, and the burden of dysphagia. M.D. Anderson Dysphagia Inventory is a self-administered questionnaire designed specifically to assess dysphagia and quality of life of patients with head and neck cancer. The questionnaires are not only used to obtain a baseline of patient’s self-reported symptoms of swallowing deficits, but also to evaluate progress if patient undergoes swallow rehabilitation.
The goal of a clinical evaluation of swallowing is to determine whether dysphagia is present, to evaluate the severity, to determine the safest oral diet and whether patient requires further instrumental evaluation. The components of a clinical assessment are: (1) history regarding the swallowing problem, (2) evaluation of the anatomy and functionality of the swallowing mechanism (oral motor exam, cranial nerve exam). Finally, (3) the oral feeding test is performed, which evaluates the oral and pharyngeal phases of swallowing and signs or symptoms of aspiration (e.g., coughing, throat clearing, changes in vocal quality, respiratory distress etc.) with different liquid and food consistencies.
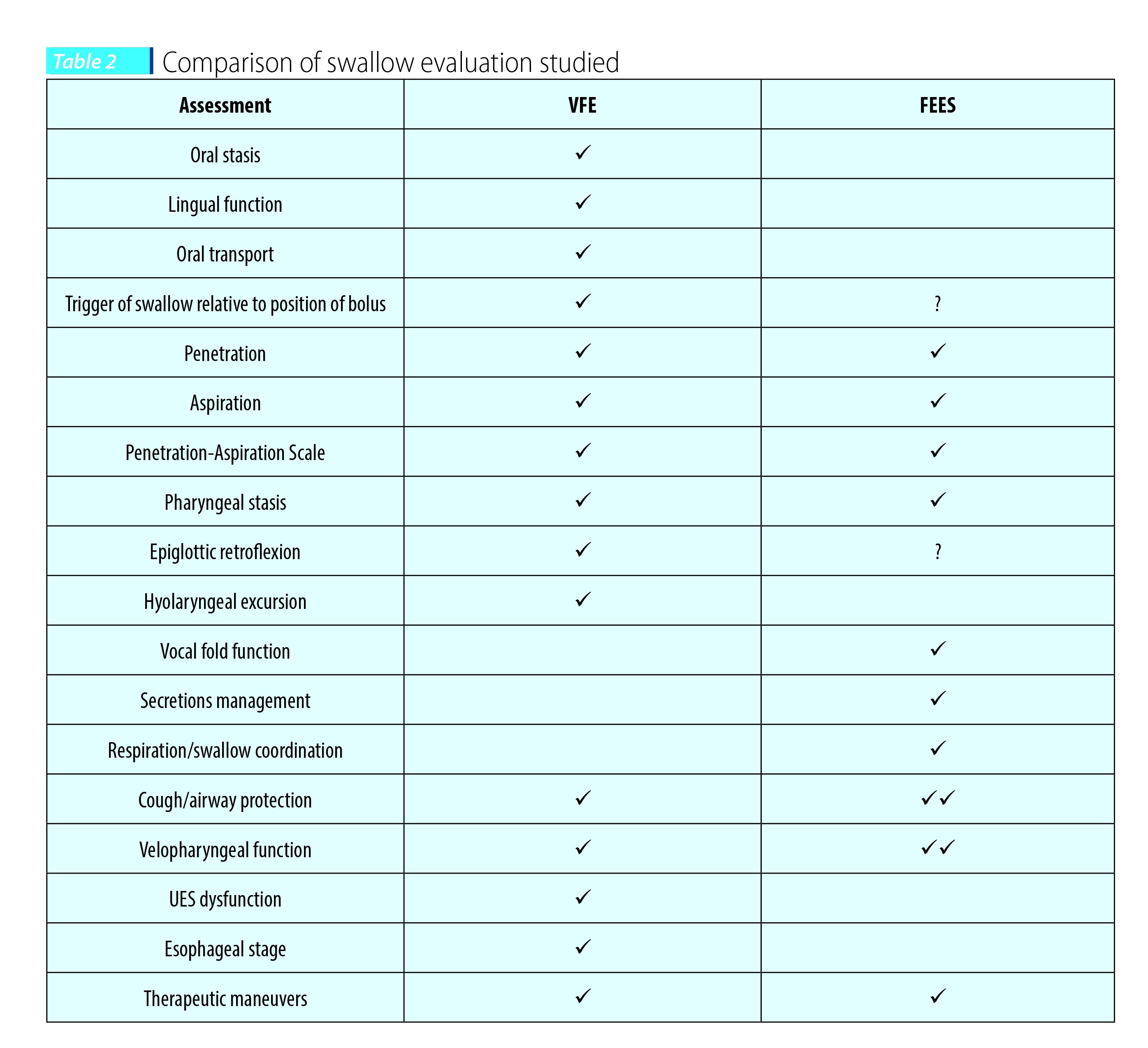
Instrumental tools in evaluation of swallowing mechanism include the Fiberoptic Endoscopic Evaluation of Swallowing (FEES) and Videofluoroscopic Swallow Study (VFSS). They are considered the gold standard tools in the evaluation of swallowing mechanism. During the FEES examination, a flexible endoscope is passed through the nose into the hypopharynx with direct visualization of the larynx and vocal cords from above. The FEES is a portable instrument and assesses the pharyngeal stage of the swallow with different food or liquid viscosities as indicated (thin liquids, thick liquids, purée and solids). It is suitable for the evaluation of swallowing at the patient’s bedside, it is quickly administered and results are available at the end of the study. A diet