Cancerul laringian local avansat. Strategii pentru prezervarea laringelui
Locally advanced laryngeal cancer. Larynx preservation strategies
Abstract
Larynx preservation in locally advanced laryngeal cancer is intended to obtain cancer control and, if possible, the preservation of laryngeal anatomy and functions (speech, swallowing and airway patency). Eligibility criteria for larynx preservation strategies are medical and surgical. Medical factors include age, performance status, nutrition status and visceral functions. Surgical factors are local factors on which depends tumor resectability and post-treatment larynx functionality. Larynx preservation strategies include radiotherapy as local treatment associated with concomitant or sequential chemotherapy. Concomitant cisplatin-fluorouracil (CDDP-FU or PF) chemoradiation and docetaxel-cisplatin-fluorouracil (TPF) induction chemotherapy followed by radiation therapy increased larynx preservation and are evidence-based validated options. After TFP (docetaxel-CDDP-FU) induction chemotherapy, cetuximab-based immunotherapy with concomitant irradiation showed no superiority to concurrent cisplatin-based chemo-radiotherapy and, consequently, this combination should not be used, except for unfit patients to platinum-based regimen.Keywords
advanced laryngeal cancerchemotherapyradiotherapybiotherapyRezumat
Prezervarea laringelui în cancerul laringian local avansat are ca obiective obținerea controlului tumoral și, în același timp, prezervarea anatomică și funcțională a laringelui (fonație, deglutiție, respirație). Criteriile de eligibilitate sunt medicale și chirurgicale. Factorii medicali includ vârsta, statusul de performanță și cel nutrițional, precum și funcțiile viscerale. Factorii chirurgicali sunt factori locali de care depind rezecabilitatea tumorală şi funcționalitatea laringelui după tratament. Strategiile terapeutice de prezervare laringiană sunt reprezentate de radioterapie ca tratament local, asociată cu chimioterapia administrată concomitent sau secvențial. Chimioterapia concomitentă pe bază de cisplatin-fluorouracil (CDDP-FU sau PF) și chimioterapia de inducție cu docetaxel-cisplatin-fluorouracil (TPF) urmată de iradiere cresc rata de prezervare laringiană și reprezintă două opțiuni terapeutice validate. Folosită după chimioterapia de inducție de tip TPF, imunoterapia pe bază de cetuximab concomitent cu iradierea nu este superioară radiochimioterapiei concomitente și prin umare nu trebuie utilizată decât la pacienții la care nu se pot administra regimuri de chimioterapie pe bază de săruri de platină.Cuvinte Cheie
cancer laringian avansatchimioterapieradioterapiebioterapieThe main goals of locally advanced laryngeal cancer treatment are local control, survival and, if possible, the preservation of laryngeal anatomy and functions (speech, swallowing and airway patency). The first therapeutic option remains total laryngectomy with lymph node dissection followed by adjuvant radiotherapy, but alternative therapeutic strategies in order to preserve the larynx are developed(1). These include radiotherapy as local treatment associated in various combinations of different systemic therapy.
The criteria for larynx preservation
The eligibility criteria for larynx preservation strategies are medical and surgical.
The medical factors are age and general health status of the patient. These factors directly influence the radiotherapy and chemotherapy tolerance. A poor radiotherapy tolerance can cause discontinuation or even stopping the irradiation, as poor chemotherapy tolerance requires dose reduction. In both cases the patient receives a suboptimal treatment responsible for a lower tumor response, a situation frequently associated with salvage laryngectomy(2).
The medical factors are:
1. Age: most studies select patients aged under 70 years, but the exclusion of older patients remains controversial. Age should always be correlated with performance status(3).
