Medicamentele şi toxicitatea hepatică la copil
Drugs and liver injury in children
Abstract
Drug toxicity represents a worldwide issue, being an important cause of morbidity and mortality in children and adolescents. It is one of the most frequent causes of acute liver injury in children, sometimes with evolution to acute liver failure (ALF) or death. The liver damage produced by toxic causes represents 20-25% of all cases of ALF in children and teenagers. The most common drug used that has liver toxicity is acetaminophen, with easy access, being released without medical prescription. Fortunately, the antidote used in acetaminophen intoxication (acetylcysteine) makes mortality to remain low among these cases. Other drugs that cause toxic hepatitis are albendazole, valproic acid, anaesthetics and antituberculosis agents. In paediatrics, the early recognition of the possible cause that led to liver injury may be the key for the correct management and for a favourable outcome. If there is a toxic cause, the emergency referral to a specialized centre is vital in these patients.Keywords
drugstoxichepatitisacute liver failure (ALF)acetaminophenchildrenteenagersRezumat
Toxicitatea secundară ingestiei de medicamente reprezintă o problemă în întreaga lume, fiind o cauză importantă de morbiditate şi mortalitate la copii şi adolescenţi. Este una dintre cele mai frecvente cauze de injurie hepatică acută la copil, cu potenţial de evoluţie către insuficienţă hepatică acută (IHA) sau deces. Toxicele reprezintă 20-25% din cazurile de IHA la copii şi adolescenţi. Paracetamolul este medicamentul implicat cel mai frecvent în hepatitele de cauză toxică din cauza faptului că în ţara noastră se eliberează fără prescripţie medicală, fiind uşor accesibil în special adolescenţilor cu gânduri suicidare. Din fericire, utilizarea N-acetilcisteinei ca antidot determină o mortalitate redusă în aceste cazuri. Alte medicamente implicate în declanşarea fenomenelor de hepatotoxicitate sunt albendazolul, acidul valproic, anestezicele, medicaţia antituberculostatică etc. Recunoaşterea timpurie de către medicul pediatru a unei posibile etiologii hepatotoxice poate reprezenta cheia către un diagnostic rapid, un tratament precoce şi un prognostic favorabil. Etiologia toxică a hepatitelor acute impune transferul de urgenţă al pacientului către un centru specializat, acest lucru având importanţă vitală, cu predilecţie în cazurile severe care evoluează cu IHA.Cuvinte Cheie
medicamentetoxicehepatităinsuficienţă hepatică acutăparacetamolcopiiadolescenţiIntroduction
Hepatotoxicity is defined as an increase of the serum levels of aminotransferases or conjugated bilirubin or cholestasis enzymes secondary to the action of an agent with hepatotoxic potential(1,2). Now, the term used for hepatotoxicity determined by drugs is drug-induced liver injury (DILI). Almost all the drugs can cause DILI, but most of the times the phenomena of hepatotoxicity are mild. Only in several cases, there is an evolution with severe liver injury. In developing countries, DILI represents the first cause of severe hepatitis evolving with acute liver failure (ALF)(3,4).
Drugs can determine hepatotoxicity by two mechanisms: toxicity dependent on the doses (acetaminophen, halothane) and idiosyncratic reactions (antibiotics, nonsteroidal anti-inflammatory drugs). They determine cellular lesions through the alteration of the hepatocyte’s membrane or the biliary canaliculus, the activation of the T lymphocytes and tissular necrosis factor alpha (TNF-alpha), the alteration of the mitochondrial function or induction of apoptosis of the hepatocyte. The clinical manifestations of drugs hepatotoxicity are gastrointestinal symptoms, jaundice, neurologic manifestations, bleeding, acute renal injury or ascites(5-9). We present some of the drugs most frequently incriminated in hepatotoxicity in children, following their mechanism of action. The main drugs that cause liver injury in children are presented in Table 1.