recommendation is then provided. The VFSS is performed in Radiology and assesses oral, pharyngeal and esophageal stages of swallowing with different food consistencies. Just as with FEES, the results are available at the completion of the exam with oral diet recommendations. Table 2 (from Glossa et al., 2014(24)) gives a quick comparison between these two swallow studies.
A third instrumental tool is the high-resolution manometry (HRM), which is used to detect esophageal dysphagia. During the HRM, a thin tube is passed through the nose into the stomach and measures pressures and timing in the pharynx and esophagus during swallowing. However, this type of exam is relatively new and it is not readily available to all speech pathologists.
Management of dysphagia
The treatment of patients with glottal insufficiency is based on the severity of the patient’s symptoms, the nature of the dysphagia, and the effectiveness of treatment options. Severe cases are often referred to otolaryngology for surgical intervention (e.g., injection laryngoplasty, medialization thyroplasty). Swallowing therapy is in general performed by a speech-language pathologist (SLP). The methods used in swallow rehabilitation include direct and indirect therapy.
Direct therapy refers to the use of head postures and maneuvers (e.g., chin-tuck, head turn, effortful swallows etc.) in the scope of changing physiology of swallow and improving airway entrance protection and pharyngeal clearance of residue. The ultimate goal is for the patient to continue on a least restrictive diet. This means that, during an evaluation, the SLP and patient find what head postures or maneuvers would work to eliminate aspiration if this is present, while ensuring that the patient’s diet has least restrictions. If these head postures fail, a modified diet may be recommended for safe swallowing (e.g., mashed soft foods, thick liquids etc.).
