Endometriosis and adenomyosis are pathologies that frequently affect women of reproductive age. For these women, obtaining a pregnancy is often difficult, some patients being advised to use assisted reproductive techniques. The ectopic endometrial-like tissue situated outside the uterine cavity responds to pregnancy-induced hormonal changes. This alters the appearance of the endometrial lesions. Therefore, the diagnosis and treatment of endometriosis in pregnant women are a real challenge. Endometriosis and adenomyosis preexistent to pregnancy have various effects on the mother and fetus, being a novel research subject in the recent years. The 2022 ESHRE Guidelines for endometriosis strongly recommend clinicians to be aware of adverse pregnancy outcomes, especially during the first trimester, in pregnancies associated with preexisting endometriosis. It is better to diagnose and treat endometriosis prior to obtaining a pregnancy, because pregnancy itself does not always improve endometriosis, as it was previously stated, and the patients often need a multidisciplinary approach. Although there are no specific follow-up protocols for pregnancies with preexisting endometriosis, specialists and their patients must be aware of the unfavorable outcomes. Our article is a review of the latest literature about the impact of endometriosis on pregnancy. We also present some examples of endometriosis that we encountered in patients admitted to our hospital.
Impactul endometriozei asupra sarcinii
Impact of endometriosis on pregnancy
First published: 31 octombrie 2022
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/ObsGin.70.3.2022.7204
Abstract
Rezumat
Endometrioza şi adenomioza sunt patologii care afectează frecvent femeile de vârstă reproductivă. Obţinerea şi menţinerea unei sarcini se fac de multe ori cu dificultate, unele paciente apelând la tehnici de reproducere umană asistată. Ţesutul endometrial ectopic este dependent de modificările hormonale induse de sarcină. Acestea schimbă aspectul leziunilor, punând uneori adevărate probleme de diagnostic şi tratament. Endometrioza şi adenomioza preexistente pot influenţa cursul sarcinii, prin diverse mecanisme care afectează mama şi fătul. Ghidul ESHRE pentru endometrioză din anul 2022 le recomandă specialiştilor să urmărească cu atenţie sporită sarcinile femeilor cu endometrioză, din cauza posibilelor efecte adverse asociate bolii, mai ales în primul trimestru. Diagnosticul şi tratamentul endometriozei şi adenomiozei înainte de obţinerea unei sarcinii sunt întotdeauna de preferat. Cazurile cu forme severe de endometrioză nu sunt influenţate pozitiv de sarcină şi necesită o abordare multidisciplinară. Cu toate că nu există protocoale speciale de urmărire a unei gravide cunoscute cu endometrioză sau adenomioză, medicul şi pacienta trebuie să ţină cont de posibilitatea apariţiei unor complicaţii grave. Acest articol prezintă un review al literaturii despre efectele endometriozei şi adenomiozei asupra mamei şi fătului. De asemenea, expunem şi exemple din experienţa noastră în domeniul formelor de endometrioză descoperite la o serie de paciente internate în spitalul nostru.
Background
Endometriosis is an inflammatory, chronic, estrogen-dependent disease that affects women worldwide. The pathological substrate is the endometrial-like tissue that grows outside the uterine cavity, responding to hormonal fluctuations.
Although women of child-bearing age are the most affected, adolescents and postmenopausal women deal with the clinical consequences and treatment requirements for this disease. The latest epidemiological statistics estimate that about 190 million women of reproductive age and adolescents currently suffer from endometriosis, as well as several postmenopausal women worldwide, but the exact prevalence is yet unknown. There is a 5-12-year delay between the beginning of symptoms and the time of a reliable diagnosis, because many symptoms are unspecific, and patients often refer to several different specialists before their correct diagnosis is being made(1). Moreover, until the latest ESHRE Guidelines published in 2022, the diagnostic gold standard for endometriosis was laparoscopy with histopathological confirmation.
The new guidelines now only recommend it in patients with negative imaging or in which the empirical treatment did not work. A special category of patients who deal with endometriosis are pregnant women. In the past years, clinical research stated that pregnancy alleviates endometriosis symptoms, but recent research shows that this is not true, and endometriosis has an important effect on pregnancy outcomes(2).
Materials and method
We searched the PubMed database for studies on adverse pregnancy outcomes and endometriosis published until August 2022. Our research is focused on miscarriage, spontaneous preterm birth, obstetric hemorrhage, preeclampsia and small for gestational age. Only English language meta-analyses, systematic studies and cohort studies were included. We also present examples of endometrial lesions found in pregnancies that were admitted to the University Emergency Hospital in Bucharest.
