Pelvic exenteration – between history and future
Exenteraţia pelviană – între istorie şi viitor
Abstract
Introduction. Advanced pelvic neoplasms are often real trials for oncologists, radiation therapists and oncological surgeons. The treatment must be individualized for each patient and it sometimes involves extensive exenterative interventions burdened with significant morbidity. Although known for more than 80 years, pelvic exenterations have undergone countless technical changes over time which led to a decrease in mortality and to improved survival and quality of life parameters. Materials and method. In this paper, we aim to carry out a review of the international publications dedicated to pelvic exenterations, following the changes undergone by the surgical technique and their impact on survival, morbidity associated with the method and on perioperative mortality. Results. The advances in surgical techniques allowed the extension of the resection in three compartments: posterior (through resection of the sacrum), lateral (through extended muscle resections, iliac vessel resections followed by various reconstructive techniques, nerve resections) and anterior (through pubic or perineal urethra resections). The development of urinary-genital reconstruction techniques also contributed to the improvement of the patients’ quality of life. Restoring the pelvic floor by using the greater omentum or other materials like breast implants, biodegradable meshes, dura mater or bladder peritoneum causes a decrease in the complications associated with the “empty pelvis syndrome”, responsible for the majority of post-pelvectomy morbidity. Finally, minimally invasive and robotic techniques cause a decrease in intraoperative blood loss and duration of hospitalization, at the cost of a longer duration of the intervention. Conclusions. The pelvectomy, an operation with a long history, in continuous reinvention and upgrading, still plays an important role in the therapy of advanced primary or recurrent pelvic cancers, being sometimes the only therapeutic method that can increase survival. However, it requires a good selection of patients and must be performed in a specialized center in order to achieve the best results and to avoid complications.Keywords
pelvectomypelvic exenterationradical pelvic surgerysacrectomyvascular resectioncomposite pelvectomyRezumat
Introducere. Neoplasmele pelviene avansate constituie de multe ori adevărate încercări pentru medicii oncologi, radioterapeuţi şi chirurgi oncologi. Tratamentul trebuie particularizat fiecărui pacient şi uneori implică intervenţii exenterative de mare amploare, grevate de o morbiditate importantă. Deşi cunoscute de peste 80 de ani, exenteraţiile pelviene au suferit de-a lungul timpului nenumărate modificări tehnice ce au determinat scăderea mortalităţii şi îmbunătăţirea parametrilor de supravieţuire şi calitate a vieţii. Materiale şi metodă. În această lucrare, ne propunem să efectuăm o trecere în revistă a publicaţiilor internaţionale dedicate exenteraţiilor pelviene, urmărind modificările suferite de tehnica chirurgicală şi impactul acestora asupra supravieţuirii, morbidităţii asociate metodei şi a mortalitaţii perioperatorii. Rezultate. Progresele tehnicilor chirurgicale au permis extensia rezecţiei în compartimentele posterior (prin rezecţia sacrului), lateral (prin rezecţii extinse musculare, rezecţii de vase iliace urmate de tehnici reconstructive diverse, rezecţii nervoase) şi anterior (prin rezecţii pubiene sau ale uretrei perineale). Dezvoltarea tehnicilor de reconstrucţie urogenitală a contribuit, de asemenea, la îmbunatăţirea calităţii vieţii pacienţilor. Refacerea planşeului pelvian folosind marele epiploon sau alte materiale (implanturi mamare, meşe biodegradabile, dura mater, peritoneu vezical) determină o scădere a complicaţiilor asociate cu „sindomul de pelvis gol”, răspunzător de majoritatea morbidităţii postpelvectomie. În final, tehnicile