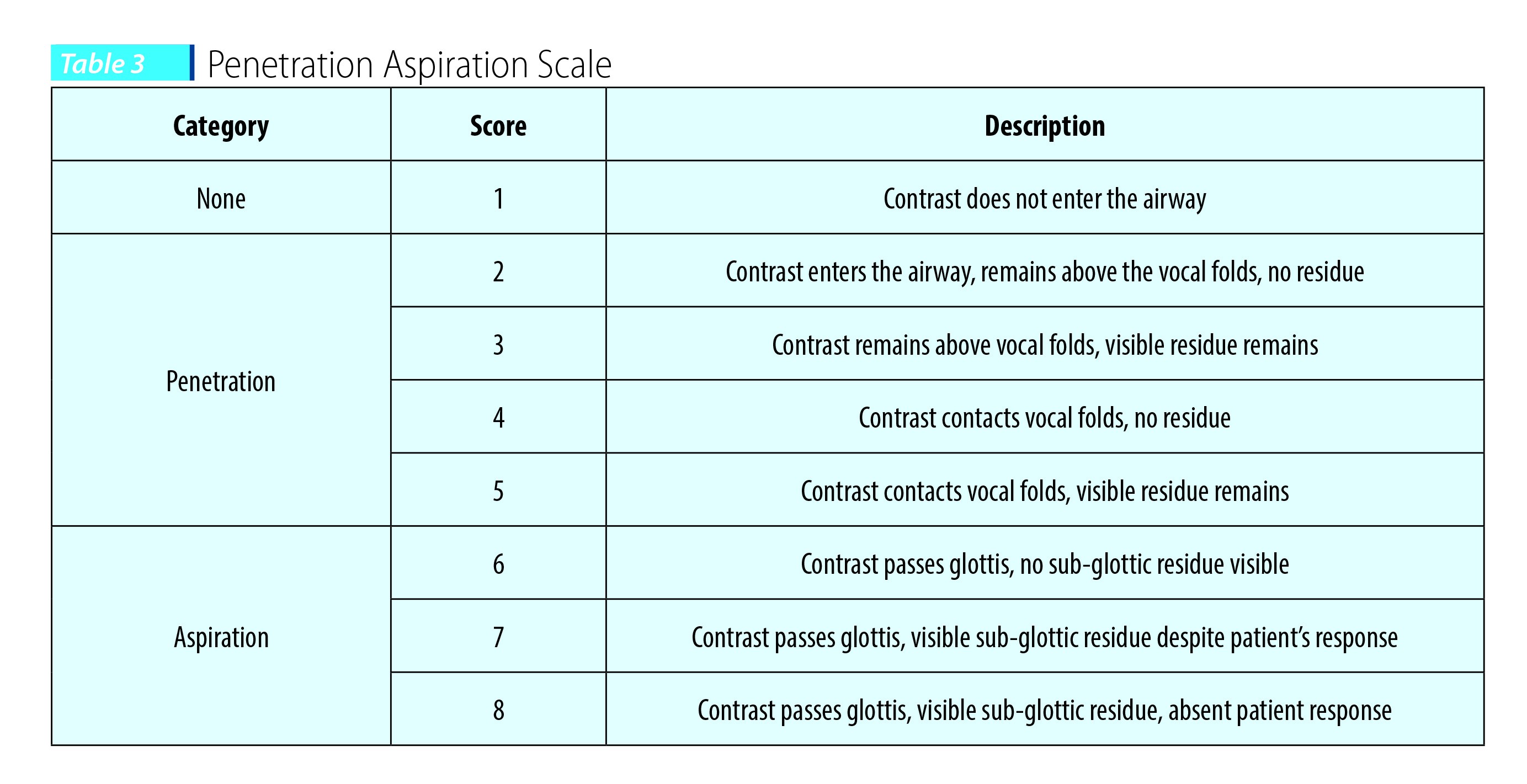
Bhattacharyya and colleagues (2003)(25) studied the effect of bolus consistency on dysphagia in unilateral vocal cord paralysis (UVFP). Thicker viscosity was found to be safer in patients with UVFP. Fifty-five patients with UVFP were studied videofluoroscopically using the Penetration Aspiration Scale (PA scale). The PA scale uses an 8-point scale to describe penetration and aspiration events. Scores are determined primarily by the depth to which material passes in the airway and by whether or not material entering the airway is expelled. Thus, a score of 1 indicates no aspiration and 8 shows silent aspiration (Table 3). The results of this study showed that the liquid bolus was penetrated in 19 (34.5%) patients and aspirated in 11 (20%) patients. However, with a paste consistency, while 12 patients penetrated (21.8%), none of the patients aspirated.
Indirect swallow therapy includes specific swallow exercises with focus on improving and strengthening the musculature involved in swallowing and timing of swallow. These therapy modalities are chosen by the SLP with the patient based on a thorough evaluation.