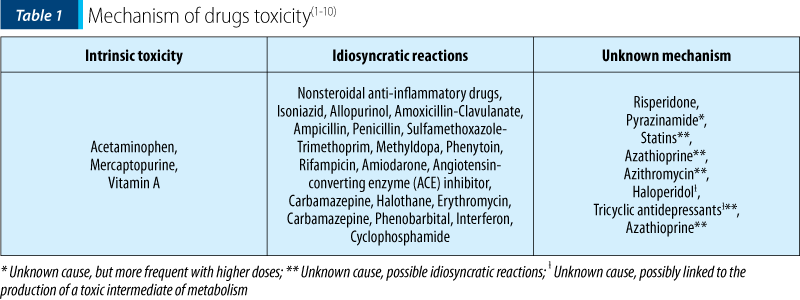
Intrinsic hepatotoxicity occurs as a result of large quantities of a hepatotoxic compound or its related metabolites. Based on the way they act upon the hepatic structure, intrinsic hepatotoxicity is classified in direct or indirect hepatotoxicity. In direct hepatotoxicity, the lesions are produced through the direct action of toxins upon the hepatocyte (cytotoxic effect) and the result is represented by the hepatic steatosis or areas of necrosis. Indirect hepatotoxicity consists of the alteration of certain intracellular synthesis metabolic processes or transport processes which determine the injury of the cellular function and, subsequently, its death. The intrinsic hepatotoxicity depends on the dose, independent of the genetic background. The symptoms occur after a short time (hours, days) from ingestion. A representative for the direct intrinsic hepatotoxicity is the carbon tetrachloride, while acetaminophen in large doses could determine cellular lesions both through direct, as well as through indirect mechanism(7-10).
Acetaminophen
Acetaminophen is the prototype of intrinsic hepatotoxicity. It is frequently used for its analgesic/antipyretic effects, but in high doses can cause severe hepatic necrosis. Fortunately, the administration of acetylcysteine (N-ACC), as an antidote in the acetaminophen intoxication, leads to a small rate of mortality in these cases. Normally, acetaminophen is metabolized directly in the liver on the glucuronic and sulphaconjugation route (95%) in metabolites that are not toxic and are eliminated through urine. When large doses are administered, these routes become oversaturated and the metabolization of acetaminophen shall be performed mainly on the P450 cytochrome route (Figure 1). The toxic metabolites resulted, such as N-acetyl-p-benzoquinoneimine (NAPQI), will determine mitochondrial disfunction and cellular death (cytotoxicity and necrosis). NAPQI depletes the glutathione reserves. As glutathione is the natural antioxidant of the liver, the result will be apoptosis, cellular lysis, and central-lobular necrosis(11-16).
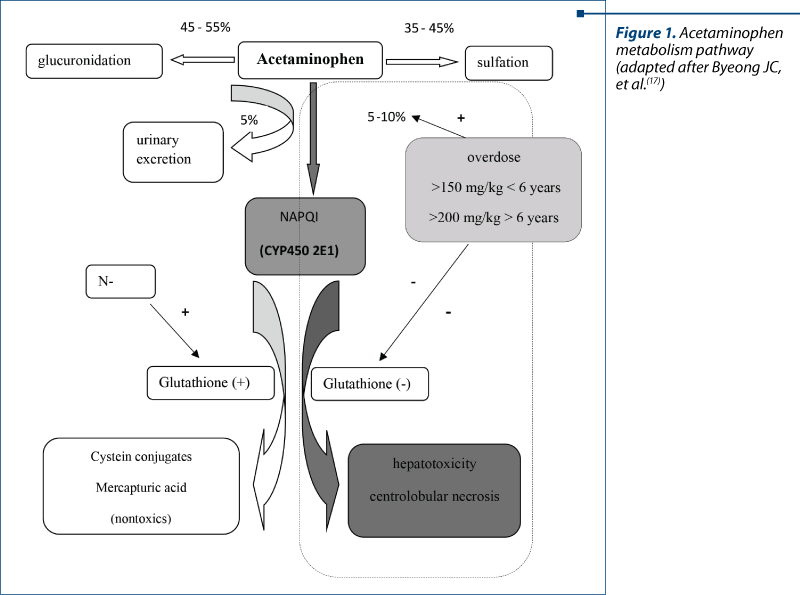
The acetaminophen toxicity is dependent on the dose. Toxic doses are over 150 mg/kg for the child below 6 years old or over 200 mg/kg for the child above 6 years old. Although the liver injury starts after 24-72 hours following the ingestion, the clinical symptoms usually occur in a late stage (after 48-96 hours from ingestion) – Table 2. These symptoms are represented by jaundice, convulsions or coma, liver failure and death(14).
