Preeclampsia is a disease characteristic only to human pregnancy, birth being its only healing method. The morbidity and mortality associated with this condition are considerable, therefore therapies aimed at preventing the development of preeclampsia, which could delay the onset of the disease or decrease its severity, are essential. Pregnant women with an increased risk of thromboembolic disease require anticoagulant treatment throughout pregnancy. The effects and pharmacokinetics of the anticoagulant treatment used are mandatory to be known, given that in some cases the teratogenic effect may be present and in all cases the adaptation of the treatment near and during labor must be individualized. The low-molecular-weight heparin (LMWH) response is more predictable than unfractionated heparin, and in the context of adverse effects and unlikely overdose, routine monitoring by biological tests is not required. For each patient undergoing anticoagulant treatment during pregnancy for the purpose of preventing thrombotic injury, there should be explained the signs and symptoms of thrombosis, taking into account the fact that, even though the thrombotic risk is very low under anticoagulant treatment, a degree of risk still persists in pregnancy. Preeclampsia risk assessment project aims to gather all the data regarding the personal and familial history, obsterical and clinical data that impact the risk of developing a hypertensive pathology in pregnancy.
Strategii de prevenire a preeclampsiei. Farmacocinetica şi farmacodinamica heparinelor cu greutate moleculară mică în sarcină
Preeclampsia prevention strategies. Pharmacokinetics and pharmacodynamics of low-molecular-weight heparin (LMWH) in pregnancy
First published: 18 mai 2020
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/Gine.28.2.2020.3176
Abstract
Rezumat
Preeclampsia este o boală caracteristică numai sarcinii umane, având ca unică metodă de vindecare naşterea. Morbiditatea şi mortalitatea asociate acestei afecţiuni sunt considerabile, astfel încât sunt esenţiale terapiile destinate prevenţiei preeclampsiei, care ar putea întârzia apariţia bolii sau scădea severitatea acesteia. Femeile însărcinate cu un risc crescut de boală tromboembolică necesită tratament anticoagulant pe toată durata sarcinii. Efectele şi farmacocinetica tratamentului anticoagulant utilizat sunt obligatoriu a fi cunoscute, având în vedere că, în unele cazuri, efectul teratogen poate fi prezent. În toate cazurile, adaptarea tratamentului în proximitatea şi în timpul travaliului este obligatorie şi trebuie individualizată. Răspunsul heparinei cu greutate moleculară mică (HGMM) este mai previzibil decât al heparinei nefracţionate, iar în contextul efectelor adverse şi al unei supradoze improbabile, nu este necesară monitorizarea de rutină prin teste biologice. Pentru fiecare pacientă care urmează un tratament anticoagulant în timpul sarcinii, în scopul prevenirii leziunilor trombotice, trebuie explicate semnele şi simptomele trombozei, ţinând cont de faptul că, chiar dacă riscul trombotic este foarte scăzut sub tratament anticoagulant, un grad de risc încă persistă în sarcină. Proiectul de evaluare a riscului de preeclampsie îşi propune să colecteze toate datele referitoare la istoricul personal şi familial, date obstetricale şi clinice care au impact asupra riscului de a dezvolta o patologie hipertensivă în sarcină, în scopul prevenţiei acesteia.
Introduction
Preeclampsia is a disease characteristic only to human pregnancy, birth being its only healing method. Despite known and intensively studied risk factors, there are numerous cases of preeclampsia that occur in the absence of any risk factors present. The prediction of the risk of developing preeclampsia is difficult, the management being most often dictated by the evolution of the case and the results of the monitoring. The switch from any other anticoagulant to low-molecular-weight heparin (LMWH) is indicated to be made preconceptionally.
The morbidity and mortality associated with this condition are considerable, therefore therapies aimed at preventing the development of preeclampsia, which could delay the onset of the disease or decrease its severity, are essential. The main causes of mortality are premature birth, intrauterine growth restriction in the context of placental dysfunction and, respectively, preeclampsia. It has been demonstrated through numerous studies the efficacy of prophylactic administration of low-dose aspirin, started before 16 weeks of gestation, in reducing the prevalence of severe preeclampsia, premature birth and intrauterine growth restriction. Since the trophoblastic invasion of the spiral arteries begins around week 8-10 of gestation and continues until around week 22, but usually reaches complete invasion at 16-18 weeks of gestation, the onset of preventive treatment is indicated at 8-10 weeks(1).