Biofeedback therapy has gained more attention recently (but has yet to be validated with efficacy data) and it is more often incorporated into indirect swallow therapy. Surface EMG uses electrodes placed submentally. During a swallow, the patient can view on a screen the efficiency of the swallow performed and whether it reached a goal set for that session. Additionally, FEES has been used successfully as a source of biofeedback whereby patient works directly to improve swallow function by the way of direct feedback, specifically in clearance of hypopharyngeal residue.
Special populations
Other modalities have been developed for special populations with glottal insufficiency, such as patients with Parkinson’s disease, who have indirectly improved swallow function and safe oral intake.
Parkinson’s disease
Lee Silverman Voice Treatment (LSVT) focuses on increasing vocal loudness and is delivered by a speech pathologist in sixteen one-hour sessions spread over four weeks. The treatment has shown to improve respiratory, laryngeal and articulatory function to maximize speech intelligibility. Direct voice rehabilitation in this population also showed improvement in swallow function. Sharkawi et al.(26) found that following LSVT, modified barium swallow study showed improvement in both oral and pharyngeal stages of swallow. More specifically, improvement was seen in temporal measures as reduced. After LSVT, there was an overall 51% reduction in the number of swallowing motility disorders. Some temporal measures of swallowing were also significantly reduced as was the approximate amount of oral residue after 3 ml and 5 ml liquid swallows. Conclusions: LSVT® seemingly improved neuromuscular control of the entire upper aerodigestive tract, improving oral tongue and tongue base function during the oral and pharyngeal phases of swallowing, as well as improving vocal intensity.
Another therapy modality, the Expiratory Muscle Strengthening Training (EMST) - Figure 1, also showed promising results in improving overall swallow function and decrease risk for aspiration.
The EMST device is a one-way, spring-loaded valve to exercise the expiratory and submental muscles. These are muscles important for breathing out forcefully, coughing, and swallowing. The treatment lasts five weeks, with participants completing 25 breaths a day (5 sets of 5 repetitions), five days per week. During the five weeks, the device is reset as the participant gets stronger.
A study conducted by Troche et al.(27), using the EMST150® with mild to moderate impaired Parkinson’s patients, showed functional and physiological improvement in swallowing. The results showed a significant reduction in the Penetration-Aspiration (PA) scores measured pre- and post-treatment with Videofluoroscopy. Eleven patients (33%) had improved PA scores following EMST. Additionally, the study showed improvement in the duration of swallow events post-treatment: onset of bolus transit, upper esophageal sphincter (UES) opening and UES closure, laryngeal opening and closure. This resulted in an overall more efficient and safer swallow.
Conclusion
Swallowing is a rapid and complex process that involves well-coordinated movements of the oral cavity, larynx, pharynx and esophagus while moving food from the mouth into the esophagus. In this fast deglutition process, laryngeal closure is the key in protecting the airways. Failure of laryngeal closure at the level of vocal cords may lead to aspiration. A thorough evaluation of voice and swallowing, both clinically and instrumentally, is critical to treating glottal insufficiency and dysphagia. The role of the speech-language pathologist is to work with the patient in eliminating risks for aspiration and finding the least restrictive oral diet for an enjoyable mea
Bibliografie
2. Inamoto, Y., et al., Evaluation of swallowing using 320-detector-row multislice CT. Part II: kinematic analysis of laryngeal closure during normal swallowing. Dysphagia, 2011. 26(3): p. 209-17.
3. Logemann, J.A., Dysphagia: evaluation and treatment. Folia Phoniatr Logop, 1995. 47(3): p. 140-64.
4. Titze, I.V., Katherine, Vocology: The Science and Practice of Voice Habilitation. 2012: National Center for Voice and Speech.
5. Myssiorek, D., Recurrent laryngeal nerve paralysis: anatomy and etiology. Otolaryngol Clin North Am, 2004. 37(1): p. 25-44, v.
6. Stemple, J.G., Leslie; Gerdeman-Klaben, Bernice, Clinical Voice Pathology: Theory and Management. 3rd ed. 2000: Singular.
