Adăugarea unui compus de platină la taxani în cancerul de sân metastatic şi beneficiul pe care îl determină pentru TNBC. Experienţa unui centru din România
Evaluating the addition of a platinum agent to a taxane in metastatic breast cancer and the benefits for TNBC. A Romanian centre experience
Abstract
Breast cancer is the most frequently diagnosed type of cancer in women worldwide and the leading cause of mortality among them. Its most aggressive subtype is triple-negative breast cancer (TNBC), accounting for almost 15-20% of all breast cancers. There is a rising trend in using platinum agents for treating breast cancer in the neoadjuvant or metastatic setting, with promising results in the TNBC subtype and/or in the presence of BRCA1/2 mutation. A retrospective study was created at the “Prof. Dr. Alexandru Trestioreanu” Institute of Oncology, Bucharest, Romania, with the purpose of evaluating the toxicity of adding carboplatin to a taxane (which already proved to be an active agent for this pathology) and to assess the TTP (time to progression) as main objective for luminal and triple-negative subtypes. We conclude that the addition of carboplatin to paclitaxel increases the haematological and gastrointestinal toxicities, but these are manageable, and the combination can be safely administered in daily practice and it also showed a significant increase in time to progression (mTTP) when used in the first-line treatment, with greater benefit seen in TNBC.Keywords
advanced breast cancerplatinum agentstriple-negative breast cancerretrospective studyRezumat
Cancerul de sân este cel mai frecvent tip de cancer diagnosticat la femeile din întreaga lume şi principala cauză de mortalitate printre acestea. Subtipul său cel mai agresiv este cancerul de sân triplu negativ (TNBC), reprezentând aproape 15-20% din toate cazurile de cancer de sân. Există o tendinţă în creştere în utilizarea agenţilor de platină pentru tratarea cancerului de sân în context neoadjuvant sau metastatic, cu rezultate promiţătoare în subtipul TNBC şi/sau în prezenţa mutaţiei BRCA1/2. A fost derulat un studiu retrospectiv la Institutul de Oncologie „Prof. Dr. Alexandru Trestioreanu”, Bucureşti, România, cu scopul de a evalua toxicitatea adăugării carboplatinei la un taxan (care s-a dovedit deja a fi un agent activ pentru această patologie) şi de a evalua TTP (timpul până la progresie) ca obiectiv principal pentru subtipurile luminale şi triplu negative. Concluzionăm că adăugarea de carboplatină la paclitaxel creşte toxicităţile hematologice şi gastrointestinale, dar acestea sunt uşor de gestionat, iar combinaţia poate fi administrată în siguranţă în practica zilnică, arătând, de asemenea, o creştere semnificativă a timpului până la progresie (mTTP) atunci când este utilizată în tratamentul de primă linie, cu beneficii mai mari observate în TNBC.Cuvinte Cheie
cancer de sân avansatagenţi de platinăcancer de sân triplu negativstudiu retrospectivIntroduction
Breast cancer is the most frequently diagnosed type of cancer in women worldwide and a vast number of women ask for a doctor’s appointment in the advanced stages of the disease or progress despite the neo-/adjuvant therapy. Its most aggressive subtype is triple-negative breast cancer (TNBC), accounting for almost 15-20% of all breast cancers. Triple-negative means that, to date, there is no targeted treatment useful in this setting because the tumors don’t express estrogen or progesterone receptors, nor HER2 overexpression(1). Thus, a solution needs to be found, and there is a rising trend in using platinum agents for treating breast cancer, with promising results in the TNBC subtype and/or in presence of BRCA1/2 mutation, especially because the carriers of these mutations develop TNBC often(2) and so they are expected to respond better to cytotoxic therapy that acts by damaging DNA and leads to cell apoptosis, such as platinum agents. Recent trials were conducted in the neoadjuvant setting (phase II GeparSixto evaluating the addition of carboplatin to standard chemotherapy in TNBC) and metastatic stage (phase III TNT trial comparing carboplatin to docetaxel for unselected TNBC patients and BRCAness subgroup) which reminded us about the added value of using platinum agents in breast cancer.