2. Performance status (WHO≤2, that means ambulatory patients) is one of the main criteria in all trials.
3. Possible comorbidities, of which the most important are nutrition status and visceral functions.
3.1. Nutrition status is evaluated based on patients current Body Mass Index and recent (in the last 6 months) weight loss. Before and/or during treatment, enteral nutrition (nasogastric feeding tube or gastrostomy) may be necessary when oral nutrition becomes insufficient. Systematic prophylactic gastrostomy before preservation therapy could reduce weight loss and malnutrition during and after treatment, but can be responsible for significant complications (wound infection, minor bleeding, peristomal wound leakage, necrotizing fasciitis, colocutaneous fistula, peritonitis)(4,5). There is no consensus in clinical practice regarding the use of enteral nutrition during conservative treatment within larynx preservation protocols: some centers place feeding tubes prophylactically in all patients before treatment, and others wait until feeding tube placement is clinically indicated (wait and see policy)(6).
3.2. Visceral functions are related mainly to renal, hepatic, cardiovascular and pulmonary comorbidities, which can pose problems for optimal chemotherapy administration.
The surgical factors are local factors on which depends tumor resectability and post-treatment larynx functionality.
1. Post-chemotherapy tumor resectability depends on cartilage and soft tissues invasion, which in turn depends on the tumor response to chemotherapy (in vivo chemosensitivity). The earlier intrinsec tumors chemoresistance is identified, the faster are avoided the ineffective treatment and unsuccessful larynx preservation. A phase II study of the University of Michigan Comprehensive Cancer Center (UMCC) proved that clinical response after one cycle of induction chemotherapy is a feasible criterion for organ preservation. Thirty-six patients with T4 squamous cell carcinoma of the larynx (cartilage invasion) were evaluated by direct laryngoscopy (under anesthesia) after the first cycle of cisplatin (100 mg/m2) on day 1 and 5-fluorouracil (1,000 mg/m2/day for 5 days) chemotherapy. Twenty-nine of them (81%) presented more than 50% clinical response, so they are considered chemosensitive and are proposed for conservative treatment. For 7 patients (19%) the response was less than 50%, so they were regarded as nonsensitive and received total laryngectomy. Of the 29 chemosensitive patients, 27 received definitive chemoradiation, obtaining complete histological response in 23 (85%). The global rate of laryngeal preservation was 58%(7).
2. Laryngeal functionality depends on initial laryngeal interventions like tracheotomy and/or gastrotomy (gastric tube).
In a non-randomized prospective study on 49 patients with stage III/IV laryngeal cancer treated by concomitant cisplatin-based chemo-radiotherapy, those who had previous tracheotomy (12 patients) had a lower rate of complete response (41.7% vs. 75%, p=0.034 ) and overall survival (median survival 12 vs. 56 months, and 3-year survival rates 6% vs. 61%, p=0.001) than patients without tracheotomy. For the patients presenting with early stridor, to which pre-radiotherapy tracheostomy is indispensable, one solution is the induction chemotherapy. The chemotherapy can decrease tumor size and, at the same time, allows a correct evaluation of in vivo tumor chemosensitivity. In the same study, all the 8 out of 9 patients with pharingolaryngeal cancer (stage III or IV) who received platinium-taxane based chemotherapy (with or without 5-FU) had no more stridor after 48 hours. After induction chemotherapy, all patients except one received concomitant chemoradiation with cisplatine every week. The radiological response rate was 62.5% and the median reduction in size of tumor was 37%(8).
Larynx preservation probability TALK score
The probability of larynx preservation can be evaluated using the TALK score developed by Sherman et al. The score uses 4 parameters: T stage (≤T3 versus T4), serum albumin level (<4 versus >4 g/dl), alcohol/liquor use (<6 cans of beer/day versus >6 cans of beer/day) and Karnofsky Performance Status (<80% versus >80%), and is calculated by summing the number of the risk factors. A TALK score of 0 predicts a good response after larynx preservation, a 1-2 TALK score predicts intermediate risk for unsuccessful larynx preservation, and a TALK score equal or greater than 3 represents a formal contraindication for larynx preservation (high risk for unsuccessful larynx preservation). Correlated with TALK score, the 3-year larynx preservation rates are 65% for score 0; 41% for score1-2; and only 6% for score 3-4(9).