ASPRE (Combined Multimarker Screening and Randomized Patient Treatment with Aspirin for Evidence-Based Preeclampsia Prevention) is one of the largest studies initiated for the study of preeclampsia prevention, which included an impressive 27,000 cases of pregnancy in the first trimester(2). The screening method used is an algorithm that uses the combination of maternal factors, mean blood pressure, uterine arterial pulsatility index, and biochemical markers such as pregnancy-associated plasma protein (PAPP-A) and placental growth factor (PLGF) between 11-13 weeks of gestation. The implemented algorithm was based on data collected from 60,000 singleton pregnancies, where it provided a detection rate of 76.6% for early preeclampsia and 38.3% for late preeclampsia, with a false positive rate of 10%(3). In addition, in the ASPRE study, women at increased risk of preeclampsia were included in a follow-up case-control study, by administering 150 mg of aspirin or placebo, starting treatment at 11-14 weeks of gestation up to 36 weeks of gestation. This study demonstrated a reduction in the incidence of early preeclampsia by 62% in the case group compared to placebo(4).
In conclusion, low-dose aspirin is recommended to be preventively administered for high-risk pregnancies of preeclampsia by both the US Preventive Services Task Force (USPSTF) and the American College of Obstetricians and Gynecologists (ACOG)(5).
The role of vitamin D in the prevention
of preeclampsia
In addition to the effect of low-dose aspirin in the prevention of preeclampsia, the effect of administering other compounds such as antioxidants, calcium supplements, fish oil, nitric oxide supplements, folic acid, nitric oxide donors or weight loss has also been studied, along with the effect of antihypertensive therapy and of the physical activity. Of all the above, it seems that only statin therapy would have a potentially beneficial effect(6). It has been speculated that the low level of vitamin D would be associated with the development of preeclampsia, but studies in this direction continue. The case-control study Vitamin D Antenatal Asthma Reduction Trial (VDAART)(7) was designed to evaluate the effect of vitamin D supplementation during pregnancy, in addition to the prenatal vitamins content for the prevention of asthma, or recurrent wheezing in newborns from parents with asthma or other atopic field. In the second phase of the study, the effect of vitamin D supplementation on pregnancy and on the incidence of preeclampsia was analyzed. In this context it has been shown that the level of serum vitamin D does not influence the risk of preeclampsia.
However, even though it did not have statistical significance, pregnant women with an increased vitamin D level, over 30 ng/ml, had a lower risk of preeclampsia. However, there is a possibility that serum vitamin D levels may be a marker for other factors that more directly affect placental function and the development of preeclampsia, and vitamin D supplementation in this context will not have a significant effect on the prognosis. Currently, the ACOG recommendations do not include routine screening of the vitamin D level in pregnancy, but encourage its measurement in high-risk cases of deficiency(8). It seems that the beneficial effect in reducing the risk of preeclampsia of vitamin D supplementation would exist only if the supplementation started prenatally and only in women with severe deficits(9).
In the long run, preeclampsia is associated with an increased risk of developing cardiovascular disease throughout life, so interventions that would decrease the prevalence of this condition provide the benefit of protection over the long-term pathology(10).