Results and discussion
1. Endometrioma
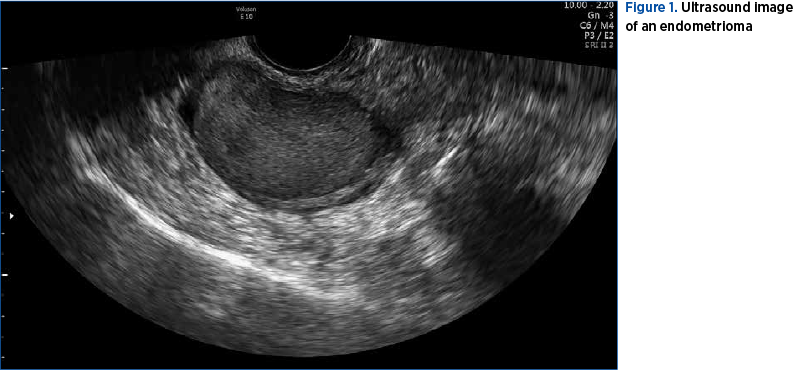
Many articles have been published along the years about endometriosis and pregnancy, but the information is difficult to categorize due to the large heterogenicity of studies and patients’ particularities. Endometriotic lesions change their appearance during pregnancy, therefore it is sometimes difficult to diagnose them correctly. The study of Ueda et al. (2010) states that in 0.5-1.2% of pregnancies, an ovarian cyst is diagnosed, but from those cysts, only 0.05-0.5% are endometriomas. The imaging aspects of endometriomas in pregnancy are different from the ones outside pregnancy due to decidualization. Sonographers may find typical or atypical cysts. Typical lesions are round cysts, with a minimum diameter of 10 mm, with thin, regular borders, with a mild echoic homogenous content, with diffuse echoes but no papillary formations. Atypical lesions are uni- or multilocular cysts, with a mild echoic content or with a “broken-glass” like structure, with vascularized intracystic papillae and a thin contour. In addition to ultrasound, MRI can be used, but biomarkers are not recommended(3). A large study published in 2015 by Maggiore et al. searched for cases of decidualized ovarian endometriosis. The meta-analysis contained articles from 1998 to 2015, but with a small number of patients. Most cases were patients who were not diagnosed with endometriosis prior to pregnancy and the lesions had an atypical imaging aspect, due to which MRI and CA125 were used for the differential diagnosis. Moreover, clinicians opted for surgical cyst removal in pregnancies under 22 weeks. Complications derived from endometrioma are part of the differential diagnosis of pelvic pain during pregnancy. Acute abdomen can occur due to torsion of enlarged cysts, rupture or infection (Maggiore et al., 2016)(1,4,5). In Figure 1, we show the ultrasound image of a large endometrioma.
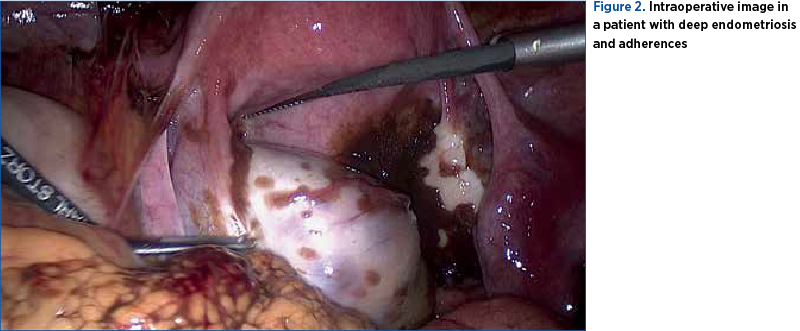
Besides ovarian endometriosis, pregnant patients may have superficial lesions or deep infiltrative lesions. Deep endometriosis is the most severe phenotype, with lesions that penetrate more than 5 mm under the surface of peritoneum, uterosacral ligaments, the ureter, bladder, and intestine, with or without occlusion or hydronephrosis. Extrapelvic endometriosis is pleural, umbilical, neural or diaphragmatic. Endometrial tissue that infiltrates in the myometrium is diffuse or focal adenomyosis, which is independently responsible for pain, bleeding and infertility. Peritoneal endometriosis can mimic carcinomatosis and bladder endometrioma must be differentiated from benign or malignant bladder tumors and calculus. Diffuse bladder endometriosis affects 1% of pregnancies. It rarely invades bladder serosa(6,7). In Figure 2, we show the intraoperative image of a patient with deep endometriosis and adherences.