Chemotherapy protocols usually used in head and neck carcinoma
There are two chemotherapy protocols:
1. Cisplatin-fluorouracil (CDDP-FU or PF) chemotherapy consists of cisplatin (CDDP, 100 mg/m2) followed by a continuous 24-hour intravenous infusion of fluorouracil (5FU, 1000 mg/m2 per day) for 5 days, repeated every 21 days.
2. The TPF chemotherapy regimen consists of docetaxel 75 mg/m2, followed by intravenous cisplatin 100 mg/m2 and fluorouracil 1000 mg/m2 per day, administered as a continuous 24-hour infusion for 4 days. This schema was compared with classical scheme of cisplatin 100 mg/m2 and fluorouracil 1000 mg/m2 per day as a continuous 24-hour infusion for 5 days.
I. Optimal chemotherapy and radiotherapy secquence: induction chemotherapy followed by radiotherapy versus alterned chemoradiation versus concurrent chemoradiation
- Question: Which is the optimal sequence of administration of CDDP-FU chemotherapy in relation to radiation: induction or alternative?
Study: The European Organization for Research and Treatment of Cancer (EORTC) Phase 3 Randomized Trial 24954 .
Four hundred and fifty patients with advanced larynx (218 patients) or hypopharynx cancer were treated by different combination of chemo- and radiotherapy. In the sequential group patients received 4 cycles of chemoterapy followed by radiotherapy, and in the alternating group the patients received reduced dose chemotherapy (cisplatin 20 mg/m2 and 5-fluorouracil 200 mg/m2 per day, on days 1-5) in weeks 1, 4, 7, and 10, alternated with three 2-week courses of radiotherapy (20 Gy per course) that were administered in weeks 2 and 3, weeks 5 and 6, and weeks 8 and 9. Salvage surgery (total laryngectomy) was reserved for patients who did not achieve at least a partial response after the first two cycles of induction chemotherapy, for those who had residual disease 2 months after radiotherapy or in case of local recurrence. After 2 cycles of CDDP-FU, good responders (response >50%) received another two cycles of CDDP-FU followed by radiotherapy (sequential group, N= 224), or 4 cycles of CDDP-FU with radiotherapy administered between cycles of chemotherapy (alternating group, N= 226). At 3 years, 39.5% of patients in the sequential group and 45.4% in the concomitant group were alive, with a functional larynx in place. The estimated 5-year overall survival were 48.5% vs. 51.9% for the sequential versus alternating groups, respectively. Regarding toxicity, grade 3 or 4 acute mucositis occurred in 32% of patients in the sequential group versus 21% of patients in alternating group and the late severe edema and/or fibrosis was observed in 16% of patients in the sequential group versus 11% of patients in the alternating group(10).
Answer and conclusions: The optimal sequence seems to be the alternative one.
- Question: Which is the optimal sequence of administration of CDDP-FU chemotherapy in relation to radiation for laryngeal preservation: induction chemotherapy followed by radiation or concomitant chemoradiation?
Study: Radiation Therapy Oncology Group (RTOG) 91-11 study.
Five hundred and twenty patients with stage III or IV glottic or supraglottic squamous cell cancer were randomly assigned to induction cisplatin/fluorouracil followed by radiotherapy (control arm), concomitant cisplatin-based chemoradiation or radiotherapy alone. Salvage laryngectomy was performed for non-responder patients (less than a partial response after two cycles of induction chemotherapy), for patients with clinical progression or biopsy-proven persistent disease after completing radiotherapy and for subsequent recurrence. After a 10.8 years median follow-up, it was proved that both chemo-radiotherapy regimens significantly improved laryngectomy-free survival compared with radiotherapy alone but without any benefit on the 10-year overall survival. Concomitant chemoradiation significantly improved the larynx preservation rate over sequential chemo-radiotherapy. Regarding toxicity, the acute mucosal toxicity during irradiation was nearly twice as frequent for concomitant chemotherapy patients compared with the other two groups, but no difference in late effects was detected(11).