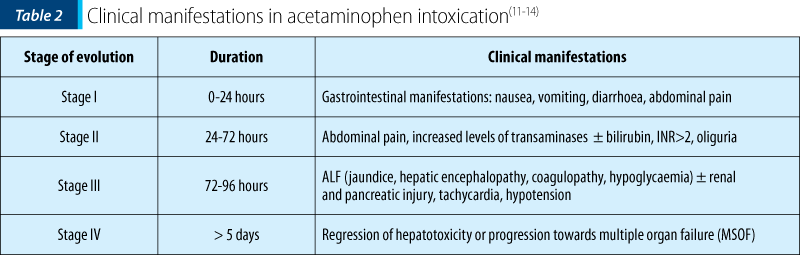
The level of acetaminophen in the blood is used for the assessment of the severity of the acetaminophen overdose and the risk of liver toxicity. The absence of toxic risk is given by a level of acetaminophen lower than 150 mg/l at 4 hours from ingestion or lower than 2.5 mg/l at 15 hours from ingestion(16). Reversely, if at 4 hours from ingestion the level of acetaminophen exceeds 200 mg/l, respectively 30 mg/l at 15 hours, then the risk of hepatic toxicity is high, and the administration of N-ACC (300 mg/kg/day) is necessary(18).
The idiosyncratic hepatotoxicity occurs in people with a genetic predisposition, independent of the dose, and involves immune-mediated phenomenon. The hepatotoxicity phenomena appear after weeks from the exposure, being secondary to a metabolic or an immune mechanism. The idiosyncratic hepatotoxicity develops as a result of certain genetic mutations that increase the susceptibility to the action of the toxic agent, the best example being isoniazid. The immune mechanism is a hypersensitivity reaction after the interaction between the toxic and immune system. Due to this reason, cutaneous rash, fever, arthralgia, eosinophilia or other autoimmune features such as an increase of immunoglobulins G (IgG) or the presence of serum antinuclear antibodies (ANA) could be associated in this type of hepatotoxicity(10).
Isoniazid (INH)
Isoniazid (INH) is a bactericide antibiotic, with intense selective action against Mycobacterium tuberculosis. It can determine hepatotoxicity through idiosyncratic reaction. INH is metabolized predominantly (50-90%) to N-acetylizoniazide (Ac-INH) and in a lower proportion to isonicotinic acid (INA). These metabolites, together with the one produced by the INH bioactivation itself, are responsible for the liver injury (Figure 2). The hepatotoxicity mechanism is independent of the dose, but is dependent on the genetic background and the onset could be at weeks/months from the administration. In case of overdosage of INH, the clinical manifestations are predominantly neurological (coma, convulsions, ataxia, psychotic reactions, death)(19-22).