Initially, platinum agents were investigated in the early 1970s in the metastatic setting, with good response rates (comparable to the ones we see in present clinical research in this area)(3), and various publications subscribe to the idea of using platinum agents in different settings also. A meta-analysis performed by Fausto Petrelli et al. compared the results of 28 studies on this matter and showed a pooled rate of 45% increase in pCR (pathologic complete response) rates among patients with TNBC to whom platinum agents were added to standard neoadjuvant chemotherapy regimes (including the aforementioned phase II GeparSixto trial)(4). As for metastatic TNBC, a retrospective study was conducted in Canada by Villarreal-Garza et al., which compared platinum-based chemotherapy (PBC) to non-PBC. In terms of overall survival (OS), the results were in favour of PBC (14.5 versus 10 months; p=0.041)(5).
Back then, in the late 1980s, newer cytotoxic agents were also discovered and started gaining approval for the treatment of various types of cancer: taxanes, paclitaxel and docetaxel (for example, docetaxel was approved by FDA in 1995 for the treatment of advanced breast cancer and was used in a continuous fashion ever since)(6), with response rates of 30-40% in all published data unrelated to any known breast cancer mutation. This was also concluded by a retrospective analysis of three clinical studies performed at the M.D. Anderson Hospital, Texas, USA, that showed a median progression-free survival with paclitaxel alone of 9 months in women with advanced breast cancer, demonstrating that paclitaxel is also an active agent in metastatic breast cancer (MBC)(7).
Thus, a fair question is raised: would combining a platinum agent and a taxane be better in terms of time to progression, response rates, survival, and how to balance the benefit-to-risk ratio in favour of the patient taking into consideration the toxicity of each agent? When chemotherapy is indicated, according to the ABC guidelines, sequential monotherapy treatment is preferred for metastatic breast cancer, with less side effects than a combination of any kind and with similar outcomes in terms of OS(8). In clinical practice, at the “Prof. Dr. Alexandru Trestioreanu” Institute of Oncology, Bucharest (IOB), Romania, many MBC patients receive combination chemotherapy, especially platinum-taxane regimes, despite the recommendation, irrespectively of the histological types of disease. The reasons for the combination are: the mechanism of action is different, no cross-resistance has ever been reported, and the activity in breast cancer is well documented for both agents(6). The purpose of this study is to evaluate the toxicity of adding carboplatin to a taxane and to assess the TTP (time to progression) as main objective.
Methodology
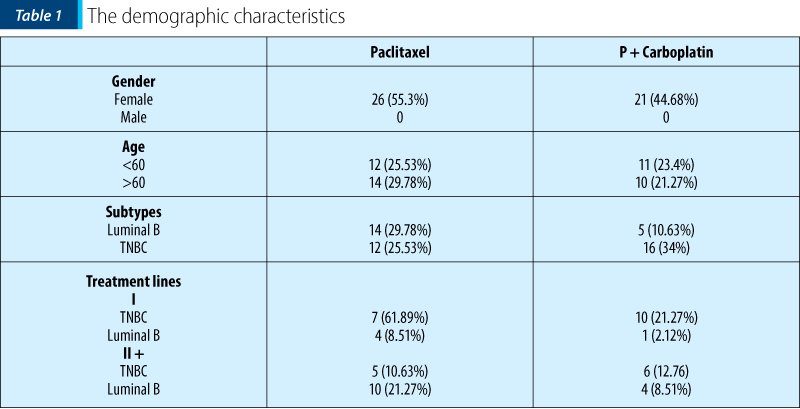
A number of 47 patients with MBC, treated between 2010 and 2018 in IOB, with TNBC or Luminal B (HER 2-) disease, were retrospectively evaluated, divided into two cohorts: A) 26 patients treated with paclitaxel (P) 80 mg/sqm weekly or P 175 mg/sqm every three weeks as monotherapy, and B) 21 patients treated with P 175 mg/sqm plus carboplatin AUC 5/6 every three weeks. Twenty-two patients were treated in the first-line setting. The patients were analyzed according to treatment arms and followed-up until disease progression after paclitaxel-containing therapy with or without a platinum agent (carboplatin). TTP was defined as period of time between starting the respective treatment and ending time based on radiologic/clinical disease progression or unaccepted toxicity(9). TTP was assessed using Kaplan-Meier methodology on evanmiller.org/ab-testing/survival-curves.html and compared between cohorts and different treatment lines/histology by using the T-Test.
