Purpose. The aim of the study was to observe and to describe the evolution of early diagnosed hearing loss in children with neonatal auditory neuropathy spectrum disorders identified by universal neonatal hearing screening.
Materials and method. In the last years, the development of universal auditory newborn screening program, that covers all maternities in our city, allowed us to test initially over 50,000 of babies and to identify and manage newborns with auditory neuropathy spectrum disorders after clinical audiological assessments. The follow-up was done for each child periodically until 18 months for cases fitted by hearing aids and until the cochlear implantation for cases with this indication.
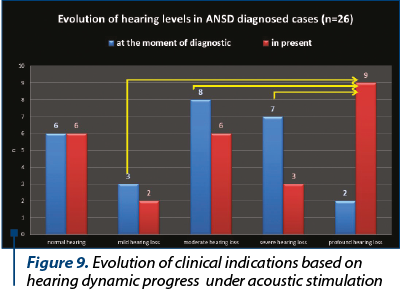
Results. We identified 26 children with auditory neuropathy spectrum disorders, six children with normal tonal hearing and 20 cases with different degrees of hearing loss. According to the results of our follow-up process, all children with normal tonal hearing kept their auditory status in time. All deaf children were hearing aid fitted, except the profound hearing loss with direct indication for cochlear implantation. In the group of children with hearing loss, we observed that 35% presented progressive forms and 15% presented fluctuating forms. 57% of progressive hearing loss group evolved to the profound deafness and cochlear implant indication. The greater the initial diagnosed degree of hearing loss, the more the children have progressed to profound deafness.
Conclusions. A lot of children with auditory neuropathy spectrum disorders present progressive hearing loss, the profound form being finally present in more than 45% of cases. The periodical follow-up is essential in the first 18 months in order to offer the best medical indication and to avoid the delay of appropriate intervention for hearing rehabilitation and speech development.
Evoluţia hipoacuziei la copiii cu tulburări din spectrul neuropatiei auditive, identificată prin screening neonatal auditiv
Evolution of hearing loss in children with auditory neuropathy spectrum disorders identified by neonatal hearing screening
First published: 30 noiembrie 2018
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/Orl.41.4.2018.2117
Abstract
Rezumat
Obiective. Scopul studiului a fost de a observa şi de a descrie evoluţia hipoacuziei diagnosticate precoce la copiii cu tulburări neonatale din spectrul neuropatiilor auditive identificate prin screeningul auditiv universal la nou-născuţi.
Materiale şi metodă. În ultimii ani, dezvoltarea unui program universal de screening pentru nou-născuţi, care acoperă toate maternităţile din oraşul nostru, ne-a permis testarea iniţială a peste 50.000 de copii. În urma evaluărilor clinice audiologice efectuate, au fost posibile identificarea nou-născuţilor cu tulburări din spectrul neuropatiei auditive şi gestionarea acestora din punctul de vedere al monitorizării evoluţiei şi al recomandării tratamentului necesar hipoacuziei. Urmărirea a fost efectuată periodic pentru fiecare copil până la 18 luni pentru cazurile protezate cu aparate auditive şi până la implantarea cohleară pentru cazurile cu această indicaţie.
Rezultate. Am identificat 26 de copii cu tulburări din spectrul neuropatiei auditive, şase cu auz normal şi 20 de cazuri cu diferite grade de hipoacuzie. Conform rezultatelor procesului nostru de urmărire, toţi copiii cu auz normal au păstrat în timp pragurile auditive. Toţi copiii hipoacuzici au fost protezaţi auditiv convenţional, cu excepţia celor cu hipoacuzie neurosenzorială profundă, cu indicaţie directă pentru implantarea cohleară. În grupul de copii cu pierdere de auz, am observat că 35% prezentau forme progresive şi 15% prezentau forme fluctuante. Din grupul cu hipoacuzie progresivă, 57% au evoluat până la surditate profundă, primind indicaţie de implant cohlear. Cu cât mai important a fost gradul hipoacuziei diagnosticate iniţial, cu atât copiii au prezentat o hipoacuzie progresivă, ajungând până la hipoacuzie profundă.