Low-molecular-weight heparin administration in pregnancy
Pregnant women with an increased risk of thromboembolic disease, such as those with an increased risk of deep vein thrombosis (DVT), cardiac pathology such as atrial fibrillation, left ventricular dysfunction or mechanical valve carriers, but also in some cases of spontaneous pregnancy loss, require anticoagulant treatment throughout pregnancy. The effects and pharmacokinetics of the anticoagulant treatment used are mandatory to be known, given that in some cases the teratogenic effect may be present and in all cases the adaptation of the treatment near and during labor must be individualized(11). The choice of the anticoagulant in pregnancy should have as a priority the safety of the mother and fetus. In this context, heparins are the most commonly used anticoagulants because they do not cross the placental barrier and do not lead to fetal anticoagulation. Low-molecular-weight heparin is indicated to be used rather than unfractionated heparin throughout pregnancy, except for the end of pregnancy period, because their route of administration is much easier and the efficacy is unquestionable. LMWH response is more predictable than that of unfractionated heparin, and in the context of adverse effects and unlikely overdose, routine monitoring by biological tests is not required(12). In contrast, unfractionated heparin is a preferential alternative in the context of the need for rapid reversibility of the anticoagulant effect, for example in preparation for emergency birth or surgery. Of course, in case of severe renal failure, with creatinine clearance less than 30 mL/min, unfractionated heparin remains the only anticoagulant option, given that LMWH clearance is almost exclusively renal, while that of unfractionated heparin is both renal and hepatic. In the presence of heparin-induced thrombocytopenia, however, when anticoagulation is absolutely necessary, the administration of X-factor inhibitors, fondaparinux, danaparoid or argatroban, may be considered as an option. Danaparoid is a low-molecular-weight heparinoid that does not cross the placenta, but conclusive studies on its safety in pregnancy are still lacking, instead it is an alternative to consider in the context of heparin-induced thrombocytopenia. Fondaparinux is a synthetic polysaccharide based on the active structure of heparin, but for which baseline studies on the safety of its administration in pregnancy and placental barrier crossing are also lacking. Vitamin K antagonists are avoidable in pregnancy because they have an increased risk of teratogenicity and crossing the placenta can cause fetal anticoagulation. Thus, if they are administered during the initial period of pregnancy, they can cause embryopathies, and in more advanced periods – fetal bleeding. In addition, vitamin K antagonists include as a side effect an increased risk of intracranial hemorrhage.

The switch from any other anticoagulant to LMWH is indicated to be made preconceptionally. For patients with chronic anticoagulant treatment, and for patients with indication of anticoagulant treatment only during pregnancy, the initiation of treatment with LMWH is recommended as soon as the pregnancy has been confirmed, in the context of the absence of vaginal bleeding.
LMWH safety in pregnancy
LMWH remains the preferential anticoagulant in pregnancy, given that human-controlled studies have shown the absence of risk (A), without demonstrating the existence of any degree of teratogenic risk in the first trimester of pregnancy or any risk of fetal impairment if it is administrated in the second or third trimester(13). Moreover, prophylactic or therapeutic LMWH administration is not associated with an increased risk of bleeding in the peripartum(14). The only adverse effect that may occur is the allergic reaction at the dermal level, with a higher incidence for dalteparin and nadroparin(15). Regarding the risk of heparin-induced thrombocytopenia, it is much lower for LMWH compared to unfractionated heparin(15). The cases of heparin-induced thrombocytopenia in pregnancy that have been described in literature belong to patients who have had episodes of heparin-induced thrombocytopenia and beyond pregnancy(16). Regarding heparin-induced osteoporosis, it is known and demonstrated that the risk for this condition associated with LMWH is much lower than that associated with unfractionated heparin. A 0.04% incidence was described by a study in this direction, with a single case of heparin-induced osteoporosis in a 36-week gestation case receiving high-dose LMWH(17).
Both LMWH and unfractionated heparin are usually administered subcutaneously, or intravenously for unfractionated heparin, with very good general tolerance to administration. Before starting the treatment, it is advisable to evaluate the status of the initial and periodic complete blood count, respectively the number of platelets, for a baseline value in case a reaction such as heparin-induced thrombocytopenia is suspected, even though the risk for this is very low as already been mentioned. Renal function is also required to be evaluated.