minim invazive şi robotice determină o scădere a pierderilor de sânge intraoperatorii şi a duratei de spitalizare, cu preţul unei durate mai lungi a intervenţiei. Concluzii. Pelvectomia, operaţie cu o lungă istorie, aflată într-o continuă reinventare şi îmbunătăţire, încă joacă un rol important în terapia cancerelor pelviene avansate primare sau recurente, fiind uneori singura metodă terapeutică ce poate duce la creşterea supravieţuirii. Operaţia necesită totuşi o bună selecţie a pacienţilor şi efectuarea procedurii într-un centru specializat, pentru a obţine cele mai bune rezultate şi a evita complicaţiile.Cuvinte Cheie
pelvectomieexenteraţie pelvianăchirurgie pelviană radicalăsacrectomierezecţie vascularăpelvectomie compozităIntroduction
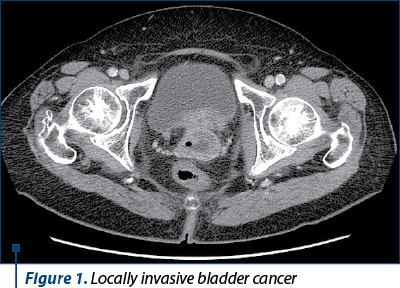
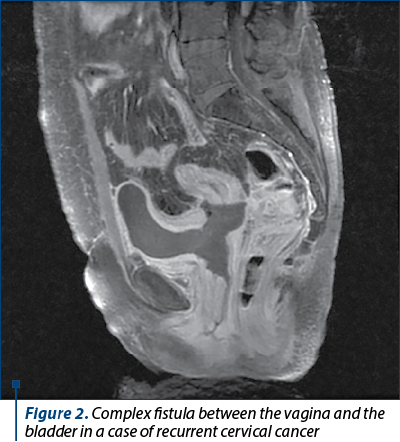
Pelvic exenteration, defined as the total or partial excision of the pelvic organs toghether with the corespondent lymph nodes, often remains the only surgical solution for certain types of locoregionally advanced primary or recurrent cancers (in Figures 1 and 2, we present typical cases with indication for pelvectomy). With a history of more than 80 years, this procedure has undergone numerous technical changes to adapt it to the goal of radicality. Although numerous medical advances have contributed to the gradual decrease in recorded mortality, the morbidity associated with this procedure remains high. However, we believe that resection is justified even in the case of recurrent pelvic localized cancers, due to the fact that, in some cases, surgery is the only therapeutic alternative that can provide a potential chance of increased survival and – why not – of cure, in happy cases.
Materials and method
In the present work, we aim to carry out a review of the international publications dedicated to pelvic exenterations, following the changes undergone by the surgical technique and their impact on survival, morbidity and perioperative mortality associated with the method.
Discussion
By the late 1940s, most of the radical interventions we perform today for cancer had already been envisioned and successfully performed. Medical advances achieved after World War II (including transfusion technique, anesthesia and intensive care, antibiotic therapy, intra- and postoperative monitoring, nutrition) made possible multiple major radical interventions that later became the standard of care for neoplastic diseases. Also, at the end of the 19th century and the beginning of the 20th century, operations preceding pelvectomies – radical cystectomy (Verhoogen, 1908), radical hysterectomy (Weirtheim, 1898) and abdominoperineal rectal resection (Miles, 1908) had already been described and successfully practiced. Thus, the set-up was optimal for the year 1948 when Al. Brunschwig published a series of 22 cases in which he practiced total pelvic exenteration, that was defined as the complete resection of the pelvic viscera and related lymphatic drainage systems(1). Although the pelvectomy was considered “the most radical surgical attack on gynecological cancers”, the procedure was accompanied by a significant mortality (22.7%) and a corresponding morbidity.
Even before Brunschwig’s writing, such interventions have already been carried out. Eugene M. Bricker performed his first total exenteration in 1940, on a 32-year-old woman with persistent carcinoma of the cervix. He observed that certain types of pelvic cancers tend to remain confined to the pelvic organs for extended periods of time before metastasizing beyond the pelvic lymph nodes. These include well-differentiated squamous cell carcinoma of the cervix, vagina and vulva, rectosigmoid adenocarcinoma and uterine sarcoma. He also found that ovarian, prostatic and – to a lesser extent – bladder and endometrial cancers had spread beyond the pelvis when adjacent viscera were involved. In the 1960s, Spratt et al. also confirmed these observations(2).