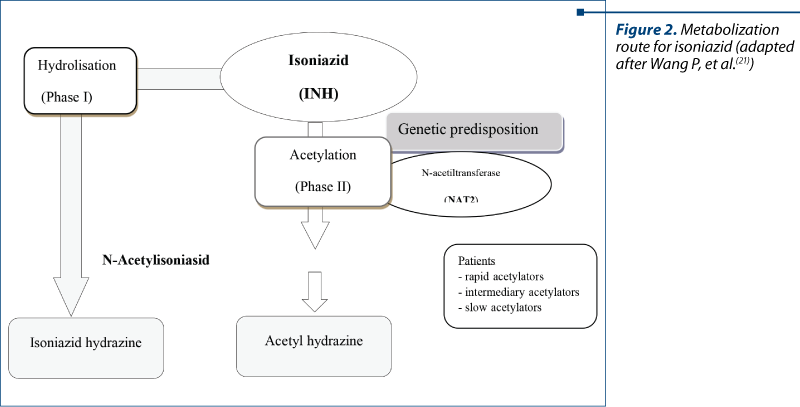
Depending on the genetic polymorphism and the presence of N-acetyltransferases gene mutation, the population is divided into slow, intermediary and fast acetylators. The slow acetylators display a higher risk of hepatotoxicity after the therapy with INH because of the prolonged exposure to the toxic element by increasing the half-life time. Certain authors sustain the hypothesis that the fast acetylators are also predisposed to toxicity because of the fast formation of large quantities of toxic metabolites (Figure 2). Other studies describe the occurrence of anti-INH or anti-P450 antibodies in patients diagnosed with ALF induced by IHN(19-21).
Valproic acid
Valproate is a fat organic acid usually used in the treatment of convulsions. Frequently, it can determine liver injury, with a slight increase of transaminases. If large doses are used, it could cause severe liver disease, such as ALF, Reye syndrome or hyperammonaemia. The mechanism of liver toxicity is the mitochondrial destruction that generates subsequently the death of hepatocytes.
Hyperammonaemia is usually present in diseases that alter the metabolism of the fatty acids or in urea cycle disorders. The antidote in the overdosage of valproic acid is the parenteral administration of carnitine. The severe forms that evolve with hyperammonaemia or neurological manifestations could benefit from extracorporeal epuration methods(23,24).
Albendazole
The administration of albendazole even in therapeutic doses could be associated with liver injuries, steatosis or periportal necrosis. Albendazole has an antiparasite effect by the increased avidity of the drug itself, as well as of its metabolites for tubulin, inhibiting the polymerisation of microtubules. Microtubules are an important component of the body of the parasite, but also represent important constituents of eukaryote cells cytoskeleton, being part of the internal structure of chilli and flagella, also involved in the intracellular movement (of the secretor vesicles, organelle, chromosomes). Linking to the tubulin, albendazole delays the formation of microtubules and, in this way, it could amplify the hepatocyte destruction. Often, albendazole is prescribed for the treatment of intestinal parasitosis, some proven, other only suspected. In a liver injury after albendazole, there is no antidote. The administration of corticoids proved its utility in the forms that associate elements of autoimmunity. The use of this drug only in very well documented cases of digestive parasitosis (hydatic cyst, aspergillosis, or severe parasite infections) could reduce the number of cases with liver injury(25-28).
Nonsteroidal anti-inflammatory drugs (NSAIDs)
NSAIDs represent one of the most frequently prescribed drugs around the world, being used in children as anti-inflammatory, analgesic or antipyretic drugs. They represent an important cause of idiosyncratic reactions, causing approximately 10% of cases of drug hepatotoxicity in both children and adults.
Aspirin induces hepatotoxicity through a dose-mediated mechanism, by direct damage to the hepatocyte, or impaired mitochondrial function. In some children, aspirin can cause Reye syndrome, a severe condition characterized by metabolic acidosis, fulminant liver failure with hepatic encephalopathy, hypoglycemia and coagulopathy. This is the reason why, at present, the aspirin use in children is limited only to exceptional cases.
In adults, an important cause of hepatotoxicity occurs secondary to diclofenac use, which is less prescribed in the pediatric population.
Ibuprofen is the most frequently used drug in children, being known for its safe properties and a very low level of hepatotoxicity. In the rare cases of hepatotoxicity that have been described, it was secondary to both mechanisms: dose-dependent hepatotoxicity or immune-mediated toxicity. Hepatotoxicity secondary to ibuprofen may range from mild transaminases to severe forms of cholestatic hepatitis with impaired liver function(29,30).