For each patient undergoing anticoagulant treatment during pregnancy for the purpose of preventing thrombotic injury, there should be explained the signs and symptoms of thrombosis, taking into account the fact that, even though thrombotic risk is very low under anticoagulant treatment, a degree of risk still persists in pregnancy. Alarm signs and symptoms are marked edema of the lower limbs symmetrically or asymmetrically, pleuritic chest pain and suggestive symptoms for central nervous system impairment. Regarding the risk of bleeding, it is recommended to switch from LMWH to unfractionated heparin before birth, with heparin discontinuation at the onset of labor for cases requiring continuous administration. In the case of premature birth, the administration of protamine sulphate can be considered as a reversal of the anticoagulant effect only if bleeding that cannot be controlled by classical uterotonic and hemostatic methods occurs(18).
Postpartum anticoagulation depends on the baseline condition and the associated thrombotic risk. In principle, for patients whose risk is increased by pregnancy, and do not require continuous anticoagulant therapy, prophylaxis for 6-8 weeks postpartum is sufficient(19).
Conclusions
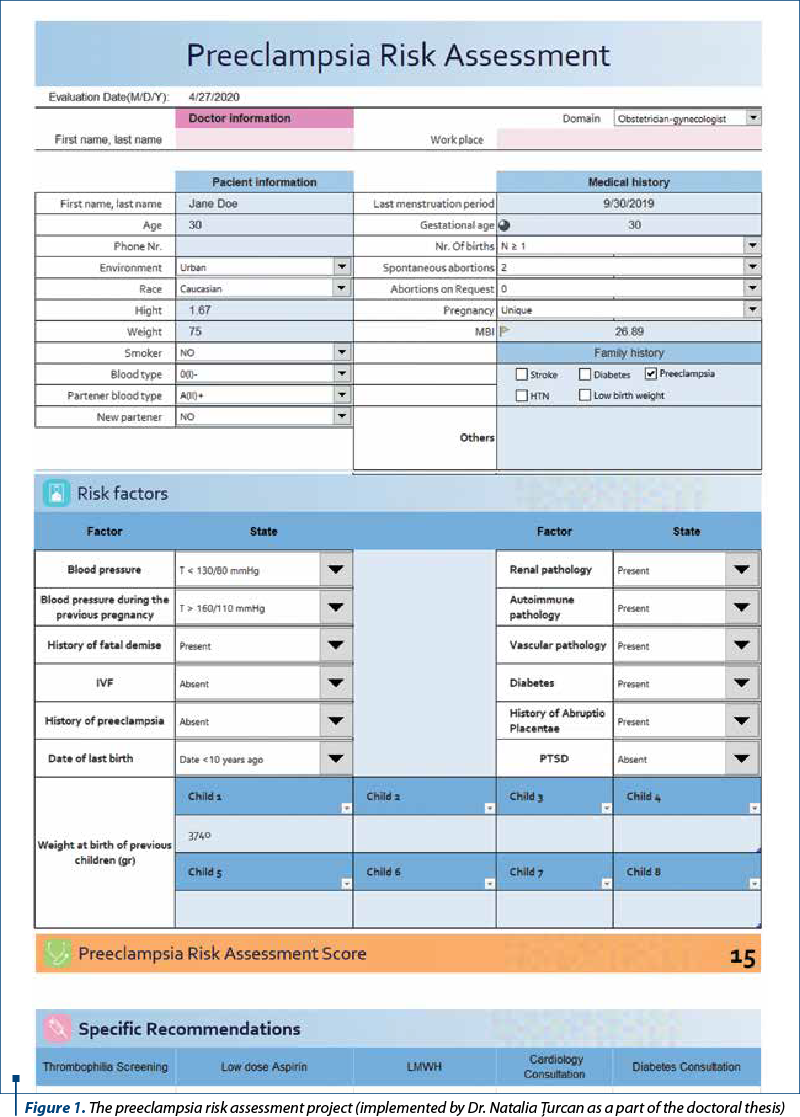
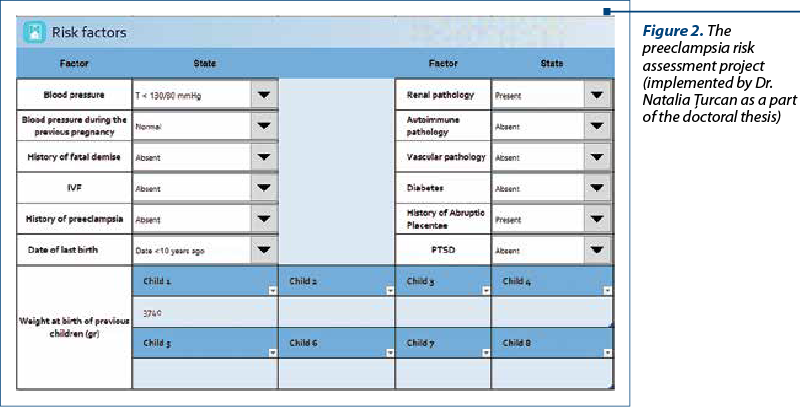
The preeclampsia risk assessment project (Figures 1-3) was implemented as a file in Excel 2016, offering the possibility to be used in any cabinet equipped with a computer without the need for special programs. The evaluation sheet in question aims to gather all the data regarding the personal and familial history, obstetrical and clinical data that impact the risk of developing a hypertensive pathology in pregnancy. This file also offers the possibility to be supplemented with information useful to the clinician; it offers the possibility to place it into the patient’s electronic file and to calculate a score that guides the prognosis regarding the development of preeclampsia.
The risk assessment sheet for preeclampsia can be completed by both the obstetrician and the family doctor who can guide the patient to a high-level obstetrics-gynaecology service specialized in monitoring and managing pregnancies at risk.