The toxicity during treatment was assessed using NCI CTCAE 5.0 criteria for evaluating adverse events (AE). The incidences of various grades of AEs were expressed as percentages from the study population and were compared using “N-1” Chi-squared test. The confidence intervals (CI) for reported toxicities were calculated using the recommendations by Altman et al. on medcalc.org.
The study was approved by the Institutional Ethical Committee. No specific ICF was used, being a retrospective analysis, but all patients signed the institutional ICF, giving consent on the full use of their medical records for research purposes.
Results
The demographic characteristics of the study population are shown in Table 1.
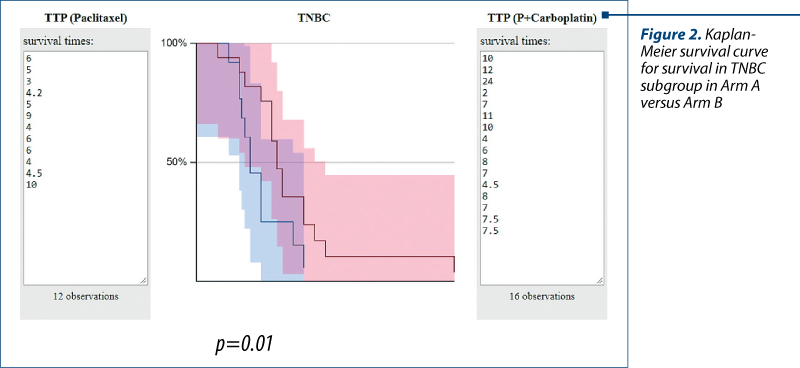
The evaluation of the median TTP (months), irrespective of subtype, in the first line for cohort A was 6.27 months (3-12) versus 10.13 months (4.5-24), p=0.04, for cohort B (Figure 1). The mTTP in the TNBC group cohort A (12 patients) was 5.55 months (4.2-10) and for the TNBC group in cohort B (16 patients), 8.03 months (2-24), p=0.01 (Table 2).
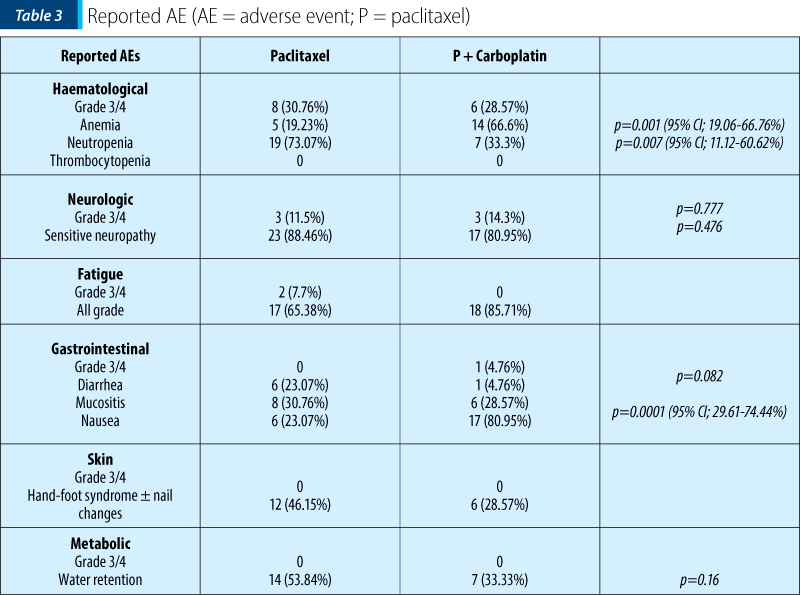
Regarding the toxicity profiles, 30.7% of patients in group A experienced grade 3/4 haematological adverse events, as compared to 28.6% in cohort B; 19% had anaemia (all grade) in the monotherapy arm, compared to 66.6% in the combination arm, p=0.001 (95% CI; 19.06-66.76%). Neutropenia (all grades) was higher with taxane alone (73% versus 33%; p=0.007; 95% CI; 11.12-60.62%). Nausea (all grades) was higher in the combination cohort (81% versus 23%, p=0.0001; 95% CI; 29.61-74.44%), while neurotoxicity grade 3/4 was almost similar, 11.5% in cohort A versus 14.3% in cohort B, p=0.777. The metabolic toxicities reported as water retention were higher in the monotherapy group (53.84% versus 33.33%; p=0,16). No significant difference between the cohorts was observed for the other reported AEs (gastrointestinal, skin, fatigue). All data are presented in Table 3.