Antibiotics
Another important cause of toxic hepatitis may be the use of antibiotics, such as penicillins, cephalosporins, clavulanic acid or macrolides. Their administration can frequently lead to cholestatic liver injury, the mechanism being both direct toxicity and hypersensitivity. The outcome of these cases is often favourable, with the remission of manifestations after stopping the administration(31-33).
Amoxicillin-clavulanate is the most frequent drug that causes idiosyncratic DILI in adults in developed countries, as cholestatic hepatitis. The combination of amoxicillin with clavulanic acid is significantly associated with a risk of DILI compared to amoxicillin alone. Once the administration is stopped, the clinical manifestations improve(34).
Cephalosporins have low hepatotoxicity potential, being described only a few cases of liver injury in children. They can cause liver damage through the direct toxic effect or by idiosyncratic reaction. More particular is ceftriaxone, which is known for its potential to cause biliary sludge with symptoms of cholecystitis and cholestatic hepatitis(35).
Sulfamethoxazole/trimethoprim (TMP-SMX) is the first-line antibiotic used for the treatment and prophylaxis of pneumonia with Pneumocystis jirovecii in immunocompromised patients or in those who have received an organ transplant. In adults, TMP-SMX may cause toxic hepatitis with an incidence of 1/11,000-45,000, while in children it is rarely reported.
Hepatotoxicity is secondary to both mechanisms: direct toxicity triggered by the accumulation of toxic metabolites and allergic response, in this case accompanied by hypersensitivity reaction manifestations. The clinical manifestations of hepatotoxicity can vary from asymptomatic forms to severe disease, with ALF. Also, in children, the vanishing bile duct syndrome was described in a few cases(36,37).
Regarding macrolides, an incidence of hepatotoxicity of 1-5% is reported in the general population, the vast majority being self-limited. In children, there are described only a few cases, including some with cholestatic hepatitis or vanishing bile duct syndrome(38). According to some authors, the risk of hepatotoxicity in children is higher after clarithromycin administration compared to azithromycin(39).
Anaesthetics
Among the anaesthetics, halothane is commonly implicated in liver injury. Hepatotoxicity after halothane is due to an idiosyncratic mechanism, appearing after repeated exposures to this substance that determines the stimulation of the immune system. The autoantibodies are produced (anti-CYP2E1) and will cause hepatocytes destruction, with fever, rash, arthralgias, or eosinophilia(40,41). Regarding the hepatotoxicity after inhaled anaesthetics, it is proved that their effect is inferior to systemic anaesthetics. Of all, sevoflurane has the least hepatotoxic potential; for this reason, it is one of the most commonly used volatile anaesthetics agents in children. In the few cases that cause hepatotoxicity, sevoflurane is similar to halothane. It is degraded in the liver to intermediate metabolites that bind to cytoplasmic proteins, resulting in complexes that stimulate autoantibodies production(42).
Conclusions
In pediatrics, the early recognition of the possible cause of liver injuries may be the key for a favorable outcome of such cases that can have also a severe evolution. These cases require significant resources and sometimes expensive treatments, from hepatic dialysis to emergency liver transplantation in severe forms. For these reasons, an early diagnosis, a specific therapy depending on the etiology or sometimes the emergency referral to a toxicology specialized centre and also the consideration of liver transplantation could be vital for these patients.
Conflicts of interests: The authors declare no conflict of interests.
Bibliografie
- Calnot A, Perret C. Liver zonation. In: Monga S.P.S. (editor). Molecular Pathology of Liver Diseases. New York, USA: Springer Science. 2011; 2: 7-16.
- Sokol JR, Narkewicz RM. Liver and pancreas. In: Hay WW, Levin JM, Sondheimer MJ, Deterding RR (editors). Current Diagnosis & Treatment Paediatrics, 20th edition, Mc Graw Hill Education, New-York. 2011; 631-651.
- Pandit A, Sachdeva T, Bafna P. Drug-Induced Hepatotoxicity: A Review. Journal of Applied Pharmaceutical Science. 2012; 02(05): 233-243.
