Toxicitate cutanată excesivă la o pacientă care a primit doxorubicină lipozomală pegilată, asociată cu sindrom Nicolau, exacerbat de obezitatea morbidă
Excessive skin toxicity in a patient receiving pegylated liposomal doxorubicin, associated with Nicolau syndrome, exacerbated by morbid obesity
Abstract
Pegylated liposomal doxorubicin (PLD) is an anthracycline anticancer agent used in ovarian cancer and a form of doxorubicin enclosed in pegylated liposomes. Although it causes less cardiotoxicity than doxorubicin, it is frequently associated with cutaneous toxicity, mainly palmoplantar erythrodysesthesia. Other forms of skin toxicity – such as maculopapular rash, intertrigo-like dermatitis, psoriasiform dermatitis and radiation recall dermatitis – have also been described. Nicolau syndrome is a rare complication following the intramuscular administration of several agents, mainly injection of nonsteroidal anti-inflammatory drugs. Obesity is not only an established risk factor for developing cancer, but can also adversely influence the course of the disease in the advanced or metastatic setting. In this article, we present the case of a 44-year-old morbidly obese woman diagnosed with platinum-resistant stage IV ovarian cancer treated with pegylated liposomal doxorubicin in a second-line setting. The patient developed severe skin toxicity caused by pegylated liposomal doxorubicin, manifested by paronychia, intertrigo-like dermatitis with ulcerations, bleeding, infection, and psoriasiform dermatitis. She also presented with associated Nicolau syndrome, due to injection of antalgics into the subcutaneous fat instead of intramuscular. These skin toxicities caused further complications, such as severe anemia and acute renal failure. We hypothesize that the morbid obesity was the main contributing factor to all of these complications. With this presentation, we want to underline the importance of maintaining a healthy body weight in cancer patients.Keywords
skin toxicitypegylated liposomal doxorubicinNicolau syndromepsoriasiform dermatitisintertrigo-like dermatitisovarian cancerobesityRezumat
Doxorubicina lipozomală pegilată (PLD) este o antraciclină utilizată în tratamentul cancerului ovarian. Deşi prezintă mai puţină cardiotoxicitate decât doxorubicina, este frecvent asociată cu toxicitate cutanată, în principal eritrodisestezia palmo-plantară. Alte forme de toxicitate a pielii – cum ar fi erupţia maculopapulară, dermatita asemănătoare intertrigo, psoriaziformă, dermatita asemănătoare celei postiradiere etc. – au fost descrise. Sindromul Nicolau este o complicaţie rară, ca urmare a administrării intramusculare a mai multor agenţi, în principal antiinflamatoare nesteroidiene. Obezitatea nu este numai un factor de risc stabilit pentru dezvoltarea cancerului, dar poate influenţa negativ cursul bolii în stadiul avansat sau metastatic. În acest articol, prezentăm cazul unei paciente de 44 de ani, cu obezitate, diagnosticată cu stadiul IV de cancer al ovarului rezistent la platină, care a urmat tratament cu doxorubicină lipozomală pegilată în linia a doua. Pacienta a dezvoltat toxicitate severă a pielii cauzată de doxorubicina lipozomală pegilată, manifestată prin paroniechie, intertrigo, ulceraţii, sângerare, infecţie şi dermatită psoriaziformă. Pacienta a prezentat, de asemenea, sindromul Nicolau, în urma injectării de antalgice în grăsimea subcutanată în loc de injectarea intramusculară. Aceste toxicităţi ale pielii au provocat complicaţii suplimentare severe, cum ar fi anemie şi insuficienţă renală acută. Ipoteza noastră este că obezitatea morbidă a fost principalul factor care a contribuit la toate aceste complicaţii. Prin această prezentare, dorim să subliniem importanţa menţinerii unei greutăţi normale la pacienţii cu cancer.Cuvinte Cheie
toxicitate cutanatădoxorubicină lipozomală pegilatăsindromul Nicolaudermatită psoriaziformăintertrigocancer ovarianobezitateIntroduction