Concluzii. Mulţi copii cu tulburări din spectrul neuropatiei auditive prezintă pierdere progresivă de auz, forma profundă fiind constatată în final la mai mult din 45% din cazuri. Urmărirea periodică este esenţială în primele 18 luni pentru a oferi cele mai bune indicaţii medicale şi pentru a evita întârzierea intervenţiilor necesare pentru reabilitarea auditivă şi pentru dezvoltarea limbajului.
Introduction
ANSD (auditory neuropathy spectrum disorders) represent a group of auditory impairments that may be caused by pathologic processes of: internal hair cells of Corti organ, cochlear-neural synaptic space or cochlear nerve fibers. Due to particular aspects in children with auditory neuropathy spectrum disorders consisting in the degree and evolution of hearing loss, audiometric configuration, speech perception and hearing aid benefit(1-5), there is not yet a consensus in the therapeutic recommendation in newborns with auditory neuropathy(6,7,8). ANSD could be early identified after the hearing neonatal screening by a comprehensive audiological assessment.
Purpose of the study
The objectives of the current study are: to report our ANSD cases resulting from neonatal hearing screening, to identify and to describe specific patterns of hearing dynamic progress and speech development evolution in ANSD cases diagnosed immediately after our hearing neonatal screening.
Materials and method
The study included all babies referred from maternities and diagnosed with auditory neuropathy in the Audiology Department of Rehabilitation Clinical Hospital Iaşi, Romania, in the last six years. In this period, within the universal auditory newborn screening program in maternities there were tested over 50,000 babies and, after the clinical audiological assessments in the audiology department, we identified and managed newborns with auditory neuropathy spectrum disorders.
ANSD cases were diagnosed on the base of the following clinical criteria: the presence of otoacoustic emissions (at least at birth) and/or abnormal or absent ABR responses, the presence of cochlear microphonics and/or abnormal or absent ABR (auditory brainstem response), mismatches between ABR thresholds and ASSR (auditory steady state response) results or free field behavioral auditory thresholds(9-12).