7. Benninger, M.S., et al., Evaluation and treatment of the unilateral paralyzed vocal fold. Otolaryngol Head Neck Surg, 1994. 111(4): p. 497-508.
8. Tabaee, A., et al., Flexible endoscopic evaluation of swallowing with sensory testing in patients with unilateral vocal fold immobility: incidence and pathophysiology of aspiration. Laryngoscope, 2005. 115(4): p. 565-9.
9. Leder, S.B. and D.A. Ross, Incidence of vocal fold immobility in patients with dysphagia. Dysphagia, 2005. 20(2): p. 163-7; discussion 168-9.
10. Leder, S.B., et al., Vocal fold immobility and aspiration status: a direct replication study. Dysphagia, 2012. 27(2): p. 265-70.
11. Woo, P., et al., Analysis of laryngoscopic features in patients with unilateral vocal fold paresis. Laryngoscope, 2016. 126(8): p. 1831-6.
12. Woo, P., Quantification of videostrobolaryngoscopic findings - measurements of the normal glottal cycle. Laryngoscope, 1996. 106(3 Pt 2 Suppl 79): p. 1-27.
13. Heman-Ackah, Y.D., et al., Quantifying the cepstral peak prominence, a measure of dysphonia. J Voice, 2014. 28(6): p. 783-8.
14. Hirano, M., Clinical examination of voice. Vol. 5. 1981: Springer.
15. Yung, K.C., I. Likhterov, and M.S. Courey, Effect of temporary vocal fold injection medialization on the rate of permanent medialization laryngoplasty in unilateral vocal fold paralysis patients. Laryngoscope, 2011. 121(10): p. 2191-4.
16. Schneider, S.L., Behavioral Management of Unilateral Vocal Fold Paralysis and Paresis. SIG 3 Perspectives on Voice and Voice Disorders, 2012. 22(3): p. 112-120.
17. Stemple, J.C., et al., Efficacy of vocal function exercises as a method of improving voice production. J Voice, 1994. 8(3): p. 271-8.
18. Verdolini, K., et al., Laryngeal adduction in resonant voice. J Voice, 1998. 12(3): p. 315-27.
19. Miller, S., Voice therapy for vocal fold paralysis. Otolaryngol Clin North Am, 2004. 37(1): p. 105-19.
20. Schindler, A., et al., Vocal improvement after voice therapy in unilateral vocal fold paralysis. J Voice, 2008. 22(1): p. 113-8.
21. Belafsky, P.C., et al., Validity and reliability of the Eating Assessment Tool (EAT-10). Ann Otol Rhinol Laryngol, 2008. 117(12): p. 919-24.
22. McHorney, C.A., et al., The SWAL-QOL and SWAL-CARE outcomes tool for oropharyngeal dysphagia in adults: III. Documentation of reliability and validity. Dysphagia, 2002. 17(2): p. 97-114.
23. Chen, A.Y., et al., The development and validation of a dysphagia-specific quality-of-life questionnaire for patients with head and neck cancer: the M. D. Anderson dysphagia inventory. Arch Otolaryngol Head Neck Surg, 2001. 127(7): p. 870-6.
24. Gossa M., S.D., Ciucci M., and Smith P. , Clinical Swallowing Evaluation Across the Lifespan: The Science & the Art, in American Speech and Hearing Association. 2014: Orlando, Florida.
25. Bhattacharyya, N., T. Kotz, and J. Shapiro, The effect of bolus consistency on dysphagia in unilateral vocal cord paralysis. Otolaryngol Head Neck Surg, 2003. 129(6): p. 632-6.
26. El Sharkawi, A., et al., Swallowing and voice effects of Lee Silverman Voice Treatment (LSVT): a pilot study. J Neurol Neurosurg Psychiatry, 2002. 72(1): p. 31-6.
27. Troche, M.S., et al., Aspiration and swallowing in Parkinson disease and rehabilitation with EMST: a randomized trial. Neurology, 2010. 75(21): p. 1912-9.