Pegylated liposomal doxorubicin (PLD) is a liposomal anthracycline-containing doxorubicin hydrochloride encapsulated in liposomes bound to their surface by methoxy polyethylene glycol (MPEG). This process is known as polyethylene, which protects the detection of liposomes by the phagocyte mononuclear system, thus increasing their circulation time in the blood. PLD was developed to reduce the risk of cardiotoxicity and myelosuppression experienced with conventional doxorubicin while maintaining anti-tumor efficacy. PLD causes lower cardiotoxicity by changing tissue distribution and decreasing the rate of drug release. Liposomes may not leave vascular space in areas of the narrow capillary junction, such as the heart muscle, but may reach tissues and organs that do not have narrow capillary junctions, such as tumor growth areas(1). One of the most common cutaneous adverse reactions reported in clinical studies for breast/ovarian cancer (50 mg/m2 at 4-week intervals) was palmoplantar erythrodysesthesia (PPE)(2). PPE – also called hand-foot syndrome – is characterized by painful papular macular rashes that include swelling, pain and sometimes skin peeling. PPE usually occurs within about 2 to 12 days after the administration of chemotherapy by the onset of dysesthesia or erythema and subsequently progresses to peeling and ulceration. Palms are most commonly affected(3). Other less common dermal manifestations would be intertrigo-like dermatitis, maculopapular rash, diffuse follicular rash, melanotic macules, and the recall phenomenon(4). Skin complications can affect the quality of life and may cause discontinuation of needle therapy which also leads to inefficiency of the therapy. In our review of the literature, we have encountered case presentations of various skin toxicities associated with pegylated liposomal doxorubicin, such as diffuse maculopapular rash associated with severe PPE(5), intertrigo-like eruptions(6,7), psoriasiform dermatitis(8) or radiation recall dermatitis(9). None of these, however, matched the severity of the current study.
Nicolau syndrome (NS) is also known as livedo-like dermatitis or embolia cutis medicamentosa, which is a rare complication following the intramuscular or intraarticular injection of different drugs. NS has been reported after the administration of various drugs, such as nonsteroidal anti-inflammatory drugs, penicillin corticosteroids and local anesthetics. It was described for the first time in literature in 1924 by Freudenthal, in patients treated with bismuth salts for syphilis(10,11). NS causes extremely severe pain at the injection site, with erythema occurring, followed by skin ulcers, or at the level of the muscular tissue which may develop until necrosis. The necrotic ulcer usually heals within a few months, with an atrophic scar. Transient neurological complications (hypoesthesia and paraplegia) have been described in one-third of the patients. Although several theories have been described in literature, the pathogenesis of NS is not fully understood, but it’s assumed to be due to arterial embolism, crystallization of drugs in blood vessels or local ischemia(11). NS has been described after attempted intramuscular administration of metamizole/dipyrone in obese patients, due to the fact that the drug was actually injected into the subcutaneous fat, causing necrosis(12). The treatment of NS varies from case to case, depending on the severity. In the early stages, debridement of the wound may be sufficient, while in the late stages corrective plastic surgery may be required. In the case of superinfected ulcers, antibiotic treatment is recommended.
The current study presents the case of a patient with metastatic ovarian cancer treated with pegylated liposomal doxorubicin who developed both skin toxicity and Nicolau syndrome, exacerbated by morbid obesity, and resulting in severe systemic complications.
Case report
We present the case of a 44-year-old woman diagnosed in October 2018 with stage IV ovarian cancer (pulmonary, pleural and peritoneal metastases), treated with pegylated liposomal doxorubicin in a second-line setting. The patient had previously received six cycles of paclitaxel-carboplatin chemotherapy and bevacizumab maintenance therapy in a first-line setting, but progressed radiologically and biochemically (CA-125) at five months after the last dose of platinum-based therapy, rendering her platinum-resistant. As comorbidities, the patient presented with idiopathic tachycardia (controlled with 50 mg metoprolol q.d.) and morbid obesity (height = 155 cm, weight = 125 kg, BMI = 52 kg/m2).