The follow-up was done for each child periodically until 18 months (at least three clinical evaluations) for cases fitted by hearing aids and until the cochlear implantation surgery for cases with this indication.
The follow-up of ANSD cases included:
auditory assessment periodic control (every three or four months), including ASSR, ABR (rarefaction and condensation polarity), free-field behavioral audiometry, tympanometry, otoacoustic emissions, free-field behavioral audiometry with hearing aids or cochlear implant;
periodic reports from speech therapy specialist (describing performances with hearing aids or with cochlear implant) regarding tonal perception, message intelligibility according to age and the level of speech production.
Results and discussion
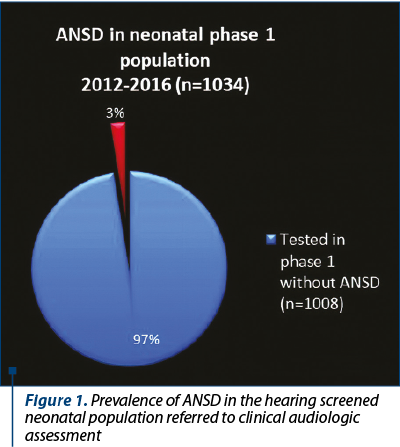
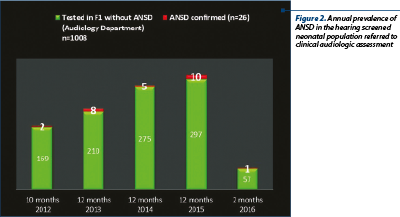
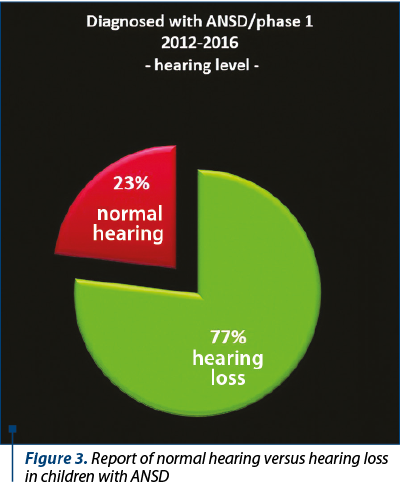
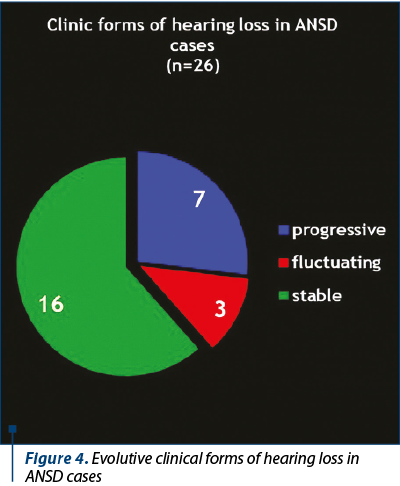
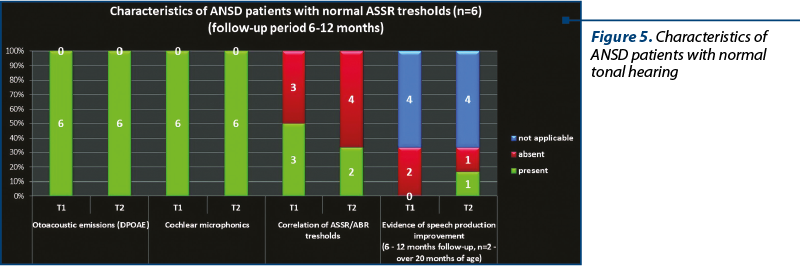
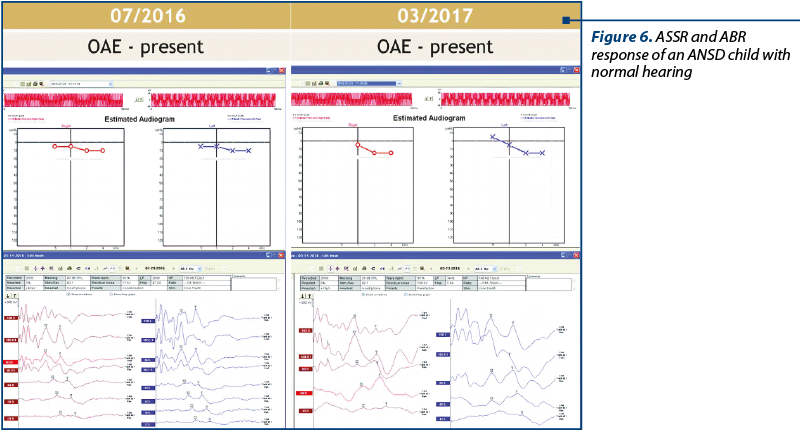
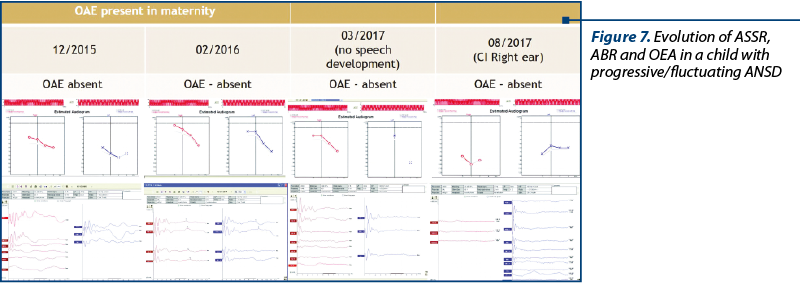
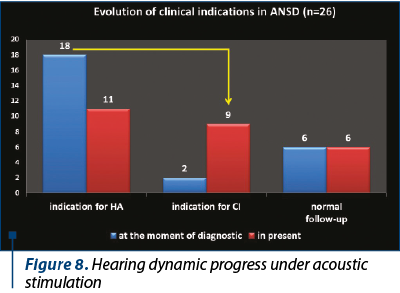
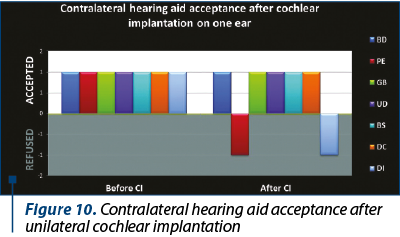
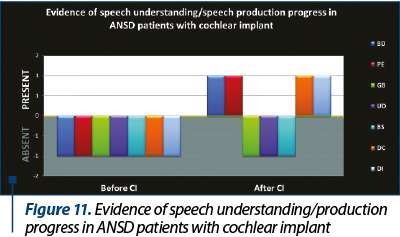
The development of universal auditory newborn screening program allowed us to test in the Department of Audiology 1034 newborns sent from maternities for otoacoustic emissions test and/or for the presence of one or more risk factors. We finally identified 26 children (3%) with auditory neuropathy spectrum disorders (Figure 1). Figure 2 shows the number of newborns identified by audiological testing during 2012 and 2016 in the first clinical phase. In our study there were diagnosed six babies with ANSD, presenting normal tonal hearing, and 20 cases were diagnosed with different degrees of hearing loss (Figure 3). In the group of children with hearing loss, we observed that seven children showed progressive forms, three presented fluctuating forms and ten children showed stable forms (Figure 4). All six cases of ANSD with normal hearing, according to the results of our follow-up process, conserved their auditory thresholds. In four cases, speech development cannot be appreciated yet (age between 8 and 15 months). In other two cases that did not develop speech production, one case showed evidence of a good evolution (Figure 5). An example of a newborn with ANSD and normal hearing is presented in