We consider this card beneficial both by providing a standardized data collection platform with the possibility of completing the evolution of pregnancy during it and, in addition, an essential tool for identifying patients at an increased risk of preeclampsia, and the possibility to act properly from the early stage of pregnancy for preventing the development of this condition.
The subset of this record is that the anamnestic and clinical data have the highest predictive weight of preeclampsia demonstrated so far.
Conflict of interests: The authors declare no conflict of interests.
Bibliografie
-
Pijnenborg R, Dixon G, Robertson WB, Brosens I. Trophoblastic invasion of human decidua from 8 to 18 weeks of pregnancy. Placenta. 1980; 191:3–19.
-
Rolnik DL, Wright D, Poon LCY, Syngelaki A, O’Gorman N, de Paco Matallana C, Akolekar R, Cicero S, Janga D, Singh M, Molina FS, Persico N, Jani JC, Plasencia W, Papaioannou G, Tenenbaum-Gavish K, Nicolaides KH. ASPRE trial: performance of screening for preterm pre-eclampsia. Ultrasound Obstet Gynecol. 2017; 50(4):492-5.
-
Akolekar R, Syngelaki A, Poon L, Wright D, Nicolaides KH. Competing risks model in early screening for preeclampsia by biophysical and biochemical markers. Fetal Diagn Ther. 2013; 33(1):8-15.
-
Rolnik DL, Wright D, Poon LC, O’Gorman N, Syngelaki A, de Paco Matallana C, Akolekar R, Cicero S, Janga D, Singh M, Molina FS, Persico N, Jani JC, Plasencia W, Papaioannou G, Tenenbaum-Gavish K, Meiri H, Gizurarson S, Maclagan K, Nicolaides KH. Aspirin versus placebo in pregnancies at high risk for preterm preeclampsia. N Engl J Med. 2017; 377(7):613-22.
-
LeFevre ML. US Preventive Services Task Force. Low-dose aspirin use for the prevention of morbidity and mortality from preeclampsia: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2014; 161(11):819–26.
-
Costantine MM, Cleary K. Eunice Kennedy Shriver National Institute of Child Health Human Development Obstetric – Fetal Pharmacology Research Units Network. Pravastatin for the prevention of preeclampsia in high-risk pregnant women. Obstet Gynecol. 2013; 121(2 pt 1):349–353.
-
Mirzakhani H, et al. Early pregnancy vitamin D status and risk of preeclampsia.J Clin Invest. 2016; 126(12):4702–15.