2. Gastrointestinal
Studies of Setubal et al. (2014) and Villa et al. (2008) found that intestinal endometriosis is present in 5-12% of women with endometriosis. Spontaneous intestinal perforations were described in the third trimester of pregnancy or in the immediate postpartum period. Adhesions and extensive decidualization that weakens intestinal wall are possible pathological mechanisms(8,9). Acute appendicitis with peritonitis is rare, but possible (Glavind et al., 2018)(10).
3. Vascular
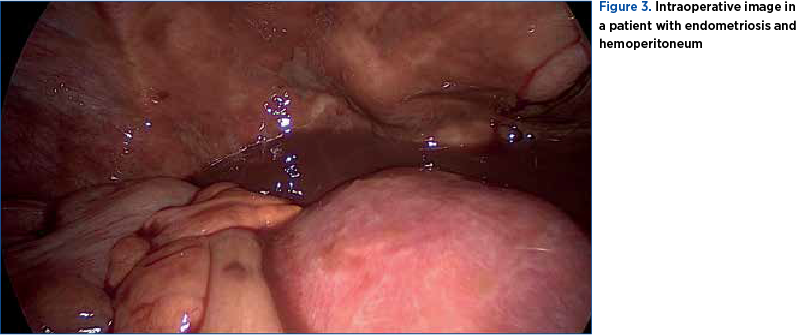
A very rare complication is spontaneous hemoperitoneum in pregnancy (SHiP). Only 20 cases were published in the literature. Bleeding occurs from pelvic endometriotic implants or ruptured vessels in the posterior uterine wall or in the parametrium. Maggiore et al. (2016) and, recently, Lier et al. (2017) found that most cases are described in the third trimester or in the postpartum, and in 94.5% of situations it led to emergency explorative laparotomy and caesarean section(1,11). In Figure 3, we show the intraoperative image in a patient with deep endometriosis and hemoperitoneum.
4. Uterine
Spontaneous uterine rupture was described in very few studies and is especially associated with previous surgery for endometriosis. The posterior uterine wall and lower segment are affected(9).
5. Urinary system
Uroperitoneum is very rare, being reported by Chiodo et al. (2015) and Maggiore et al. (2008). The extensive decidualization of severe deep endometriosis with serosa perforation can cause urohemoperitoneum(4,12). In Figure 4, we show the ultrasound image in a patient with stage IV endometriosis, bladder endometrioma and a positive pregnancy test.
6. Ectopic pregnancy
Recent studies comprising meta-analyses of cohort and case-control studies found an increased association between endometriosis and ectopic pregnancy. Interestingly, they could not find sufficient data to draw conclusions about the stage or type of endometriosis, mode of conception and ectopic pregnancy(3).
7. Spontaneous abortion
Studies published in the 1980s suggested that women with endometriosis had an increased risk for miscarriage, but with conflicting results. Recent studies found an increased risk for miscarriage as well, but confounding factors, such as assisted reproductive techniques (ART), adenomyosis and other causes of infertility, were mentioned. Vercellini et al. studied 928 patients and concluded that women with adenomyosis had a 31% increased risk for miscarriage and women without adenomyosis had a 14% increased risk(7,13). Saraswat et al. studied a large cohort of 14,655 patients and found that women with endometriosis had a significantly increased risk for spontaneous abortion compared to those without endometriosis(14). Horton et al., in a more recent and elaborated study, concluded that women with adenomyosis and endometriosis had an increased risk for miscarriage compared to controls(15). The latest ESHRE Guidelines recommend clinicians to be aware of this risk, although the recommendation is based on low/moderate quality data(2).
8. Preterm birth
Women with endometriosis were more likely to have premature rupture of membranes and preterm birth, according to larger studies published between 2009 and 2019 (Fernando et al., 2009; Horton et al., 2019). This was observed in both women with and without ART, but definite conclusions cannot be drawn(15-17).
9. Placenta praevia and obstetric hemorrhage
Placenta praevia was described in many studies about endometriosis in pregnancy. A possible explanation might be an alteration of uterine contractions in women with endometriosis which leads to abnormal blastocyst implantation. Horton et al., in 2019, found that antepartum hemorrhage is with 80% more frequent in women with endometriosis(15). Healy et al., in 2010, concluded that women with endometriosis had a 1.7-fold increased risk for placenta praevia. In more recent studies, the results are contradictory(18). Maggiore et al., in 2016, found no association between endometriosis and placental abruption and antepartum hemorrhage(1). Horton et al. (2019) and Lalani et al. (2018) concluded that the risk is increased for placental abruption, but not in postpartum hemorrhage in women with endometriosis compared to controls(15,19). In Figure 5, we present the intraoperative image in a pregnant patient with deciduosis and endometriosis lesions.