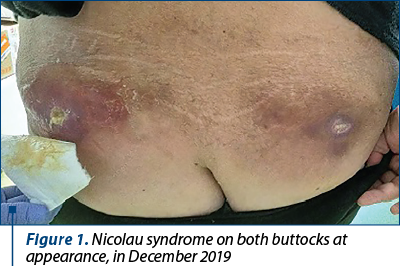
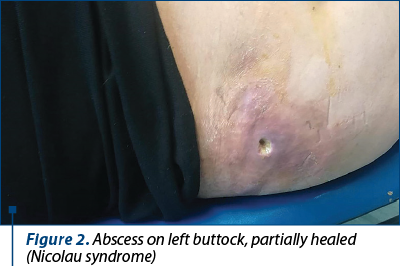

The second-line therapy with pegylated liposomal doxorubicin was started in December 2019. At chemotherapy initiation, the patient’s tumor marker CA-125 was 1361 U/mL (November 2019). The first cycle was very well tolerated, with no immediate toxicity. However, the patient complained about abdominal pain, presumably due to peritoneal metastases. Oral tramadol and Piafen® (metamizole/dipyrone + pitofenone + fenpipramide) were prescribed, but the patient refused this medication, claiming that it caused her abdominal discomfort, and decided to switch to a parenteral administration. The patient’s sister administered the tramadol subcutaneously and Algifen® intramuscular, three times daily. Within a week, the patient developed bilateral abscesses on the buttocks. These needed surgical debridement and antibiotic therapy (oral amoxicillin prescribed by the patient’s family physician) and led us to delay the second planned cycle of therapy with two weeks (Figures 1 and 2). The most likely cause of these abscesses was that the Algifen® injections were actually administered within the fatty tissue – due to the morbid obesity, the gluteal muscle was not reached. This type of complication has been previously described as Nicolau syndrome and has been seen in obese patients after injection of NSAIDS or metamizole/dipyrone(12). After the abscesses healed and the chemotherapy was resumed, the patient has been instructed to urgently lose weight and to avoid intramuscular injections at all costs. Despite the warning, the patient’s sister administered another injection of Algifen® in the patient’s right thigh, causing another abscess. In spite of the treatment delay, the disease seemed to respond to chemotherapy, as the tumor marker CA-125 decreased from 865 U/mL (December 2019) to 523 U/mL (January 2020). The third cycle of chemotherapy was planned for January, but was delayed three days due to a diarrheal syndrome (the patient did not come to the clinic at the scheduled date, only informing us of her diarrhea).

When she came to the clinic, the diarrheal syndrome was completely relieved, but she presented with extensive cutaneous lesions, as follows:
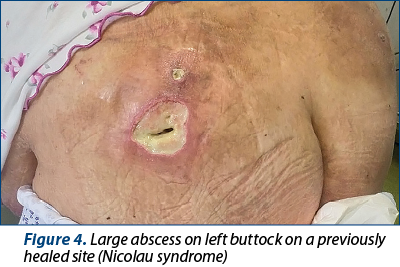
1. A new abscess on a previously healed site of the right buttock (presumed due to Nicolau syndrome) – Figures 3 and 4.
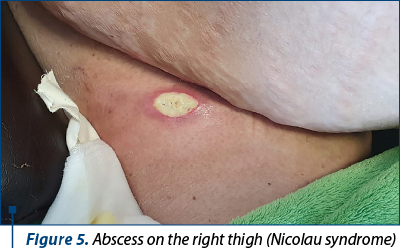
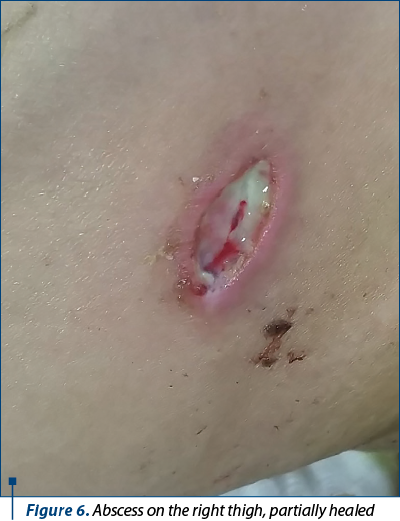
2. Abscess on the right thigh (presumed due to Nicolau syndrome) – Figures 5 and 6.
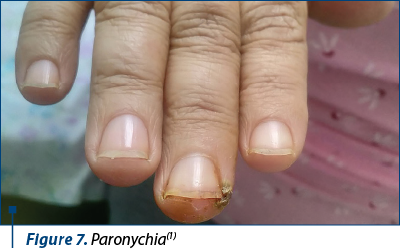
2. Paronychia – Figures 7 and 8.
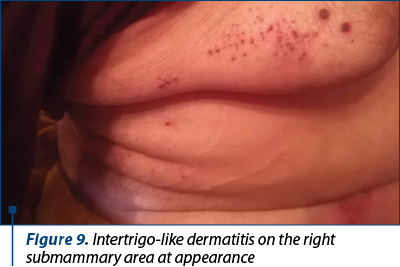
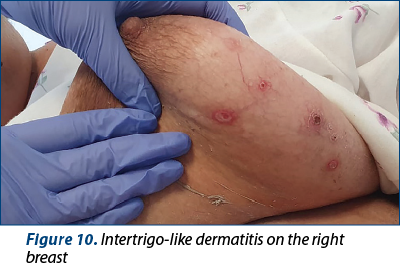
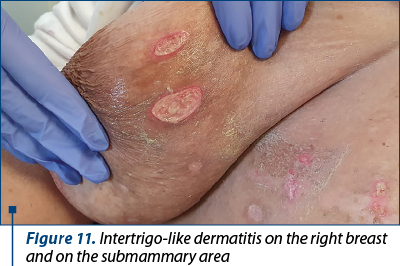
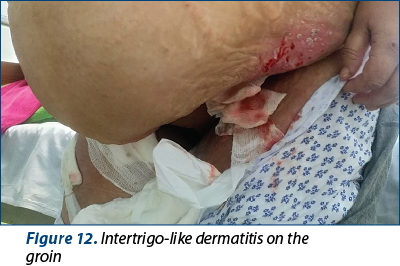
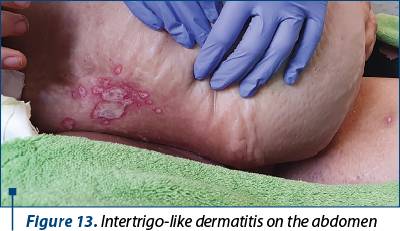
3. Intertrigo-like dermatitis with ulcerations, bleeding and infection in the following sites: right submammary area (Figures 9, 10 and 11), inguinal (Figure 12), under the abdominal skin flaps (Figure 13).