- Dhawan A. Aetiology and Prognosis of Acute Liver Failure in Children. Liver Transplantation. 2008; 14(2): S80-S84.
- Grama A, Burac L, Cainap SS, Aldea C, Delean D, Bulata B, et al. Acute liver failure in children: aetiology and evolution. Arch Dis Child. 2019; 104(Suppl 3): A1-A428.
- Bansai S, Dhawan A. Acute liver failure. Curr Pediatr. 2006; 16: 36-42.
- Mehta N, Ozick LA, Gbadehan E. Drug-induced hepatotoxicity. http://emedicine.medscape.com.
- Colleti J Jr, Azevedo RT, de Calvalho WB. Pediatric Acute Liver Failure: Current Perspectives. Liver Research. 2017; 2(1): 14-15.
- Grama A, Aldea C, Burac L, Delean D, Boghitoiu D, Bulata B, et al. Acute liver failure secondary to toxic exposure in children. Arch Med Sci. 2019; doi:10.5114/aoms.2019.87716
- Roth RA, Ganey PE. Intrinsic versus Idiosyncratic Drug-Induced Hepatotoxicity – Two Villains or One. J Pharmacol Exp Ther. 2010; 332(3): 692-697.
- Farrell S, Miller MA. Acetaminophen Toxicity. Available at: emedicine.medcape.com. Accesed March 2020.
- Yoon E, Babar A, Choudhary M, Kutner M, Pyrsopoulos N. Acetaminophen-Induced Hepatotoxicity: A Comprehensive Update. J Clin Transl Hepatol. 2016; 4(2): 131-142.
- Mazaleuskaya LL, Sangkuhl K, Thorn FC, FitzGerald AG, Altman R, Klein ET. PharmGKB summary: Pathways of acetaminophen metabolism at the therapeutic versus toxic doses. Pharmacogenet Genomics. 2015; 25(8): 416-426.
- Hinson J, Roberts WD, James PL. Mechanisms of Acetaminophen-Induced Liver Necrosis. Handb Exp Pharmacol. 2010; (196): 369-405.
- Mirochnitchenko O, Weisbrot-Lefkowitz M, Reuhl K, Chen L, Yang C, Inouy M. Acetaminophen Toxicity Opposite Effects of two forms of glutathione peroxidase. J Biol Chem. 1999; 274: 10349-10355.
- Rumack-Mathew nomogram. Available at: https:/www.emnote.org/emnotes/rumack-matthew-nomogram
- Byeong JC, Seung MM, Kim HS. Antidote for acetaminophen poisoning: N-acetyl cysteine. J Korean Med Assoc. 2013; 56 (12): 1067-1075.
- Heard KJ. Acetylcysteine for Acetaminophen Poisoning. N Engl J Med. 2008; 359(3): 285-292.
- Drug record. Clinical and Research Information of Drug-Induced Liver Injury. Available at: https://livertox.nih.gov/Isoniazid.htm
- Kompoliti K, Horn S. Drug-Induced and Iatrogenic Neurological Disorders. In: Goetz C. (ed). Textbook of Clinical Neurology (Third Edition), Philadelphia, USA, Sauders Elsevier 2007: 1285-1318.
- Wang P, Pradhan K, Zhong X, Ma X. Isoniazid metabolism and hepatotoxicity. Acta Pharm Sin B. 2016; 6(5): 384-392.
- Preziosi P. Isoniazid: Metabolic Aspects and Toxicological Correlate. Curr Drug Metab. 2007; 8: 839.
- Pirmohamed M, Leeder SJ. Anticonvulsant agents. In: Kaplowitz N, DeLeve LD, editors. Drug-induced liver disease, 3rd ed. Amsterdam: Elsevier; 2013: 423-41.
- Nasa P, Sehrawat D, Kansal S, Chawla R. Effectiveness of haemodialysis in a case of severe valproate overdose. Indian J Crit Care Med. 2011; 15(2): 120-122.