-
ACOG Committe on Obstetric Practice. ACOG Committee Opinion No. 495: Vitamin D: screening and supplementation during pregnancy. Obstet Gynecol. 2011; 118(1):197–8
-
Bohîlţea RE, Zugravu CA, Neacsu A, Navolan D, Berceanu C, Nemescu D, Bodean O, Ţurcan N, Baroş A, Cîrstoiu MM. The prevalence of vitamin D deficiency and its obstetrical effects. A prospective study on Romanian patients. Rev Chim. 2019; 70(4):1228-33.
-
McDonald SD, Malinowski A, Zhou Q, Yusuf S, Devereaux PJ. Cardiovascular sequelae of preeclampsia/eclampsia: a systematic review and meta-analyses. Am Heart J. 2008; 156(5):918–30.
-
Ţurcan N, Bohîlţea R, Neacsu A, Baroş A, Cîrstoiu MM. The role of anticoagulant therapy in the prevention of preeclampsia. Pharmacokinetic and pharmacodinamic mechanisms. Rev Chim. 2019; 70(4):1424-28.
-
Ţurcan N, Bohîlţea R, Cîrstoiu MM. Actualities on prediction and prevention of preeclampsia. Literature review. Gineco.eu. 2017; 13:156-61.
-
Forestier F, Daffos F, Rainaut M, Toulemonde F. Low molecular weight heparin (CY 216) does not cross the placenta during the third trimester of pregnancy. Thromb Haemost. 1987; 57(2):234.
-
Bates SM, Rajasekhar A, Middeldorp S, et al. American Society of Hematology 2018 guidelines for management of venous thromboembolism: venous thromboembolism in the context of pregnancy. Blood Adv. 2018; 2(22):3317-59.
-
Greer IA, Nelson-Piercy C. Low-molecular-weight heparins for thromboprophylaxis and treatment of venous thromboembolism in pregnancy: a systematic review of safety and efficacy. Blood. 2005; 106(2):401-7.
-
Huhle G, Geberth M, Hoffmann U, Heene DL, Harenberg J. Management of heparin-associated thrombocytopenia in pregnancy with subcutaneous r-hirudin. Gynecol Obstet Invest. 2000; 49(1):67-9.
-
Hunt BJ, Doughty HA, Majumdar G, et al. Thromboprophylaxis with low molecular weight heparin (Fragmin) in high risk pregnancies. Thromb Haemost. 1997; 77(1):39-43.
-
Bohîlţea R, Ţurcan N, Ionescu C, Toader O, Nastasia S, Neculcea D, Movileanu I, Munteanu O, Cîrstoiu M. The incidence of prematurity and associated short-term complications in a multidisciplinary emergency hospital from Romania.5th SRUOG Congress. 2017; 105-12.
-
Kearon C, Ginsberg JS, Julian JA, et al. Comparison of fixed-dose weight-adjusted unfractionated heparin and low-molecular-weight heparin for acute treatment of venous thromboembolism. JAMA. 2006; 296(8):935-42.
Articole din ediţiile anterioare
Importanţa alimentaţiei echilibrate înainte şi în timpul sarcinii
Alimentaţia femeii însărcinate este o parte esenţială a sarcinii pentru o bună dezvoltare materno-fetală. Totuşi, se pare că între 20% şi 30% din ...
Corelaţii anatomoclinice şi ecografice în hipertensiunea arterială indusă de sarcină
Hipertensiunea arterială indusă de sarcină este diagnosticată prin creşterea tensiunii arteriale sistolice peste 140 mmHg şi a celei diastolice...
Diagnosticarea şi prevenţia preeclampsiei - review de literatură
Preeclampsia este o patologie asociată sarcinii, care are o prevalenţă de 5-8%. Este una dintre cauzele majore de mortalitate şi morbiditate matern...
Valoarea predictivă a dozării sFlt, PIGF versus testarea raportului sFlt/PIGF la pacientele cu risc de preeclampsie
Hipertensiunea arterială este cea mai frecventă problemă medicală întâlnită în timpul sarcinii, complicând 2-3% din sarcini. Patogeneza preeclampsi...