Discussion
As expected, the addition of carboplatin to a taxane (paclitaxel) had a better mTTP when used in the first line, it was better observed in the TNBC subgroup (Figure 2), and the result subscribes to all data presented before in clinical trials and mentioned above. Luminal B subgroup had almost the same mTTP with monotherapy (paclitaxel) versus combination: 7 months versus 7.5 months.
The benefit for TNBC patients was seen in unselected population, unrelated to BRCA status, and was given by adding the platinum agent, probably due to its mechanism of action and the similar phenotype which TNBC shares with breast cancer patients with BRCA mutations for which we have strong data to suggest using a platinum agent.
A retrospective analysis conducted at the Curie Institute, Paris, by L. Staudacher et al., included 143 patients with metastatic breast cancer treated with platinum-based chemotherapy. A better response rate was observed in the TNBC arm, but with no improvement on OS or substantially on PFS versus non-TNBC: TTP was 8 months for TNBC and 7 months for non-TNBC. As we can acknowledge, mTTP was the same as in our retrospective analysis for TNBC and also for Luminal B – 7.5 months(10).

A meta-analysis conducted by Miao Liu et al. showed a mPFS in three metastatic breast cancer studies of 6 to 12 months for TNBC when a platinum agent was added, which subscribes to our results(1).
A phase II trial conducted by Perez et al. included 53 patients with metastatic breast cancer for the first-line treatment with paclitaxel and carboplatin and evaluated their response and treatment toxicity. The study concluded a 7.3 months of mTTP in unselected population and a tolerable toxicity profile(12). If it were to be made a direct comparison with our retrospective study, the mTTP in the first line, irrespectively of tumor subtype, showed a benefit of 3 months over the US study, and this could be due to the selection bias and the low number of participants.
In her paper on the value of paclitaxel in breast cancer, A.E. Perez also evaluated the combination of the two drugs, carboplatin and paclitaxel, comparing the results from various clinical trials in which this combination was used. As we could also conclude from our small retrospective study, the toxicity profile of the combined therapy was tolerable, with less haematological side effects than expected, the grade 3-4 haematological AEs being almost similar in the two cohorts. Based on three retrospective trials performed in USA, A.E. Perez concluded that combining carboplatin with paclitaxel did not add extraneurotoxicity or leukopenia/thrombocytopenia than a patient on monotherapy would have expected(12). Carboplatin is known to have less neurological side effects than cisplatin and this didn’t overlap with the well documented neurotoxicity which taxanes have, and this is also observed in our study, neurotoxicity being almost the same between the two cohorts.
Decatris et al. talked about a protective effect on the platelet count with the combination therapy, as in any of the trials he evaluated retrospectively, severe thrombocytopenia was not observed(3). This situation was also seen in our study, as no thrombocytopenic AEs was reported.