If in 1952, Chiricuţă called the pelvectomy “the surgery of despair”. In the hands of experienced surgeons, such as Brunschwig, Appleby, Britnall, Flocksand and many others, pelvic exenteration has gradually transformed from an experiment reserved for desperate clinical situations into a possibility of healing for selected patients with advanced pelvic cancers, otherwise incurable. Bricker developed in the 1950s the ileal conduit for post-pelvectomy urinary diversion, this technique representing a major advance in pelvic surgery(3). At the same time, medical advances made the operation safer, with the mortality rate falling from over 20% in the 1940-1960s to less than 10% in the 1970s. The continuing interest in the subject led in 1973 to the first comprehensive publication on exenterative surgery of the pelvis that presented a series of over 500 cases (Spratt et al., 1973)(4).
Between the years 1990 and 2010, multiple series of patients’ cases were published in whom pelvic exenterations were performed for various locally advanced neoplasias, noting the reduction of mortality rates (both global and perioperative – the latter tending towards 0), with a maintained risk of morbidity. Reduced operative mortality rates have allowed surgeons to safely extend the operation to include portions of the bony pelvis(6-8). Even when the exenteration is extended to include bony portions of the pelvis (the so-called “composite” pelvic exenteration), the morbidity remains unchanged, while the mortality tends toward 0%.
Medical developments have placed this radical operation in a different perspective. Pelvic exenteration, today, should be seen as an operation with potential healing, and not only as a palliative effort. Publications on pelvectomies attest to the increasing use of this procedure internationally, even in the community hospitals(9-12).
The efforts made to perform curative resections, especially in cases of tumor recurrence in already treated patients, have been pushed to new limits in the last decades, including new anatomical structures, that previously prohibited the resection. The importance of R0 resection on survival is highlighted in numerous publications and meta-analyses(13). Clear resection margins have been shown to be the most important predictor of both long-term survival and subsequent quality of life of patients. For this reason, obtaining a resection without residual microscopic tumor became the primary goal of radical pelvectomies(13,14). The extension of the resection targeted three compartments that we will describe below: the posterior compartment, the lateral compartment, and the anterior compartment.
Posterior compartment
The first attempts at posterior extension of the pelvectomy were made by Brunschwig in the 1960s. However, the technique was abandoned until the 1980s due to associated mortality and morbidity(15,16). The treatment of neoplasias infiltrating the presacral fascia should include “en bloc” sacrectomy rather than trying to skin off the fascia from the bone surface, due to the risk of microscopic residual tumor(17).
Multiple sacral resection techniques have been described: sacrectomy by abdomino-lithotomy (a procedure used when the bone trans-section level is above the saco-iliac joint and which provides very good access to the lateral pelvic wall and to the vasculo-nervous structures from this level), ventral decubit sacral resection after performing the abdomino-perineal time (a procedure used when the bony cross-sectional plane is found below S3), segmental sacrectomy (in which an initial posterior disconnection of the affected sacral segments is performed and then followed by a completion excision by abdomino-lithotomy, thus avoiding high sacrectomy, which makes it possible to preserve unaffected nerve roots and reduce post-procedural sacro-pelvic instability(18)).
Sacral resection techniques are associated with an R0 resection rate of over 70%, which translates into a five-year survival of 35% in patients who undergo this procedure for reccurent rectal cancer. The price is a major morbidity of about 40%, but with 0% mortality(17). The level of the bone trans-section does not influence the probability of achieving an R0 resection, acording to the same authors. The Mayo Clinic even published a study on a group of nine patients with recurrent rectal neoplasm who underwent sacrectomy associated with pelvic exenteration, R0 resection being possible in all cases, a fact translated into a rate of median survival of 31 months.
Lateral compartment
Lateral neoplastic extension raises the most difficult problems regarding the surgical technique, due to the proximity of major pelvic vascular-nervous structures, which associates an increased risk of neurological dysfunctions and catastrophic bleeding, especially in cases where exenterative efforts are carried out in the context of previous interventions or after pelvic tissue irradiation. In most centers, lateral neoplastic extension remains a relative contraindication to pelvectomy(19-21).