10. Intrauterine growth restriction (IUGR), small for gestational age (SGA) babies and neonatal complications
The studies of Fernando et al. (2009) and Conti et al. (2015) report a two-fold increased incidence of SGA babies in women with endometriosis compared to controls. The possible explanations were placental insufficiency and abnormal placental implantation(17,21). Larger studies, on more than 1.5 million pregnancies (Exacoustos et al., 2015; Stephenson et al., 2016), did not find an association between SGA and endometriosis(21). Mochimaru et al. (2015) found an association between SGA and adenomyosis(23). Recent studies (Horton et al., 2019) did not find an increased risk of SGA and endometriosis(15).
11. Caesarean section
The latest studies found that the incidence of caesarean section is higher in women with endometriosis, possibly due to previous surgical interventions, labor dystocia, malpresentation and ART (Exacoustos et al., 2016; Horton et al., 2019)(15,22). Clinical experience shows that caesarean scar endometriomas are also encountered in some patients with endometriosis who give birth by caesarean section. These patients usually address their physician after two or more years after surgery to investigate the appearance of a round, painful mass located under the scar tissue. In Figure 6, we present the postoperative image of an endometriosis lesion located on the caesarean scar two years after the patients’ first birth by caesarean section.
Conclusions
Endometriosis remains one of the main causes of chronic pelvic pain and general discomfort for women. In clinical practice, patients with a wide range of symptoms, apparently unrelated to a gynecological substrate, actually have endometriosis. There is an increased need for a wider approach of the disease, considering that these women come not only with pelvic pain, but with infertility and psychological problems, as well. Pregnancy is not a cure for the disease, and the new ESRHE Guidelines recommend clinicians not to induce this idea. Pregnancy modifies the aspect of endometriotic lesions and, in some cases, results in adverse outcomes. When possible, it is better to diagnose and approach the lesions before obtaining a pregnancy. The new ESHRE Guidelines, based on clinical findings, recommend clinicians and patients to be aware that complications induced by endometriosis during pregnancy are indeed rare, but potentially severe. To this date, there is no specific protocol for the pregnancy with endometriosis, but more studies are needed to improve the management and quality of life of these patients.
Conflict of interests: The authors declare no conflict of interests.
Bibliografie
-
Maggiore U, Ferrero S, Mangili G, Bergamini A, Inversetti A, Giorgione V, et al. A systematic review on endometriosis during pregnancy: diagnosis, misdiagnosis, complications, and outcomes. Hum Reprod Update. 2016;22:70–103.
-
Becker CM, Bokor A, Heikinheimo O, et al. ESHRE guideline: endometriosis. Hum Reprod Open. 2022;2022(2):hoac009.
-
Ueda Y, Enomoto T, Miyatake T, Fujita M, Yamamoto R, Kanagawa T, et al. A retrospective analysis of ovarian endometriosis during pregnancy. Fertil Steril. 2010;94:78–84
-
Maggiore U, Remorgida V, Sala P, Vellone VG, Biscaldi E, Ferrero S. Spontaneous uroperitoneum and preterm delivery in a patient with bladder endometriosis. J Minim Invasive Gynecol. 2015;22:701–702.
-
Maggiore U, Scala C, Tafi E, Racca A, Biscaldi E, Vellone VG, et al. Spontaneous fertility after expectant or surgical management of rectovaginal endometriosis in women with or without ovarian endometrioma: a retrospective analysis. Fertil Steril. 2017;107:969–76.e5.
-
Vercellini P, Vigano P, Somigliana E, Fedele L. Endometriosis: pathogenesis and treatment. Nat Rev Endocrinol. 2014;a10:261–275.
-
Vercellini P, Consonni D, Dridi D, Bracco B, Frattaruolo MP, Somigliana E. Uterine adenomyosis and in vitro fertilization outcome: a systematic review and meta-analysis. Hum Reprod. 2014b;29:964–977.
-
Setubal A, Sidiropoulou Z, Torgal M, Casal E, Lourencco C, Koninckx P. Bowel complications of deep endometriosis during pregnancy or in vitro fertilization. Fertil Steril. 2014;101:442–6.
-
Villa G, Mabrouk M, Guerrini M, Mignemi G, Colleoni GG, Venturoli S, et al. Uterine rupture in a primigravida with adenomyosis recently subjected to laparoscopic resection of rectovaginal endometriosis: case report. J Minim Invasive Gynecol. 2008;15:360–1.