Answer and conclusions: Concomitant chemoradiation significantly improved the larynx preservation rate versus sequentially chemo-radiotherapy. This attitude is the standard of care in USA.
II. Optimal induction chemotherapy followed by radiotherapy
- Question : Does induction CDDP-FU chemotherapy followed by radiotherapy permit laryngeal conservation for responder patients?
Study: Veterans Affairs Laryngeal Cancer Study Group, prospective randomized trial.
Three hundred and thirty-two patients with advanced (stage III or IV) laryngeal squamous cell carcinoma were evaluated after two cycles of CDDP-FU induction chemotherapy and the responders (more than 50% reduction in the sum of the longest tumor dimension and no progression of any neck adenopathy) received a third cycle of chemotherapy and definitive radiation, while the non responders underwent immediate total laryngectomy and neck dissections followed by radiation therapy. Overall survival was identical (68% survival at 2 years) for both groups, but total larynngectomy was superior in terms of local control (the recurrence rate was 2% in the surgery group versus 12% in the chemotherapy group). It is evidenced that larynx was preserved in 64% of patients assigned to induction chemotherapy. Salvage laryngectomy for recurrent cancer was performed on 11 patients(12).
Answer and conclusions: Induction CDDP-FU chemotherapy permits larynx preservation for the good responder patients.
- Question: Does adding docetaxel to CDDP-FU induction chemotherapy increase laryngeal preservation?
Study: Groupe d’Oncologie Radiothérapie Tête Et Cou (GORTEC) 2000-01 study.
Two hundred and thirteen patients with untreated stage III or IV larynx or hypopharynx invasive squamous cell carcinoma received three cycles of induction cisplatin-fluorourcil with or without docetaxel chemotherapy followed by radiation therapy for responders. Long-term follow-up confirms that adding docetaxel to cisplatin-fluorouracil induction chemotherapy increased larynx-preservation (74% vs. 58.1% at 5 years and 70.3% vs. 46.5% at 10 years, p=0.01) and larynx dysfunction-free survival (67.2% vs. 46.5% at 5 years and 63.7% vs. 37.2% at 10 years, p=0.001). Overall survival, disease-free survival, and locoregional control rates were not statistically improved in the TPF vs. the PF arm. From the toxicity point of view, the TPF combination induced more grade 2 alopecia (19.4% vs. 2%) and more neutropenia (31.5% vs. 17.6%), including febrile neutropenia (10.9% vs 5.8%), while the PF combination is responsible for more grades 3 and 4 stomatitis (7.8% vs. 4.6%), thrombocytopenia (7.8% vs. 1.8%), and renal dysfunction (creatinine elevation 2% vs. 0%)(13).
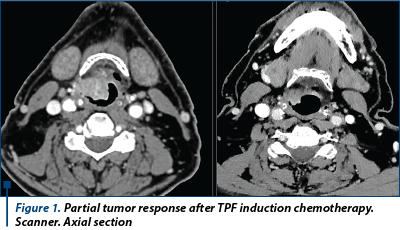
Answer: Docetaxel-cisplatin-fluorouracil (TPF) induction chemotherapy followed by radiation therapy increased larynx-preservation and larynx dysfunction-free survival and should be recommended for medically fit patients as a standard of care.
III. Induction chemotherapy followed by radio-chemotherapy
- Question: Is TPF induction chemotherapy superior to induction PF chemotherapy ?
Study: TAX 324 open-label phase 3 trial.
One hundred and sixty-six patients with locally advanced stage III-IV laryngeal or hypolaryngeal cancer were randomized to receive TPF versus PF induction chemotherapy followed by weekly carboplatin concomitant radiochemotherapy. After a median follow-up of 72.2 months, it was found that TPF induction chemotherapy was superior to classical PF in terms of overall survival (5-year OS 52% versus 42%, and median survival 70.6 months versus 34.8 months) and progression-free survival (median 38.1 months vs. 13.2 months)(14).