However, techniques of proximal ligatures of the internal iliac vessels followed by “en bloc” resection of muscular (piriformis and internal obturator muscles), bony (sciatic spine) or nerve (the sciatic nerve) structures have been described, which allow moving the resection to a lateral plane, unaltered by previous interventions or irradiation. Adopting such techniques, case series were reported in which R0 resection was possible in 66.5% of cases, a fact translated into a median survival rate of 41 months(22).
When the process also affects the external iliac vessels, they can also be resected, the subsequent arterial reconstruction being done by various techniques of vascular surgery with autologous or synthetic grafts(23,24). As a rule, it is not necessary to restore the external iliac vein, because it is frequently thrombosed preoperatively and the collateral circulation could be developed over time.
The extensive excision of the lateral compartment results in a significant morbidity of almost 30%(22). However, we note the fact that in patients in whom resection of the external iliac vessels is necessary, the morbidity reaches 50%, being due to complications specific to vascular grafts, 24% of cases requiring reintervention. However, the non-functionality of the grafts occurs in few cases, the amputation of the lower limb being rarely necessary(23).
Complex vascular techniques have also been described in the case of pelvectomies performed for palliative purposes, with the aim of improving the patients’ symptoms. A publication from Turkey describes a total pelvectomy performed on a 35-year-old female patient with locally advanced persistent cervical neoplasm and associated with significant lower limb edema and pain not controlled by medication at this level. The intervention was associated with an arterial and venous ileo-femoral bypass, which resulted in the improvement of local pain and the dramatic decrease in the circumference of the pelvic limb from 75 to 44 cm, postponing the amputation for five months(25). Although the indications for palliative pelvectomies are controversial, they can be considered according to the needs of each patient, after realistically discussing the prospects with the patient in question.
Anterior compartment
Anterior extension of the resection is most often required in male patients with recurrent rectal cancer in which the dissection of the primary tumor was performed very close to the prostate. In such cases, to achieve an R0 resection, the surgeon is obliged to approach the urethra perineally and resect it “en bloc” with the pubic bone (the latter being the subject of a total or partial resection)(17).
The feasibility of pubic bone resection techniques in pelvectomies was proven in a series of 29 patients, where R0 resection was achieved in 76% of cases, with an overall survival rate of 53%(26).
Although accompanied by significant morbidity, these anterior extensions of pelvectomies, like posterior and lateral extensions, represent a curative treatment option for carefully selected patients operated in specialized centers.
Urogenital and pelvic floor reconstruction
With advances in overall survival, considerations of body image and socio-familial integration of patients have been raised. Patients’ expectations about social relationships, professional life, sexuality, recreational activities, as well as the acceptance of stomas influenced by individual (gender, culture, geographical origin(27)) and socioeconomic factors (availability and compensation by insurance systems of materials necessary for stoma care) will determine the choice of reconstructive techniques. Numerous techniques have been developed and enrich the therapeutic arsenal.
For the evacuation of urine after pelvectomies, urinary diversions externalized directly through the skin or indirectly through a digestive segment (ureterostomy or nephrostomy) were used. However, these are not considered reconstructive techniques. Among the reconstructive techniques, we mention the ureterosigmoidostomy (described by Coffey in 1934(28)), the “double barrel” wet colostomy technique (originally described by Brunschwig and adapted by Carter in 1989 to reduce severe complications such as pyelonephritis and metabolic imbalance(29)), the ileal Bricker(30), “self-catheterizing” continent bag Miami, Indiana or Mainz type(31-33), and orthotopic neobladder(34).
It has recently been suggested that indocyanine green can be used to assess the vascularity of urinary-digestive anastomoses and, thus, play a role in assessing the risk of complications in patients undergoing such a reconstructive technique(35).
Vaginal reconstruction is usually performed using two techniques: a musculoperitoneal composite transversus muscle and rectus abdominis (TRAMP) or a rectus abdominis inverted inferior musculocutaneous flap (TRAM). The second method is credited with a lower complication rate(36).
A good part of the complications after pelvic exenteration is due to the so-called “empty pelvis syndrome”, defined as all the symptoms that appear as a direct result of the empty space resulting from the resection of the pelvic organs, including here the formation of abscesses, hematomas, fistulizations or obstructions of intestinal loops trapped in the pelvis, dehiscences of the perineal wound, hernias and damage to the viscera descending into the pelvis from subsequent treatments addressed to neoplastic disease such as postradicular enteritis.