-
Glavind MT, Forman A, Arendt LH, Nielsen K, Henriksen TB. Endometriosis and pregnancy complications: a Danish cohort study. Fertil Steril. 2017;107:160–6.
-
Lier M, Malik RF, van Waesberghe J, Maas JW, van Rumpt-van de Geest DA, Coppus SF, et al. Spontaneous haemoperitoneum in pregnancy and endometriosis: a case series. BJOG. 2017;124:306–12.
-
Chiodo I, Somigliana E, Dousset B, Chapron C. Urohemoperitoneum during pregnancy with consequent fetal death in a patient with deep endometriosis. J Minim Invasive Gynecol. 2008;15:202–4.
-
Vercellini P, Parazzini F, Pietropaolo G, Cipriani S, Frattaruolo M, Fedele L. Pregnancy outcome in women with peritoneal, ovarian and rectovaginal endometriosis: a retrospective cohort study. BJOG. 2012;119:1538–43.
-
Saraswat L, Ayansina DT, Cooper KG, Bhattacharya S, Miligkos D, Horne AW, et al. Pregnancy outcomes in women with endometriosis: a national record linkage study. BJOG. 2017;124:444–52.
-
Horton J, Sterrenburg M, Lane S, Maheshwari A, Li TC, Cheong Y. Reproductive, obstetric, and perinatal outcomes of women with adenomyosis and endometriosis: a systematic review and meta-analysis. Hum Reprod Update. 2019;25;592-632.
-
Fernando S, Soh PQ, Cooper M, Evans S, Reid G, Tsaltas J, Rombauts L. Reliability of visual diagnosis of endometriosis. J Minim Invasive Gynecol. 2013;20:783-789.
-
Fernando S, Breheny S, Jaques AM, Halliday JL, Baker G, Healy D. Preterm birth, ovarian endometriomata, and assisted reproduction technologies. Fertil Steril. 2009;91:325–30.
-
Healy DL, Breheny S, Halliday J, Jaques A, Rushford D, Garrett C, et al. Prevalence and risk factors for obstetric haemorrhage in 6730 singleton births after assisted reproductive technology in Victoria Australia. Hum Reprod. 2010;25:265–74.
-
Lalani S, Choudhry AJ, Firth B, Bacal V, Walker M, Wen SW, Singh S, Amath A, Hodge M, Chen I. Endometriosis and adverse maternal, fetal and neonatal outcomes, a systematic review and meta-analysis. Hum Reprod. 2018;33:1854-1865.
-
Conti N, Cevenini G, Vannuccini S, Orlandini C, Valensise H, Gervasi MT, et al. Women with endometriosis at first pregnancy have an increased risk of adverse obstetric outcome. J Matern Fetal Neonatal Med. 2014;9:1–4.
-
Exacoustos C, Lauriola I, Lazzeri L, de Felice G, Zupi E. Complications during pregnancy and delivery in women with untreated rectovaginal deep infiltrating endometriosis. Fertil Steril. 2016;106:1129–35.e1.
-
Exacoustos C, Luciano D, Corbett B, DeFelice G, Di Feliciantonio M, Luciano A, Zupi E. The uterine junctional zone: a 3-dimensional ultrasound study of patients with endometriosis. Am J Obstet Gynecol. 2013;209;248.e1–248.e7.
-
Mochimaru A, Aoki S, Oba MS, Kurasawa K, Takahashi T, Hirahara F. Adverse pregnancy outcomes associated with adenomyosis with uterine enlargement. J Obstet Gynaecol Res. 2015;41:529–33.
Articole din ediţiile anterioare
Managementul terapeutic al glioblastomului în timpul sarcinii: prezentare de caz subliniind caracteristicile clinice şi radiologice la diagnostic şi după intervenţie
Heterogeneous malignant cerebral tumors include the following types: anaplastic astrocytoma, glioblastoma multiforme type, gliosarcoma and analplas...
Sarcina şi BPA – o problemă nerezolvată
Bisfenolul A (BPA) a fost clasificat ca disruptor endocrin în ultimii ani şi asistăm la tot mai multe raportări ale efectelor adverse materne şi fe...
Myasthenia gravis şi sarcina
Myasthenia gravis (MG) is an autoimmune disorder affecting nearly 1 million individuals worldwide, being diagnosed typically in the second and thir...
Incidenţa endometriozei şi a endometriozei atipice în cazul tumorilor ovariene epiteliale
Mai multe studii anterioare au identificat o asociere între endometrioză şi dezvoltarea carcinoamelor ovariene. Acest studiu urmăreşte prevalenţa e...