Conclusions
We conclude that the addition of carboplatin to paclitaxel increases the haematological and gastrointestinal toxicities, as it was acknowledged in all studies from literature, but not as high as expected. They are manageable and the combination can be safely administered in daily practice.
Although the selection bias cannot be accounted for, in our limited experience, the carboplatin plus taxane combination showed a significant increase in time to progression when used in the first-line treatment for MBC. Further research is needed, through a prospective randomised comparative trial to define the real benefit and toxicity of adding platinum agents in the first line to paclitaxel monotherapy in metastatic breast cancer.
Conflicts of interests: The authors declare no conflict of interests.
Bibliografie
- Liu M, Mo QG, Wei CY, Qin QH, Huang Z, He J. Platinum-based chemotherapy in triple-negative breast cancer: A meta-analysis. Oncol Lett. 2013; 5(3):983-991. doi:10.3892/ol.2012.1093
- Sørlie T, Tibshirani R, Parker J. Repeated observation of breast tumor subtypes in independent gene expression data sets. PNAS. 2003, July 8; 100 (14) 8418-842. https://doi.org/10.1073/pnas.0932692100
- Decatris MP, Sundar S, O’Byrne KJ. Platinum-based chemotherapy in metastatic breast cancer: current status. Cancer Treat Rev. 2004; 30(1):53-81. doi:10.1016/S0305-7372(03)00139-7.
- Petrelli F, Coinu A, Borgonovo K, et al. The value of platinum agents as neoadjuvant chemotherapy in triple-negative breast cancers: a systematic review and meta-analysis. Breast Cancer Res Treat. 2014; 144: 223. https://doi.org/10.1007/s10549-014-2876-z
- Villarreal-Garza C, Khalaf D, Bouganim N, et al. Platinum-based chemotherapy in triple-negative advanced breast cancer. Breast Cancer Res Treat. 2014; 146, 567-572. https://doi.org/10.1007/s10549-014-3033-4
- Ojima I, Lichtenthal B, Lee S, Wang C, Wang X. Taxane anticancer agents: a patent perspective. Expert Opin Ther Pat. 2016; 26(1):1-20.
- Miller KD, Sledge GW. Taxanes in the Treatment of Breast Cancer: A Prodigy Comes of Age. Cancer Investigation. 1999; 17:2, 121-136. DOI: 10.1080/07357909909011727
- Cardoso F, Senkus E, Costa A, Papadopoulos E, Aapro M, André F, Harbeck N, Aguilar Lopez B, Barrios CH, Bergh J, Biganzoli L, Boers-Doets CB, Cardoso MJ, Carey LA, Cortés J, Curigliano G, Diéras V, El Saghir NS, Eniu A, Fallowfield L, Francis PA, Gelmon K, Johnston SRD, Kaufman B, Koppikar S, Krop IE, Mayer M, Nakigudde G, Offersen BV, Ohno S, Pagani O, Paluch-Shimon S, Penault-Llorca F, Prat A, Rugo HS, Sledge GW, Spence D, Thomssen C, Vorobiof DA, Xu B, Norton L, Winer EP. 4th ESO-ESMO International Consensus Guidelines for Advanced Breast Cancer (ABC 4). Annals of Oncology. 2018 Aug; 29(8):1634–1657. https://doi.org/10.1093/annonc/mdy192
- Saad ED, Katz A. Progression-free survival and time to progression as primary end points in advanced breast cancer: often used, sometimes loosely defined. Annals of Oncology. 2009 March; 20(3):460–464. https://doi.org/10.1093/annonc/mdn670
- Staudacher L, et al. Platinum-based chemotherapy in metastatic triple-negative breast cancer: the Institut Curie experience. Annals of Oncology. 2011 Apr;22(4):848–856.
- Perez EA. Paclitaxel in Breast Cancer. The Oncologist. 1998; 3:373-389.
- Perez EA, Hillman DW, Stella PJ, et al. A phase II study of paclitaxel plus carboplatin as first-line chemotherapy for women with metastatic breast carcinoma. Cancer. 2000; 88(1):124-131.