- Shah C, Mahapatra A, Shukla A, Bhatia S. Recurrent acute hepatitis caused by albendazole – case report. Trop Gastroenterol. 2013; 34 (1): 38-39.
- Freire JF, Rocha LMC, Lima LG, Raposo PRA. Subfulminant Acute Liver Failure by Albendazole: Case Report. J Med Cases. 2015; 6(8): 342-345.
- Choi GY, Yang HW, Cho SH, Kang DW, Go H, Lee WC. Acute drug-induced hepatitis caused by albendazole. J Korean Med Sci. 2008; 23(5): 903-905.
- Zuluaga IJ, Marin Castro EA, Perez Cadavid CJ, Gutierrez CRJ. Albendazole-induced granulomatous hepatitis: a case report. J Med Case Rep. 2013; 7: 201.
- Manov I, Motanis H, Frumin I, Iancu TC. Hepatotoxicity of anti-inflammatory and analgesic drugs: ultrastructural aspects. Acta Pharmacol Sin. 2006; 27: 259-72.
- Bessone F. Non-steroidal anti-inflammatory drugs: What is the actual risk of liver damage?. World J Gastroenterol. 2010; 16(45): 5651-5661.
- Hautekeete ML. Hepatotoxicity of antibiotics. Acta Gastroenterol Belg. 1995; 58: 290-6.
- Presti ME, Janney CG, Neuschwander-Tetri BA. Nafcillin-associated hepatotoxicity. Report of a case and review of the literature. Dig Dis Sci. 1996; 41: 180-4.
- Derby LE, Jick H, Henry DA, Dean AD. Erythromycin-associated cholestatic hepatitis. Med J. 1993; 158: 600-2.
- Donati M, Motola D, Leone R, Moretti U, Stoppa G, Arzenton E, et al. Liver Injury Due to Amoxicillin vs. Amoxicillin/Clavulanate: A Subgroup Analysis of a Drug-Induced Liver Injury Case-Control Study in Italy. J Hepatol Gastroint Dis. 2017; 3:1.
- LiverTox: Clinical and Research Information on Drug-Induced Liver Injury [Internet]. Bethesda (MD): National Institute of Diabetes and Digestive and Kidney Diseases; 2012-. Cephalosporins. Available from: https://www.ncbi.nlm.nih.gov/books/NBK548666
- Abusin S, Johnson S. Sulfamethoxazole/Trimethoprim induced liver failure: a case report. Cases J. 2008; 1(1): 44.
- Hyun JC, Hye JJ, Kyu SK, Dae YG, Jae YKK. Immunoallergic drug-induced hepatitis: lessons from halothane. J Hepatol. 1997; 26(S1): 5-12.
- LiverTox: Clinical and Research Information on Drug-Induced Liver Injury [Internet]. Bethesda (MD): National Institute of Diabetes and Digestive and Kidney Diseases; 2012-. Macrolide Antibiotics. Available from: https://www.ncbi.nlm.nih.gov/books/NBK548398/
- Ferrajolo C, Verhamme KMC, Trifirò G, ‘t Jong GW, Picelli G, Giaquinto C, et al. Antibiotic-Induced Liver Injury in Paediatric Outpatients: A Case-Control Study in Primary Care Databases. Drug Saf. 2017; 40(4): 305-315.
- Lo SK, Wendon J, Mieli-Vergani G, Williams R. Halothane-induced acute liver failure: continuing occurrence and use of liver transplantation. Eur J Gastroenterol Hepatol. 1998; 10: 635-9.
- Rajan S, Garg D, Cummings KC 3rd, Krishnaney AA. Hepatotoxicity after sevoflurane anaesthesia: a new twist to an old story. Br J Anaesth. 2019; 122(4): e63-e64.
- LiverTox: Clinical and Research Information on Drug-Induced Liver Injury. Bethesda (MD): National Institute of Diabetes and Digestive and Kidney Diseases; 2012-. Sevoflurane. Available from: https://www.ncbi.nlm.nih.gov/books