To avoid these complications, a whole series of reconstructive techniques have been devised using various materials to fill the pelvic cavity: the great omentum(37), mammary implants(38), musculocutaneous flaps(39-42), dura mater(43) biodegradable mesh(44) or bladder peritoneum(45).
Minimally invasive and robotic surgery
With the publication by Lim in 2009 of the first robotic-assisted laparoscopic total pelvic exenteration(46), the path is opened to the minimally invasive approach of advanced pelvic neoplasias. Although the surgeon must operate in narrow pelvic spaces, often dissecting fibrotic and fragile tissues previously subjected to radiation therapy or surgery, robotic technology can help the surgeon cope with these difficult conditions, especially thanks to the 3D vision, articulated movement, ergonomic improvements and stabilization of tremor.
A 2018 meta-analysis by the Pelvex Collaborative(47), comparing minimally invasive and open abdominal pelvic exenterations, revealed a shorter operative time of an average of 83 minutes in favor of the open approach, while the blood loss (median 550 versus 2300 ml) and hospitalization (22 versus 28 days; p=0.04) were shorter in favor of the minimally invasive approach.
However, at the present time, we believe that additional studies are needed to clarify the role of minimally invasive and robotic techniques of pelvectomy.
Conclusions
Although the extremely advanced pelvic cancers commonly observed in the past decades have become much rarer as a result of the implementation of early cancer detection and treatment practices, pelvic exenteration, with its many technical variations, retains an important place in treatment of advanced pelvic malignancies or recurrences and in selected cases with complications of neoplastic diseases, remaining for certain patients the only available resource.
Pelvic exenteration is indicated as primary or salvage therapy for locally advanced pelvic cancers that have not metastasized beyond the anatomical limits of resection. Case selection is critical to the success of the procedure, given the significant associated morbidity and the patient’s subsequent quality of life which may be greatly diminished.
As early as 1950, Bricker(3) stated that “if we cannot leave a patient in a functional state compatible with a comfortable existence, we cannot morally justify performing this operation”. Any factor which may have a negative impact on postoperative function, such as lack of mobility, inability to self-care, and dependence on peers, must be taken into account. These social factors must be carefully evaluated in repeated discussions with the patient and his/her family to ensure that everyone realistically understands the implications of the proposed operation(48).
However, accepting the costs related to morbidity and quality of life, pelvectomy remains the only option in selected cases that can ensure the long-term survival.
Conflicts of interests: The authors declare no conflict of interests.
Bibliografie
-
Brunschwig A. Complete excision of pelvic viscera for advanced carcinoma: a one-stage abdominoperineal operation with end colostomy and bilateral ureteral implantation into the colon above the colostomy. Cancer. 1948;1(2):177–83.
-
Spratt JS, Watson FR, Pratt JL. Characteristics of variants of colorectal carcinoma that do not metastasize to lymph nodes. Dis Colon Rectum. 1970;13(3):243–6.
-
Bricker EM. Bladder substitution after pelvic evisceration. Surg Clin North Am. 1950;30(5):1511–21.
-
Spratt JS, Butcher HR, Bricker EM. Exenterative surgery of the pelvis. Philadelphia: WB Saunders; 1973.
-
Dias AR, Nahas RC Modified supralevator pelvic exenteration for the treatment of locally advanced rectal cancer with vaginal and uterine invasion, Surg Today. 2013 Jun;43(6):702-4. doi: 10.1007/s00595-012-0298-2.
-
Lopez MJ, Luna-Pérez P. Composite pelvic exenteration: is it worthwhile? Ann Surg Oncol. 2003;11(1):27–33.
-
Wanebo HJ, Gaker DL, Whitehill R, et al. Pelvic recurrence of rectal cancer. Ann Surg. 1987; 205:482–94.
-
Yamada K, Ishizawa T, Kiyoshi N, et al. Pelvic exenteration and sacral resection for locally advanced primary and recurrent rectal cancer. Dis Colon Rectum. 2002;45(8):1078–84.