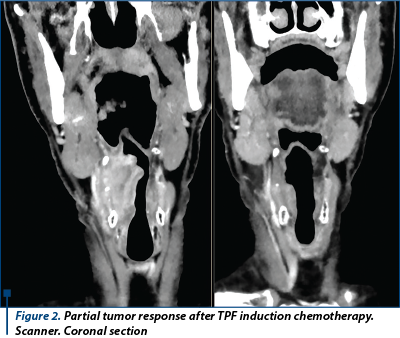
Answer: Induction chemotherapy with TPF provides long-term survival benefit compared with PF in locally advanced head and neck cancer. Patients who are candidates for induction chemotherapy should be treated with TPF.
IV. Induction chemotherapy followed by concurrent bio-radiotherapy
- Question: After TFP (docetaxel-CDDP-FU) induction chemotherapy, is concurent bioradiotherapy superior (benefit/tolerance) to concurent cisplatin-based chemoradiotherapy?
Study: TREMPLIN.
One hundred and sixteen previously untreated patients with stage III to IV larynx/hypopharynx squamous cell carcinoma received three cycles of TPF induction chemotherapy. The poor responders (<50% response) underwent salvage surgery, while the responders (>50% response) were randomly assigned to concurrent cisplatin+RT (arm A, 60 patients) or concurrent cetuximab+RT (arm B, 56 patients).
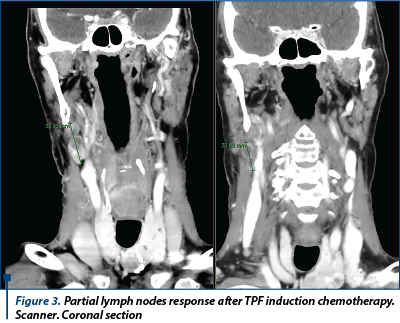
Cetuximab (Erbitux) is a recombinant chimeric monoclonal antibody that binds to the human epidermal growth factor receptor and blocks the activation of receptor-associated kinases, which results in cell growth inhibition, induction of apoptosis, and decreased vascular endothelial growth factor production. With concurrent radiation, the initial dose of cetuximab is 400 mg/m2 i.v. administrated one week before irradiation started, followed by 250 mg/m2 i.v. weekly. There was no significant difference in larynx preservation (95% vs. 93%), larynx function preservation (87% vs. 82%) at 3 months and overal survival at 18 months (92% vs. 89%) between cisplatin-radiotherapy and cetuximab-radiotherapy groups(15).
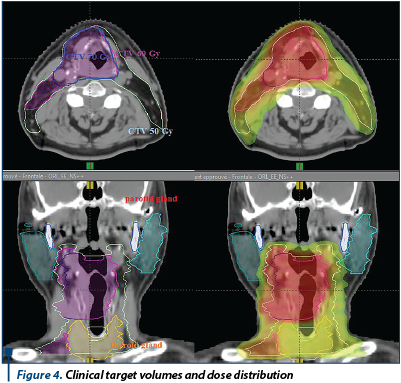
Answer and conclusion: There is no evidence that one treatment was superior to the other. The best regimen following induction TPF chemotherapy is still to be determined.
Post radio-chemotherapy follow-up
The initial assessment after radiotherapy should occur between 2 and 3 months after the last day of radiotherapy and has to evaluate two major criteria:
1. Local control. It is evaluated by imaging methods (Computed Tomography, Magnetic Resonance Imaging or Positron Emission Tomography) correlated with endoscopy. Routine biopsy is not recommended.
2. Laryngeal/pharingeal function. Baseline assessment of vocal cord fixation and swallowing function (e.g., a barium esophagram) should be performed.
Related to potential chemotherapy (platinum molecules) toxicity, hearing and renal dysfunction should be assessed at 6 months after the end of treatment(16).