Figure 6, where we can observe the presence of cochlear microphonics even 8 months later than the diagnosis moment. All deaf children were hearing aid fitted, except for those with profound hearing loss with direct indication for cochlear implantation. For mild/moderate/severe hearing loss we used the hearing aids as the first choice. At the follow-up, when we found stable hearing loss thresholds with evidence of auditory gain and good speech development, hearing aids remained the best solution, with a periodic follow-up. When we identified a progressive hearing loss (proved after hearing aid fitted), we recommended unilateral cochlear implantation (the existence of a low speech development rate was not an argument against the evidence of hearing loss progressivity). In the group of children with hearing loss, we observed three cases with fluctuating hearing loss (Figure 7 represents the fluctuations in ASSR thresholds level in a case with auditory neuropathy). For these children, the evaluation of the limits of fluctuation determined some difficulties for hearing aid fitting, and the evaluation of speech development showed us no evidences of good evolution. Depending on the available time or age of the patient, our indication was for unilateral cochlear implant. In our group, we did not find reversible hearing loss cases. In the group of progressive hearing loss, 57% (9 cases) evolved under acoustic stimulation by hearing aids to profound deafness and cochlear implant indication (Figure 8, Figure 9). The greater the initial diagnosed degree of hearing loss, the more the children have progressed to profound deafness. In cases with bimodal stimulation, two patients refused the contralateral hearing aid after few months from the switch-on of their cochlear implant. They are included among patients with evidence of good performances in speech development (Figure 10). Two patients with cochlear implant indication refused the surgery and they are still hearing aid users, with no evidence of speech development (Figure 11). Four out of seven of cochlear implanted children showed evidence of good speech development until now – following survey is necessary. Patients with ANSD fitted by hearing aid without cochlear implant indication present different levels of progress in speech understanding and production.