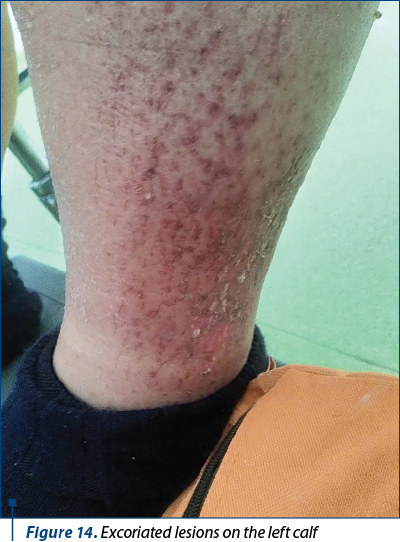
4. Excoriated lesions on the left calf (Figure 14).
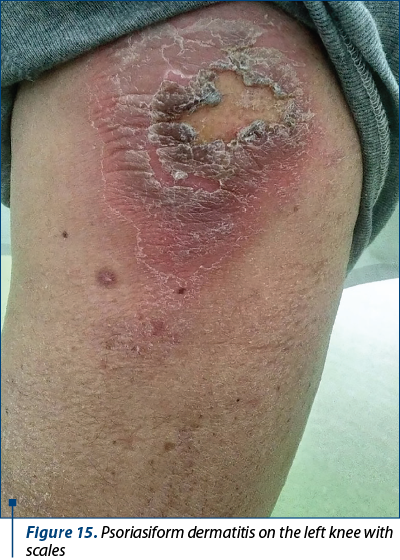
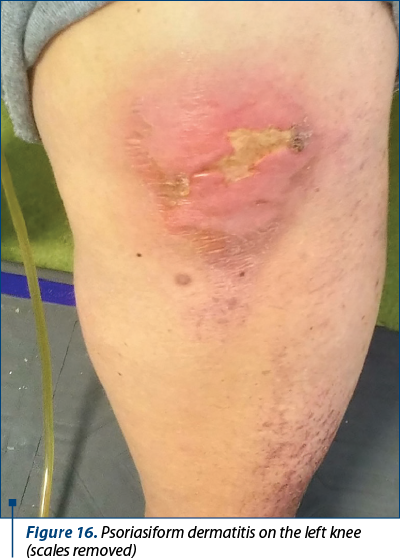
5. Psoriasiform dermatitis of the left knee (Figures 15 and 16).
Curiously, there was no sign of PPE, which is the most common cutaneous toxicity associated with PLD.
The patient mentioned that the lesions appeared about 10 days prior, preceded by intense burning and pruritus two days before they became visible. As complications of the infection, the patient also had severe anemia (Hb = 5.8 g/dl) and acute renal failure (creatinine: 2.42 mg/dL, associated with oliguria: 400 ml/24 hours). However, there were no signs of sepsis. The patient was apyretic and the blood cultures were sterile. Serum ferritin levels were measured and were found to be 9 times above the upper normal limit (2618.8 ng/mL; upper normal limit: 291 ng/mL), so the cause of the anemia is not iron deficiency. We determined it to be anemia of inflammation, a form of anemia that commonly develops in the setting of infection, inflammatory disease or malignancy, due to inefficient erythropoiesis combined with a shortened erythrocyte lifespan caused by enhanced erythrophagocytosis by cytokine-activated macrophages(13). The acute renal failure was most likely caused by dehydration in the context of diarrhea. The patient also admitted to having avoided oral hydration during the diarrheal episode, due to a misplaced belief that this would decrease the frequency of her watery stools. Despite the morbid obesity, the patient consistently had normal blood sugar levels and there is no suspicion of diabetes mellitus that would explain the poor healing of the skin lesions.
The skin lesions were treated with povidone-iodine ointment and tetracycline ointment, showing signs of improvement. The anemia was corrected via transfusion, hemoglobin levels rising to 8.4 mg/dL after one unit of packed red blood cells. The acute renal failure resolved after two days of intensive intravenous hydration and loop diuretics, creatinine levels decreasing to 0.53 mg/dL, and urine output returning to normal (2000 ml/24 hours). As of now, oncological treatment cannot be resumed as the multitude of cutaneous lesions are not healed yet.
Discussion
Intertrigo-like and psoriasiform dermatitis have been described before in relation to pegylated liposomal doxorubicin(6-8), but never in such a severe form, or leading to such devastating complications. We hypothesize that the main risk factor for this patient was morbid obesity, creating many skin flaps. Obesity in itself is a major risk factor for intertrigo(14) and intertrigo-like dermatitis. The morbid obesity also created mobility problems for the patient, interfering with local hygiene. Obesity is one of the most common metabolic diseases and is often associated with many other ailments, including many types of cancer, therefore it is regarded by the World Cancer Research Fund (WCRF) and the American Institute for Cancer Research (AICR) as a recognized risk factor for several cancers(15). In terms of ovarian cancer, obesity is associated with an increased risk of cancer and may even lower survival in advanced disease(16). Unfortunately, the general population often fails to perceive obesity as a risk factor for cancer(17). Conversely, cancer patients are usually much more worried about weight loss than weight gain(18). We firmly believe that the morbid obesity was the main risk factor leading to severe dermatological complications for this patient.
Conclusions
With the presentation of this case, we want to underscore the often neglected risks that morbid obesity poses for oncological patients and the way it exacerbates the dermatological toxicity of both anti-cancer agents and of antalgic treatments. Oncological patients are often neglecting weight-loss strategies, due to the common misconception that advanced cancer is more dangerously associated with weight loss(18). It is of paramount importance that cancer patients strive to maintain healthy body weight, irrespective of disease stage.
Acknowledgements. We would like to thank the patient for allowing us to publish her case.
Consent. The written informed consent was obtained from the patient for the publication of this case report and for any accompanying images. A copy of the written consent is available for review.
Conflicts of interests: The authors declare no conflict of interests.