Conclusions and recommendations
Larynx preservation is possible and must be proposed to selected patients who meet eligibility medical and surgical criteria for this strategy. Several randomized clinical trials have proved that concomitant chemoradiation significantly improved the larynx preservation rate versus sequential chemo-radiotherapy and therefore this attitude became the standard of care in USA. However, docetaxel-cisplatin-fluorouracil (TPF) induction chemotherapy followed by radiation therapy increased larynx preservation and larynx dysfunction-free survival and should be recommended for medically fit patients as a standard of care. Nowadays, there is no evidence that concurrent bioradiotherapy is superior to concurrent cisplatin-based chemo-radiotherapy, and this combination should not be used, except for unfit patients to platinum-based regimen.
Bibliografie
1. LefebvreJL, Ang KK. Larynx Preservation Consensus Panel Larynx preservation clinical trial guidelines, Head Neck, 31 (April (4) (2009), pp. 429-441.
2. Denaro N, Russi EG, Lefebvre JL, Merlano MC. A systematic review of current and emerging approaches in the field of larynx preservation. Radiother Oncol. 2014 Jan; 110(1):16-24.
3. K. Kian Ang, Larynx Preservation Clinical Trial Design: Summary of Key Recommendations of a Consensus Panel, The Oncologist 2010;15 Suppl 3:25-9. doi: 10.1634/theoncologist.2010-S3-25.
4. Silander E, Nyman J, Bove M, Johansson L, Larsson S, Hammerlid E. Impact of prophylactic percutaneous endoscopic gastrostomy on malnutrition and quality of life in patients with head and neck cancer: a randomized study. Head Neck. 2012 Jan; 34(1):1-9. doi: 10.1002/hed.21700. Epub 2011 Mar 3.
5. Atasoy BM, Yonal O, Demirel B, Dane F, Yilmaz Y, Kalayci C, Abacioglu U, Imeryuz N. The impact of early percutaneous endoscopic gastrostomy placement on treatment completeness and nutritional status in locally advanced head and neck cancer patients receiving chemoradiotherapy. Eur Arch Otorhinolaryngol. 2012 Jan;269(1):275-82. doi: 10.1007/s00405-010-1477-7. Epub 2011 Apr 7.
6. Bozec A, Benezery K, Chamorey E, Ettaiche M, Vandersteen C, Dassonville O, Poissonnet G, Riss JC, Hannoun-Lévi JM, Chand ME, Leysalle A, Saada E, Sudaka A, Haudebourg J, Hebert C, Falewee MN, Demard F, Santini J, Peyrade F. Nutritional status and feeding-tube placement in patients with locally advanced hypopharyngeal cancer included in an induction chemotherapy-based larynx preservation program. Eur Arch Otorhinolaryngol. 2016 Sep; 273(9):2681-7. doi: 10.1007/s00405-015-3785-4. Epub 2015 Sep 22.
7. Francis P. Worden, Jeffrey Moyer, Julia S. Lee, Jeremy M. G. Taylor, Susan G. Urba, Avraham Eisbruch, Theodoros N. Teknos, Douglas B. Chepeha, Mark E. Prince, Norman Hogikyan, Amy Anne D. Lassig, Kevin Emerick, Suresh Mukherji, Lubomir Hadjiski, Christina I. Tsien, Tamara H. Miller, Nancy E. Wallace, Heidi L. Mason, Carol R. Bradford, and Gregory T. Wolf. Chemoselection as a Strategy for Organ Preservation in Patients with T4 Laryngeal Squamous Cell Carcinoma with Cartilage Invasion, Laryngoscope. 2009 Aug; 119(8): 1510–1517.