Conclusions
Universal hearing screening in newborns offers the chance to identify children with auditory neuropathy, requiring a different diagnosis and therapeutic approach compared to patients with regular inner ear hearing loss. Auditory neuropathy spectrum disorders cannot be diagnosed only on the base of screening auditory tests in maternity; the presence of different hearing loss risk factors is an important argument to indicate clinical audiological tests. ANSD was found in 3% of the newborns referred to our audiology department for clinical audiological tests (risk factors or “refer” result in screening tests), 23% presented normal tonal hearing and 77% presented different levels of hearing loss. We found different clinical forms of hearing loss: stable hearing level (16 cases), progressive hearing level (seven cases) and fluctuating hearing level (three cases). Because 35% of the patients with progressing hearing level switched from different degrees of hearing level to the profound one, we consider the periodical follow-up essential in the first 18 months in order to offer the best medical indication and to avoid the delay of appropriate intervention for hearing rehabilitation and speech development.
Conflict of interests: The authors declare no conflict of interests.
Bibliografie
- Mathai JP. Behavioural and Electrophysiological Correlates of Aided Performance in Individuals with Late Onset Auditory Neuropathy Spectrum Disorder: A Review. J Audiol Otol. 2018 Oct;22(4):171-177. doi: 10.7874/jao.2018.00031. Epub 2018 Sep 20.
- Narne VK, Prabhu P, Chandan HS, Deepthi M. Gender differences in audiological findings and hearing aid benefit in 255 individuals with auditory neuropathy spectrum disorder: a retrospective study. J Am Acad Audiol. 2016; 27:839–45.
- Mathai JP, Yathiraj A. Audiological findings and aided performance in auditory neuropathy spectrum disorder: a retrospective study. J Hear Sci. 2013; 3:18–26.
- Mathai JP, Yathiraj A. Performance-intensity function and aided improvement in individuals with late-onset auditory neuropathy spectrum disorder. Ear Hear. 2017; 38:e109–17.
- Yuvaraj P, Mannarukrishnaiah J. Audiological profile of adult persons with auditory neuropathy spectrum disorders. J Audiol Otol. 2016; 20:158–67.
- Narne VK, Prabhu P, Chandan HS, Deepthi M. Audiological profiling of 198 individuals with auditory neuropathy spectrum disorder. Hearing Balance Commun. 2014; 12:112–20.
- Ngo RY, Tan HK, Balakrishnan A et al. Auditory neuropathy/ auditory dys-synchrony detected by universal newborn hearing screening. International Journal of Pediatric Otorhinolaryngology. 2006; 70, 12991306.
- Jiang ZD. Neural conduction impairment in the auditory brainstem and the prevalence in term babies in neonatal intensive care unit. Clin Neurophysiol. 2014; http://dx.doi.org/10.1016/j.clinph.2014.10.147.
- Cozma S et al. Six years of universal neonatal hearing screening at Iaşi – the results of an interdisciplinary partnership. ORL.ro. 2015; VIII, no. 27, 26-31.
- Starr A, Picton TW, Sininger Y, Hood LJ, Berlin CI. Auditory neuropathy. Brain. 1996; 119 (Pt 3):741–53.
- Berlin CI, Hood LJ, Morlet T, Wilensky D, Li L, Mattingly KR, et al. Multi-site diagnosis and management of 260 patients with auditory neuropathy/dys-synchrony (auditory neuropathy spectrum disorder). Int J Audiol. 2010; 49:30–43.
- Lalayants MR, Brazhkina NB, Geptner EN, Kruglov AV, Barrlyak VV, Tavartkiladze GA. Auditory evoked potentials in children with auditory neuropathy spectrum disorder. Vestn Otorinolaringol. 2018; 83(4):15-20. doi: 10.17116/otorino201883415.
Articole din ediţiile anterioare
Impactul hipoacuziei
Hipoacuzia reprezintă un handicap senzorial frecvent întâlnit, care are un impact major și complex nu numai asupra persoanei cu deficiență de auz, ...
6 ani de screening auditiv neonatal universal la Iaşi - rezultatele unui parteneriat interdisciplinar
Programul de detecţie a hipoacuziei la nou-născuţi se desfăşoară la Iaşi în mod neîntrerupt din anul 2008. La baza succesului acestui program stă p...