Bibliografie
- Mangana J, Zipser MC, Conrad C, Oberholzer PA, Cozzio A, Knuth A, French LE, Dummer R. Skin problems associated with pegylated liposomal doxorubicin – more than palmoplantar erythrodysesthesia syndrome. Eur J Dermatol. 2008; 18:566–570.
- Green AE, Rose PG. Pegylated liposomal doxorubicin in ovarian cancer. Int J Nanomedicine. 2006; 1:229–239.
- Farr KP, Safwat A. Palmar-plantar erythrodysesthesia associated with chemotherapy and its treatment. Case Rep Oncol. 2011; 4:229–235, doi:10.1159/000327767.
- Cady FM, Kneuper-Hall R, Metcalf JS. Histologic patterns of polyethylene glycol-liposomal doxorubicin-related cutaneous eruptions. Am J Dermatopathol. 2006; 28:168–172, doi:10.1097/01.dad.0000199880.71481.0f.
- Kubicka-Wołkowska J, Kędzierska M, Lisik-Habib M, Potemski P. Skin toxicity in a patient with ovarian cancer treated with pegylated liposomal doxorubicin: A case report and review of the literature. Oncology Letters. 2016; 12(6):5332-5334.
- Totsuka M, Watanabe Y, Asai C, Takahashi S, Ishikawa H, Takamura N, et al. Case of severe bullous erythema including intertrigo‐like eruptions with angioedema induced by pegylated liposomal doxorubicin. The Journal of Dermatology. 2019; 46(6):535-539.
- Sánchez Henarejos P, Ros Martínez S, Marín Zafra G, Alonso Romero J, Navarrete Montoya A. Intertrigo-like eruption caused by pegylated liposomal doxorubicin (PLD). Clinical and Translational Oncology. 2009; 11(7):486-487.
- Kar S, Prasad K, Yadav N, Madke B. Liposomal doxorubicin-induced palmoplantar erythrodysthesia syndrome. Indian Dermatology Online Journal. 2015; 6(5):366.
- Jimeno A, Ciruelos E, Castellano D, Caballero B, Rodriguez-Peralto J, Cortés-Funes H. Radiation recall dermatitis induced by pegylated liposomal doxorubicin. Anti-Cancer Drugs. 2003; 14(7):575-576.
- Nischal KC, Basavaraj HB, Swaroop MR, Agrawal DP, Sathyanarayana BD, Umashankar NP. Nicolau syndrome: An iatrogenic cutaneous necrosis. J Cutan Aesthet Surg. 2009 Jul-Dec; 2(2): 92–95.
- Senel E. Nicolau syndrome as an avoidable complication. Journal of Family and Community Medicine. 2012; 19(1):52.
- Dadaci M, Altuntas Z, Ince B, Bilgen F, Tufekci O, Poyraz N. Nicolau Syndrome after Intramuscular Injection of Non-Steroidal Anti-Inflammatory Drugs (NSAID). Bosnian Journal of Basic Medical Sciences. 2015; 15(1).
- Nemeth E, Ganz T. Anemia of Inflammation. Hematology/Oncology Clinics of North America. 2014; 28(4):671-681.
- Metin A, Genç Dilek N, Gunes Bilgili S. Recurrent candidal intertrigo: challenges and solutions. Clinical, Cosmetic and Investigational Dermatology. 2018; Vol. 11:175-185.
- Vucenik I, Stains J. Obesity and cancer risk: evidence, mechanisms, and recommendations. Annals of the New York Academy of Sciences. 2012; 1271(1):37-43.
- Liu Z, Zhang TT, Zhao JJ, Qi SF, Du P, Liu DW, Tian QB. The association between overweight, obesity and ovarian cancer: a meta-analysis. Japanese Journal of Clinical Oncology. 2015 Dec; 45(12):1107-15.
- Soriano R, Ponce de León Rosales S, García R, García-García E, Méndez JP. High knowledge about obesity and its health risks, with the exception of cancer, among Mexican individuals. J Cancer Educ. 2012; 27:306-311.
- Zaleta A, McManus S, LeBlanc T, Buzaglo J. Perceptions of unintentional weight loss among cancer survivors. Journal of Clinical Oncology. 2018; 36(7 suppl):138-138.