-
Kecmanovic DM, Pavlov MJ, Kovacevic PA, et al. Management of advanced pelvic cancer by exenteration. Eur J Surg Oncol. 2003;29(9):743–6.
-
Chen HS, Sheen-Chen SM. Total pelvic exenteration for primary local advanced colorectal cancer. World J Surg. 2001;25(12):1546–9.
-
Wiig JN, Poulsen JP, Larsen S, et al. Total pelvic exenteration with preoperative irradiation for advanced primary and recurrent rectal cancer. Eur J Surg. 2002;168(1):42–8.
-
Vitelli CE, Crenca F, Fortunato L, et al. Pelvic exenterative procedures for locally advanced or recurrent colorectal carcinoma in a community hospital. Tech Coloproctol. 2003;7(3):159–63.
-
Simillis C, Baird DL, Kontovounisios C, et al. Systematic Review to Assess Resection Margin Status After Abdominoperineal Excision and Pelvic Exenteration for Rectal Cancer. Ann Surg. 2017;265:291-9
-
Young JM, Badgery-Parker T, Masya LM, et al. Quality of life and other patient-reported outcomes following exenteration for pelvic malignancy. Br J Surg. 2014;101(3):277-87.
-
Wanebo HJ, Marcove RC. Abdominal sacral resection of locally recurrent rectal cancer. Ann Surg. 1981;194(4):458-71.
-
Takagi H, Morimoto T, Kato T, et al. Pelvic exenteration combined with sacral resection for recurrent rectal cancer. J Surg Oncol. 1983;24(3):161-6.
-
Yee Chen Lau, Kilian G.M. Brown, Peter Lee Pelvic Exenteration for locally advanced and recurrent rectal cancer – How much more? Journal of Gastrointestinal Oncology. 2019;10(6):1207-14.
-
Brown KG, Solomon MJ, Austin KK, et al. Posterior high sacral segmental disconnection prior to anterior en bloc exenteration for recurrent rectal cancer. Tech Coloproctol. 2016;20(6):401-4.
-
Rahbari NN, Ulrich AB, Bruckner T, et al. Surgery for locally recurrent rectal cancer in the era of total mesorectal excision: is there still a chance for cure? Ann Surg. 2011;253(3):522-33.
-
Pawlik TM, Skibber JM, Rodriguez-Bigas MA. Pelvic exenteration for advanced pelvic malignancies. Ann Surg Oncol. 2006;13:612-23.
-
Mirnezami AH, Sagar PM, Kavanagh D, et al. Clinical algorithms for the surgical management of locally recurrent rectal cancer. Dis Colon Rectum. 2010;53(9):1248-57.
-
Solomon MJ, Brown KG, Koh CE, et al. Lateral pelvic compartment excision during pelvic exenteration. Br J Surg. 2015;102(13):1710-7
-
Brown KG, Koh CE, Solomon MJ, et al. Outcomes After En Bloc Iliac Vessel Excision and Reconstruction During Pelvic Exenteration. Dis Colon Rectum. 2015;58(9):850-6.
-
Brown KG, Koh CE, Solomon MJ, et al. Spiral saphenous vein graft for major pelvic vessel reconstruction during exenteration surgery. Ann Vasc Surg. 2015;29(6):1323-6.
-
Tatar B, Yalçın Y, Erdemoğlu E. Palliative pelvic exenteration using iliofemoral bypass with synthetic grafts for advanced cervical carcinoma. Turk J Obstet Gynecol. 2019;16(1):80-83.
-
Austin KK, Herd AJ, Solomon MJ, et al. Outcomes of Pelvic Exenteration with en Bloc Partial or Complete Pubic Bone Excision for Locally Advanced Primary or Recurrent Pelvic Cancer. Dis Colon Rectum 2016;59(9):831-5.
-
Simmons KL, Maekawa A, Smith JA. Culture and psychosocial function in British and Japanese people with an ostomy. J Wound Ostomy Continence Nurs. 2011;38(4):421–7.
-
Coffey RC. Transplantation of the ureters into the large intestine.Submucous implantation method. Personal studies and experiences. Br J Urol [Internet]. 1931;3:353–428.