8. Vijay Maruti Patil, Vanita Noronha, Amit Joshi, Vamshi Muddu, Bhavesh Poladia, Bharat Chauhan, Kumar Prabhash, Devendra Arvind Chaukar, Pankaj Chatturvedi, Gouri Pantvaidya, Shashikant Juvekar, Anil D’cruz, Induction Chemotherapy in Locally Advanced Pharyngolaryngeal Cancers with Stridor: Is It Feasible and Safe? Chemother Res Pract. 2012; 549170. doi: 10.1155/2012/549170. Epub 2012 Aug 12.
9. Eric J Sherman, Susan G. Fisher, Dennis H. Kraus, Michael J Zelefsky, Venkatraman E. Seshan, Bhuvanesh Singh, Ashok R Shaha, Jatin P Shah, Gregory T Wolf, David G Pfister. TALK score: Development and validation of a prognostic model for predicting larynx preservation outcome, The Laryngoscope, Volume 122, Issue 5, May 2012, 1043–1050.
10. JL Lefebvre, F Rolland, M Tesselaar, E Bardet, CR Leemans, L Geoffrois, P Hupperets, L Barzan, D de Raucourt, D Chevalier, L Licitra, F Lunghi, R Stupp, D Lacombe, J Bogaerts, JC Horiot, J Bernier, JB. Vermorken, and for the EORTC Head and Neck Cancer Cooperative Group and the EORTC Radiation Oncology Group, Phase 3 Randomized Trial on Larynx Preservation Comparing Sequential vs. Alternating Chemotherapy and Radiotherapy, J Natl Cancer Inst. 2009 Feb 4; 101(3): 142–152.
11. AA Forastiere, Q Zhang, RS Weber, et al. Long-term results of RTOG 91-11: a comparison of three nonsurgical treatment strategies to preserve the larynx in patients with locally advanced larynx cancer. Journal of Clinical Oncology, 31 (2013), pp. 845–852.
12. The Department of Veterans Affairs Laryngeal Cancer Study Group. Induction Chemotherapy plus Radiation Compared with Surgery plus Radiation in Patients with Advanced Laryngeal Cancer.N Engl J Med 1991; 324:1685-169.
13. Janoray G, Pointreau Y, Garaud P, Chapet S, Alfonsi M, Sire C, Jadaud E, Calais G. Long-term Results of a Multicenter Randomized Phase III Trial of Induction Chemotherapy With Cisplatin, 5-fluorouracil, ± Docetaxel for Larynx Preservation, J Natl Cancer Inst. 2015 Dec 16; 108(4).
14. Posner MR, Norris CM, Wirth LJ, Shin DM, Cullen KJ, Winquist EW, Blajman CR, Mickiewicz EA, Frenette GP, Plinar LF, Cohen RB, Steinbrenner LM, Freue JM, Gorbunova VA, Tjulandin SA, Raez LE, Adkins DR, Tishler RB, Roessner MR, Haddad RI, TAX 324 Study Group. Sequential therapy for the locally advanced larynx and hypopharynx cancer subgroup in TAX 324: survival, surgery, and organ preservation., Ann Oncol 2009 May; 20(5):921-7.
15. Jean Louis Lefebvre, Yoann Pointreau, Frederic Rolland, Marc Alfonsi, Alain Baudoux, Christian Sire,Dominique de Raucourt, Olivier Malard, Marian Degardin, Claude Tuchais, Emmanuel Blot, Michel Rives, Emile Reyt, Jean Marc Tourani, Lionel Geoffrois, Frederic Peyrade, Francois Guichard, Dominique Chevalier, Emmanuel Babin, Philippe Lang, Francois Janot, Gilles Calais, Pascal Garaud, Etienne Bardet. Induction Chemotherapy Followed by Either Chemoradiotherapy or Bioradiotherapy for Larynx Preservation: The TREMPLIN Randomized Phase II Study, Journal of Clinical Oncology 2013 January, vol 31, nr 7, 833-845.
16. Lefebvre JL, Ang KK; Larynx Preservation Consensus Panel. Larynx preservation clinical trial design: key issues and recommendations--a consensus panel summary. Head Neck. 2009 Apr; 31(4):429-41.