-
Carter MF, Dalton DP, Garnett JE. Simultaneous diversion of the urinary and fecal streams utilizing a single abdominal stoma: the double-barreled wet colostomy. J Urol. 1989;141(5):1189–91.
-
Bricker EM. Bladder substitution after pelvic evisceration. Surg Clin North Am. 1950;30(5):1511–21.
-
Bejany DE, Politano VA. Stapled and nonstapled tapered distal ileum for construction of a continent colonic urinary reservoir. J Urol. 1988;140:491–4.
-
Rowland RG, Mitchell ME, Bihrle R, et al. Indiana continent urinary reservoir. J Urol. 1987;137(6):1136–9.
-
Thüroff JW, Alken P, Engelmann U, et al. The Mainz pouch (mixed augmentation ileum ‘n zecum) for bladder augmentation and continent urinary diversion. Eur Urol. 1985;11(3):152–60.
-
Lilien OM, Camey M. 25-Year experience with replacement of the human bladder (Camey procedure). J Urol. 1984;132(5):886–91.
-
Bizzarri N, Foschi N, Loverro M, et al. Indocyanine Green to Assess Vascularity of Ileal Conduit Anastomosis During Pelvic Exenteration for Recurrent/Persistent Gynecological Cancer: A Pilot Study. Front Oncol. 2021;11:727725.
-
Rietjens M, Maggioni A, Bocciolone L, Sideri M, Youssef O, Petit JY. Vaginal reconstruction after extended radical pelvic surgery for cancer: comparison of two techniques. Plast Reconstr Surg. 2002;109(5):1592-7; discussion 1598-9.
-
Nilsson PJ. Omentoplasty in abdominoperineal resection: a review of the literature using a systematic approach. Dis Colon Rectum. 2006;49(9):1354–61.
-
Farrell RA, Maguire PJ, Freyne A, Siddiki S, Saadeh FA. Use of an artificial mammary implant in total pelvic exenteration. Int J Gynecol Cancer. 2021;31(8):1186–7.
-
Jacombs ASW, Rome P, Harrison JD, Solomon MJ. Assessment of the selection process for myocutaneous flap repair and surgical complications in pelvic exenteration surgery. Br J Surg. 2013;100(4):561–7.
-
van Ramshorst GH, Young JM, Solomon MJ. Complications and impact on quality of life of vertical rectus abdominis myocutaneous flaps for reconstruction in pelvic exenteration surgery. Dis Colon Rectum. 2020;63(9):1225–33.
-
Holm T, Ljung A, Haggmark T, Jurell G, Lagergren J. Extended abdominoperineal resection with gluteus maximus flap reconstruction of the pelvic floor for rectal cancer. Br J Surg. 2007;94(2):232–8.
-
Abdou AH, Li L, Khatib-Chahidi K, et al. Free latissimus dorsi myocutaneous flap for pelvic floor reconstruction following pelvic exenteration. Int J Colorectal Dis. 2016;31(2):385–91.
-
Donato D, Jarrell M, Averette H, Malinin T, Sevin B, Girtanner R. Reconstructive techniques in gynecologic oncology: the use of human dura mater allografts. Eur J Gynaecol Oncol. 1988;9(2):135–9.
-
Lee P, Tan WJ, Brown KGM, Solomon MJ. Addressing the empty pelvic syndrome following total pelvic exenteration: does mesh reconstruction help? Colorectal Dis. 2019;21(3):365–9.
-
Wang Y, Wang P. Novel technique with bladder peritoneum to prevent empty pelvic syndrome after laparoscopic pelvic exenteration for gynecologic malignancies: Three case reports. Medicine. 2021;100:49(e28200).
-
Lim PC. Robotic assisted total pelvic exenteration: a case report. Gynecol Oncol. 2009;115(2):310-311.
-
The PelvEx Collaborative. Minimally invasive surgery techniques in pelvic exenteration: a systematic and meta-analysis review. Surg Endosc. 2018;32(12):4707-4715.
-
Lopez M, Barrios L. Evolution of Pelvic Exenteration. Surg Oncol Clin N Am. 2005;14:587–606.





